peeterv/iStock/Getty Images
Modern marvels
Of all the science headlines of the 1920s, “Ice Cream from Crude Oil” may best capture the era’s unbridled enthusiasm for chemistry. Edwin E. Slosson, the first head of the news service that would become Science News, reported that fats can be made by breaking up and rearranging the molecules of mineral oil. Synthetic ice cream was just one of the wonders that could lie around the corner.

Slosson went on to describe the potential to create aromatics, flavorings, nitroglycerine for dynamite, plastics, drugs and dyes “of infinite variety.” Petroleum would become increasingly valuable “as a source of substances for which man has hitherto been dependent upon the chance bounty of nature.”
Petroleum-based ice creams never became the new it thing, yet Slosson’s reporting was on point. The last century has witnessed a dramatic leap in the ability to study and synthesize matter. From our homes and cities to our electronics and clothing, much of what we interact with every day is made possible through the manipulation, recombination and reimagination of the basic substances nature has provided.

“The world is unrecognizable from 100 years ago,” says Anna Ploszajski, a materials scientist and author of Handmade: A Scientist’s Search for Meaning Through Making. And that, she says, is “simply because of the materials that we have around, let alone all of the new ways we use them.”
The turn of the 20th century set the stage for a new era of materials. Organic chemists learned how to turn coal into a variety of industrial chemicals, including dyes and perfumes. Later, motivated by wartime demand, chemists honed their craft with poison gas, explosives and propellants, as well as disinfectants and antiseptics —so many new products that World War I was often called “the chemist’s war.” And at a fundamental level, the new century also ushered in greater understanding of chemical bonds and the atom, its constituents and its behavior.
In the decades that followed, approaches in chemistry and physics combined with engineering to give rise to a new field, now called materials science. A boost from the U.S. government, which was seeking new materials for space and military technologies during the Cold War, led to academic departments that brought together physicists, chemists, metallurgists and electrical engineers — anyone interested in materials.

An extensive survey of the field, put together by the National Academy of Sciences in the 1970s and titled Materials and Man’s Needs, defined materials as “substances having properties which make them useful in machines, structures, devices and products.” Here’s how the report described the pace of research: “The transitions from, say, stone to bronze and from bronze to iron were revolutionary in impact, but they were relatively slow in terms of the time scale. The changes in materials innovation and application within the last half century occur in a time span which is revolutionary rather than evolutionary.”
Alongside this new science came new and improved scientific tools. Scientists can now see materials at a much finer scale, with the electron microscope making individual atoms visible. X-ray crystallography unveils atomic arrangements, allowing for a better understanding of materials’ structure. And equipment such as chromatographs and mass spectrometers let today’s scientists untangle mixtures of chemicals and identify the compounds within. Francis Aston first took advantage of a mass spectrometer in his study of isotopes in 1919, but for a long time the tool was seen by some chemists as, according to a description by mass spectrometrist Michael Grayson, “an unexplainable, voodoo, black magic kind of a tool.”
Many new materials were birthed from basic curiosity and serendipity. But new techniques also made way for targeted innovation. Today materials can be designed from scratch to solve specific problems. And explorations of the properties of solid substances — for instance, how matter interacts with heat, light, electricity or magnetism — along with iterations of design have further contributed to the stuff that surrounds us, giving way to transistors, transition lenses, touch screens and hard disk drives. Explorations into how matter interacts with biological tissues have yielded coronary stents, artificial skin and also hip replacements, including metal mélanges that are tough and nonreactive when they sit against bone.
“Materials science and engineering over the millennia have gone from ‘What do you find outside the cave?’ to the beginnings of materials-by-design,” Stephen H. Carr of Northwestern University told Science News in the 1980s.

The outputs of such efforts are all around us. Take air travel, for example, and the global interconnectivity it introduced. It’s possible thanks to alloys that are lightweight and robust. And today’s personal connectivity — via smartphones and computers — came with transistors made of silicon. Their small size and low power requirements brought computing to work, and then into our homes and pockets. An abundance of plastic housewares and comfy athleisure options are made possible via improvements in polymers.
The examples below highlight just a handful of ways that advances in materials over the last century have transformed our daily lives — we’ve focused on travel, connectivity and convenience. We could have chosen countless others.
Yet innovation hasn’t come without consequences. For each tale of progress, there are also stories of the marks people have left on this planet. While enabling humans to flourish, many new substances have become pollutants, from PCBs to plastics. However people go about addressing these environmental problems, other new materials will inevitably play a role in the solutions.
— Carolyn Wilke
How we move
It was the summer of 1940, the early days of the Battle of Britain. Nazi Germany’s air force, the Luftwaffe, began a months-long attack on the British Isles that eventually included the nightly bombing raids known as the Blitz. Going into the battle, the Luftwaffe believed it had the upper hand; in battles in France, the Germans had dominated in the air. Little did they know the Allies had a secret weapon — and it was in their fuel tanks.

As Germans began flying over England, they were surprised to find the tables had turned. One important factor was not the planes themselves, but the octane rating of the fuel they ran on. The British Spitfires and Hurricanes that the Germans had outmaneuvered in France could now climb higher and fly faster thanks to fuel made with a newly developed process called catalytic cracking.
Catalysts boost chemical reactions by reducing the energy needed to get them going. French mechanical engineer Eugene Houdry had developed a catalytic process in the late 1930s to make high-octane fuel, which can withstand higher compression and allows engines to deliver more power. Simply increasing the octane rating of aviation fuel from 87 to 100 gave the Allies a crucial edge.
Houdry wasn’t the first to attempt to use catalysts to bust the big molecules of heavy fuels into smaller ones to improve performance. But as an avid road racer, he had a special interest in high-quality gasoline. He studied hundreds of catalysts until he landed on aluminum- and silicon-based materials that could do the busting more efficiently than an existing process that relied on heat. When he tested his gasoline in his Bugatti racer, he reached speeds of 90 miles per hour.
Shortly after catalytic cracking was first demonstrated at industrial scale in 1937, Science News Letter, the predecessor of Science News, described it as “a radically new refining process held to be of the greatest importance to the entire petroleum industry.” By then Houdry had moved to the United States and joined Socony-Vacuum Oil and Sun Oil to build pilot plants. The first large-scale “Houdry unit” began operating at Sun Oil’s Marcus Hook refinery in Pennsylvania, and units multiplied from there.

In the following decades, catalytic cracking and improvements to the process Houdry pioneered would contribute to the reign of automobiles, and catalytic cracking still produces much of the gas that cars guzzle today.
But all that driving soon began to take a toll on the environment. When the hydrocarbon molecules in gasoline burn, the engine exhaust contains small amounts of harmful gases: poisonous carbon monoxide, nitrogen oxide that can cause smog and acid rain, as well as unburned hydrocarbons. With so many vehicles hitting the road, cities such as Los Angeles choked on smog in the 1940s and ’50s.
Houdry, after helping to fuel those vehicles, looked again to catalysts to deal with the pollution that internal combustion engines caused. He designed a catalytic converter.
“When first considered, the problem seems simple,” Houdry wrote in a 1954 patent application. “A great number of catalysts can be used for the reaction. By simply placing one of these catalysts in the exhaust line under controlled conditions, the exhaust fumes can be cleaned.” The catalysts, precious metals such as platinum or palladium, provide docking sites for the harmful gases to hang onto; there, reactions involving oxygen convert them to less harmful forms. But, as Houdry pointed out, the high temperatures, mechanical shock and other conditions in an engine complicate the problem.
In the 1950s, Houdry set out his solution, outlining a series of reactions, materials and conditions necessary for a working catalytic converter. But he was ahead of his time. For years, the adoption of catalytic converters in automobiles was stymied by leaded gasoline, which improved octane ratings but gummed up the catalysts’ surfaces. Within a few decades of the passage of the Clean Air Act of 1970, which led to requirements for catalytic converters and lead-free fuel, the devices helped clear the air in cities around the world.
Taking to the air
Air travel for the masses encountered a different dilemma: lightening the load. The earliest airplanes gained lift at the turn of the 20th century on wings of fabric and wood, but to really soar, airplanes needed light but strong materials. The first aircraft designed for passengers, the Ford Trimotor — nicknamed the Tin Goose — took to the air in 1926 with help from aluminum alloys.

Alloys have existed since ancient times. Bronze Age artisans combined copper with arsenic or tin in crucibles to make tools, jewelry and more. From there, advances coincided with the ability to melt metals at higher and higher temperatures, eventually leading to steel. Scientists since have studied how materials’ structures and properties — including desirable features like strength, bendability and resistance to corrosion — vary with composition, temperature and processing.
The fuselage of the Tin Goose contained a newly developed alloy named duralumin, a contraction of “Dürener” (for the company that originally made it) and “aluminum.” Developed by Alfred Wilm, a German metallurgist seeking a lighter alloy that could replace brass in ammunition cartridges, duralumin combined aluminum with copper, magnesium and manganese and got its strength from a series of heat treatments.
After adjusting the amount of magnesium in his mixture one day in 1906, Wilm left for the weekend, charging his assistant to test the hardness again on Monday. To their surprise, they discovered a new, never-before-seen feature of some alloys: Simply sitting at room temperature, the material had gotten harder as it sat.
In 1926, Science News Letter described the promise of materials such as duralumin for safer dirigibles, which would carry large numbers of passengers into the air: “Of these sound materials, strong and light girders must be built. So light that a man can carry one of them in his hand and yet so strong that they will carry loads of thousands of pounds.” The rigid frame of the Hindenburg, for example, was made of duralumin.

But dirigibles and duralumin were just the beginning. The 20th century has seen an explosion in types of alloys and their applications, from stainless steel cutlery to the titanium alloys used in prosthetics and pacemakers to crucial components of vehicles. Today’s jet engines are built of superalloys, which can withstand infernal temperatures.
Plastics and composites have also helped planes shed weight. While alloys melt metals and sometimes other types of elements together, composites combine materials with very different properties — such as glass and plastic — by suspending one in the other or sandwiching them together, for instance. Because they can be tuned to be light and strong, composites have made their way into parts all over planes, from the engine to the wings. Boeing’s 787 Dreamliner, which debuted in 2007, is made up of 50 percent composites by weight.
Composites also show up all over today’s cars, from dashboards to bumpers, with various materials glued together to better protect driver and passengers. “The incredible use of adhesives in cars to create crumple zones so that the passenger compartment is safer,” for example, is “directly out of materials,” says Mark Jones, a chemist and member of the National Historic Chemical Landmarks committee of the American Chemical Society.
And to space
When the Apollo 11 astronauts reentered Earth’s atmosphere after their historic moonwalk in 1969, the outer surface of their module skyrocketed to somewhere around 5,000° Fahrenheit, hot enough to incinerate any living thing. But inside, the astronauts sat at comfortable temperature around 70° F or so, protected by a heat shield made of a material called Avcoat.
Avcoat is a type of reinforced plastic, made from an epoxy resin filling a honeycomb fiberglass network. A layer of it, which varied in thickness from 0.5 to 3 inches, dissipated heat away from the command module’s metal components, keeping temperatures inside from rising.
Research advances that led to alloys and composites, as well as plastics — many of which ended up in cars and planes — got a boost from a desire to get to space. Spacefaring vehicles have to be not only strong and lightweight, but also withstand extreme temperatures.
Spotlight
Core memory weavers and Navajo women made the Apollo missions possible
The stories of the women who assembled integrated circuits and wove core memory for the Apollo missions remain largely unknown.
A new NASA spaceship called Orion, destined to take people to the moon and beyond, again uses Avcoat. But engineers have come up with a far more efficient process to make it. Instead of injecting Avcoat individually into each fiberglass cell, the Avcoat material is now made in pieces, which are machined to size before they are bonded onto a heat shield more than 16 feet in diameter. An Orion test mission to the moon could launch in early 2022.
Space materials push composites to the max. One extreme composite is Nextel. Sheets of it, woven of the ceramic fiber and Kevlar, another materials innovation, protect the International Space Station from debris and meteoroids. In other cases, materials invented for air and space travel have stuck around on Earth. Memory foam, developed in the late 1960s to cushion pilots and passengers during takeoff and landing, is now better known for its use in mattresses. And space blankets, which arrived that same decade to protect electronics from drastic swings of temperature, show up in emergency kits and are donned by marathoners after a race.
— Carolyn Wilke

How we connect
For a testament to the power of materials to connect us, just look at an iPhone. “The iPhone contains about 75 elements from the periodic table — a huge proportion of all the atoms that we know about in the universe are in an iPhone,” says Anna Ploszajski, a materials scientist and author of Handmade: A Scientist’s Search for Meaning through Making.

Some of those are rare-earth elements, a set of 17 metallic elements mostly on the outskirts of the periodic table. Though they are difficult to mine and process, rare earths are sought after because they lend unusual magnetic, fluorescent and electrical properties to materials made from them. Neodymium, for example, mixed with other metals makes the strongest magnets known — these magnets make your cell phone vibrate and its speakers produce sound.
Despite the challenges and environmental hazards associated with mining them, these elements show up in a lot of other 20th century applications, too. Rare earths are in color televisions, camera lenses, fiber-optic cables, nuclear reactors, nickel-metal hydride batteries, aircraft engines, PET scanners and much more.
Tiny switches
A more familiar element made widespread cell phones and other personal computing possible – silicon.
As a semiconductor, silicon conducts electricity better than materials such as ceramics and glass do, but not as well as metals. This in-between status makes it possible to control how electrons zip around a semiconductor, a control that’s ideal for creating electrical switches for circuits in radios, televisions or computers. In the 1930s and 1940s these and other electronic devices relied on bulky, breakable glass vacuum tubes that weren’t all that reliable. Decades of semiconductor research pointed to another, slimmer way.
The first semiconductor switch, dubbed the transistor, was made of germanium and invented at Bell Laboratories in 1947. But teams at Texas Instruments and Bell Labs were both eyeing silicon instead. Silicon holds up under higher temperatures (the temperatures that might show up in military applications — in a rocket, for example). Silicon is also less likely than germanium to leak current when a switch is supposed to be off. But achieving silicon success depended on the creation of high-purity forms and, as with germanium transistors, the ability to tune its electrical properties by incorporating other atoms.

Though the two teams independently developed silicon transistors, Texas Instruments’ Gordon Teal gets the credit as his announcement came first, in May of 1954.
At a conference in Dayton, Ohio, toward the end of the day’s talks, Teal matter-of-factly revealed his company’s success. “Contrary to what my colleagues have told you about the bleak prospects for silicon transistors,” he said, “I happen to have a few of them here in my pocket.” His announcement, which followed other talks suggesting that the devices were years away, jolted the audience, which stampeded to the back of the room for copies of Teal’s talk, and out to the telephone booth to share the news.
Strung together
Our attention-sucking phones are right in front of our faces. But out of sight are the fiber optics that relay messages around the world in a flash.
All the glass strung out in the world’s optical cables could tether Earth to Uranus and then some, stretching some 4 billion kilometers. These cables ferry messages across countries and continents and across the seafloor. Optical fiber “really has strung the world together in a new way,” says Ainissa Ramirez, a materials scientist and author of the book The Alchemy of Us. Messages from across the Atlantic used to come by boat, she says, then came copper cables to relay telegraph dispatches in the 1840s. The first live telephone traffic sent through fiber-optic cables was in 1977 in Long Beach, Calif. Now e-mails from abroad arrive nearly instantaneously thanks to thin-as-hair optical fibers. People “have a new relationship with the dispatches that we get as a result of technology,” Ramirez says. “Of course, the internet is changing everything.”
Typically made of glass, though sometimes of plastic, these strands carry information in light coursing through their cores. A second layer prevents the light’s escape by bouncing it inward. The glass has to be ultrapure, free from foreign atoms, and so is grown from the bottom up. Gas molecules are deposited onto a rod that is later removed, a strategy figured out in the 1970s.
In a 1975 Science News article, reporter John Douglas hailed this technology that could carry many more messages than existing clogged communications systems. “Where wires once sufficed … a glut of messages has begun to tax the intrinsic limits of present technology.” Other elements are added into the glass that help amplify the signals. The composition is tuned to a tee to provide the necessary optical properties. The light moving through optical fibers often comes from lasers, another materials-driven technology.
And more
The list of materials that helped put oceans of information at our fingertips goes on and on.
The polymer Teflon, invented by accident in 1938 at DuPont and famous for its use in nonstick kitchenware, now also shows up in the minuscule microphones in our phones and computers. A Bell Labs effort discovered that when Teflon is coated with aluminum, it holds enough of a charge to pick up sound without being electrified.
Sometimes, new materials started out as chemical curiosities, with researchers then figuring out how the materials could be useful. The liquid crystal displays in today’s smartphones and televisions, for example, were born from research in the 1960s exploring how electricity jostles molecules in a liquid. That shake-up changes the molecules’ interaction with light, an innovation first put to use in a calculator’s display.

And powering our phones are lithium-ion batteries that became commercially available in 1991, after nearly two decades of iterations that are ongoing still. Researchers sought materials that more readily give up electrons for the batteries’ anodes and materials that better attract them from the cathodes, while also swapping materials to keep the batteries lightweight and reduce the likelihood of explosions.
All of these developments and more led to today’s abundance of electronic devices. But ever more improvements, and our constant urge to upgrade, creates a new problem: “How do we unmake this stuff and recycle those substances safely?” asks Ploszajski.
— Carolyn Wilke
How plastics boomed
In a quest to connect with the omnipresence of plastics, Susan Freinkel, author of Plastic: A Toxic Love Story, pledged to go a day without touching any. Glimpsing her plastic toilet seat, Freinkel gave up the experiment mere moments after it began and instead spent the day cataloging page upon page of plastic stuff she encountered.
Plastics covered her body — in yoga pants, sneakers and eyeglasses. Plastic made up the entire interior of her minivan and parts of kitchen appliances. Plastic packaging protected her food, and after eating, she dumped her trash in a plastic bin. Even the walls around her contained plastics, from the paint to the synthetic insulation.

Today, we’re awash in plastics. Yet at the beginning of the 20th century, only a handful of plastics had made their way into homes.
The story of commercial plastics began in the 1860s, when John Wesley Hyatt, seeking a substitute for the ivory popularly used in billiard balls, landed on a material later called celluloid. It made its way into combs, other novelties and photographic film. But cellulose, a natural substance, was at its heart; it wasn’t fully synthetic.
The first fully synthetic plastic, Bakelite, arrived in 1907, discovered unexpectedly as Belgian-born chemist Leo Baekeland sought an alternative to the natural shellac that insulated electrical cables. Celluloid was a suitable substitute for ivory and tortoiseshell, but sleek, shiny Bakelite gleamed with modernity. It quickly made its way into a host of products, including the casings for radios, jewelry and telephones. A new era of innovating on nature’s materials, rather than merely mimicking them, was born, Freinkel writes.
Yet it wasn’t until the 1920s that researchers started to understand plastics’ chemical nature. Plastics are a type of polymer, large molecules made of repeating units. At the time, what gave natural polymers like cellulose, shellac and rubber their properties remained unknown. Inventors seeking new human-made substitutes for these materials could land on something similar, but it was a trial-and-error endeavor. Credit for changing all that goes to the German organic chemist Hermann Staudinger.

From experiments on natural rubber, Staudinger showed that large, heavy molecules could be formed by the linking of many smaller molecules into chains. As Science News put it in 1953, when Staudinger was awarded the Nobel Prize in chemistry: “The way the molecules regiment themselves determines the differences between springy rubber, hard plastic and tough fiber.” It might sound obvious today, but it was a contentious claim when Staudinger proposed it. Chemists at the time thought that what we now call macromolecules were simply aggregates of smaller molecules.

Staudinger’s ideas gradually gained acceptance and formed a basis for new research on polymers. In the following decade, industrial chemists worked to figure out the chemical reactions needed to create new polymers, plastics among them. One early success story was nylon, invented in 1935 as a substitute for silk. American women were introduced to nylon stockings in 1940 and they proved a hit — in a year, nylons accounted for 30 percent of the hosiery market.
But it was World War II that drastically increased demand for plastics. The military turned to the new industry to make substitutes for strategic materials such as glass, brass or steel, says Freinkel. World War II called for nylon’s diversion to military uses, so women offered up their stockings to be recycled. “Great piles of stockings retired after faithful and intimate service are awaiting resurrection — thousands of pounds of them,” reported a 1943 Science News article.

Though small at the start of the war, the plastics industry got better at making its wares and boosted production. Processes such as injection molding, which spurts melted plastic into a mold “sort of like a Play-Doh Fun Factory,” Freinkel says, made it possible to mass-produce plastic. A technique called blow-molding, invented in the 1930s and based on the same principle as glass blowing, also helped, making a quick way to form plastic bottles.
As wartime demand dried up, the plastics industry began to bring more plastics to the people. “That’s really where you start to get this flood of plastic into everyday life,” Freinkel says.
The promise of plastic was on display in 1946. Held in New York City, the first National Plastics Exhibition featured wares made of the wonder material, and some 87,000 people visited. “Thousands of people lined up to go to this trade show and walk through this conference hall and gawk at stuff that had an almost magical quality,” says Freinkel. Visitors saw durable nylon fishing line and window screens in a riot of colors. Such items seemed transformative — mass-produced plastic offered “a new way to have the good life on the cheap.”

Studies of the properties of polymers and how to make them continued over the following decades. In the early 1950s, researchers at Phillips Petroleum Company figured out how to use chromium oxide catalysts to transform products left over from the refining of petroleum into polypropylene and polyethylene. Heat- and shatter-resistant, the high-density polyethylene produced by this process is used in products such as hula hoops and baby bottles. It’s one of the most commonly used plastics.

We’ve come a long way from the days of celluloid and Bakelite. Tens of thousands of plastic compounds exist today. We now produce an excess of 380 million metric tons of plastic a year — that’s more than a hundred pounds of what’s typically very lightweight stuff for every person on the planet every year.
— Carolyn Wilke
Can we clean up?
By the mid-1960s, researchers started noticing plastic pieces in the ocean, says science writer Susan Freinkel, author of Plastic: A Toxic Love Story. Now plastic pollution is found virtually everywhere, in bits wafting in the winds, on Earth’s tallest mountain and as trash piling up on the seafloor.

Plastics, born from chemistry labs and now ubiquitous, provide a quintessential example of the journey from material marvel to environmental nuisance. But they’re not the only problem.
The organic chemistry advances of the early 1900s made new and exciting materials possible, but also allowed people to make more and more materials that weren’t recyclable, says Thomas Le Roux, a historian at the French National Centre for Scientific Research in Paris and coauthor of the 2020 book The Contamination of the Earth. By the 1970s, new disposable products, from pens to razors to packaging, signaled an ease of life. “It was modern to throw away what we buy,” he says.

The consequences of this easy-come, easy-go relationship with our stuff soon appeared in the environment. Our unabated demand for fossil fuels, used not only as fuel but as raw materials for making plastics, releases emissions that contribute to Earth’s changing climate.
Many of our modern substances, plastics and beyond, were created to solve problems, says Mark Jones, a chemist and member of the National Historic Chemical Landmarks committee of the American Chemical Society. For instance, before the 1930s, air conditioning and refrigeration relied on ammonia, which is flammable and toxic. That changed with the introduction of Freon and other chlorofluorocarbons, or CFCs for short, which were created by chemists in the 1920s. These molecules appeared to have little effect on living things. “They were presumed to be incredibly safe,” says Jones, who recently retired from a role in communications at Dow Chemical.
But Freon and its CFC cousins had unforeseen consequences on the atmosphere when they escaped air conditioning and refrigeration systems. In the 1980s, scientists discovered a hole in Earth’s ozone layer, created when CFCs rise to the stratosphere, break down and react with ozone, destroying it. In solving one problem, humanity found itself with another.

There are parallels with how catalytic cracking played out. Mechanical engineer Eugene Houdry invented the process, which ultimately helped enable abundant automobile travel. But, concerned with the health risks automobile pollution created, he invented a catalytic converter to clean up emissions.
The history of science offers up abundant examples of solutions begetting new problems. Polychlorinated biphenyls, or PCBs, which proved useful as insulators in electronics, can cause serious health problems when they enter the environment, including cancer. The compounds that allow food to slide without sticking out of kitchen pans belong to a family that have earned the title “forever chemicals” for their tendency not to degrade. Lithium mining, which has increased with demand for lithium-ion batteries, guzzles water and can release harmful chemicals that contaminate ecosystems and poison drinking wells. Other battery ingredients, such as cobalt, are mined unethically — sometimes using child labor.
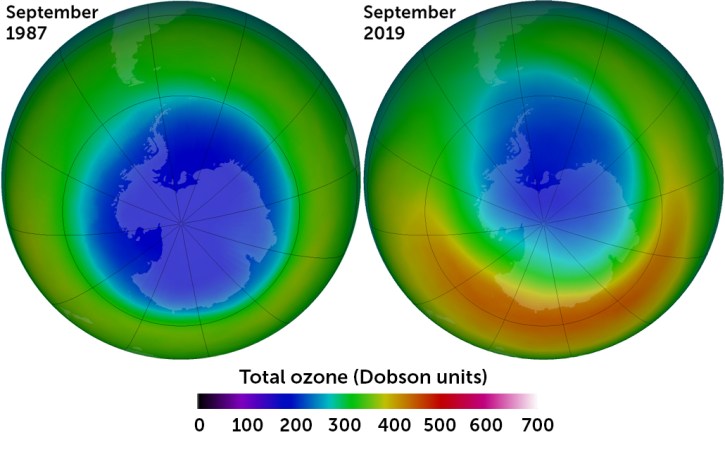
Perhaps the largest unintended consequence that humankind faces today is climate change. Human activities — factories, mining, growing food, traveling, using air conditioning and heating to keep indoor climates comfortable — have released greenhouse gas emissions which have heated the world by around 1.25 degrees Celsius since preindustrial times. The world is already experiencing extreme weather events linked to climate change.
Clearly chemists and materials scientists have contributed to these problems. But they will inevitably be part of finding solutions as well. There’s a cyclical nature to the promise and perils of new molecules and materials. “The entire history of chemistry is, ‘Hey, look what I can do! Darn, I wish I hadn’t done it that way! But I have another way I can do it.’ And that keeps us kind of moving forward,” Jones says.

Chemists are now creating plastics that break down on purpose and can be recycled more easily. Materials scientists are developing better membrane materials to filter pollutants out of water. Engineers are deploying new materials to capture carbon dioxide at smokestacks. New iron-based catalysts could someday convert carbon dioxide into jet fuel, potentially cutting greenhouse gas emissions from air travel. And researchers continue to innovate to turn more and more of the solar spectrum into energy, and so cut our reliance on fossil fuels.
Chemistry and materials innovation can’t solve all our problems. People’s choices will also matter. Weighing the risks and rewards associated with new materials will require recognition of the potential problems, regulations to combat them, willpower, collaboration and collective action. History holds plenty of lessons, but it’s not yet clear whether we’ll learn from them.
— Carolyn Wilke
From the archive
-
Ice Cream from Crude Oil
Edwin E. Slosson, director of Science Service, reports on “some of the achievements and hopes of modern chemistry” from a meeting in Philadelphia. “Ice cream made from crude oil is one of the many marvels….”
-
Fabrics of the Future
Chemists are fashioning fibers and fabrics from cow’s milk, trees, coal, fish and sand – with stranger sources still to come.
-
Laws of Matter Up-to-Date
Chemist Helen Miles Davis outlines 74 facts of matter known in the late 1940s.
-
Shineless Stainless Steel
Reporter Ann E. Ewing describes the many benefits of black rustless steel.
-
Impurity Virtue in Some Materials
A very brief description of the materials and applications that benefit from doping.
-
The Tailored Molecule
The transistor serves as an example of the coming age of “molecular engineering” – “designing a new material to fit a special purpose.”
-
Fibers and More Fibers
A look into the world of new polymer fibers, and the drivers behind their development.
-
Electron’s Eye View
A look at the new views the electron microscope has offered in the decades since its development.
-
Bright Future for a Serendipitous Chemical Discovery
This story about “microcrystalline polymers” offers a window into the promise of polymer chemistry.
-
Catch the Sun
Award-winning writer Janet Raloff describes the current state and future of solar energy.
-
Lessons Learned from a Lady
Statue of Liberty restorations gave scientists an in-the-field look at how materials perform.
-
Making the Right Stuff
Instead of relying on serendipity and trial and error, scientists are learning to assemble atoms and molecules into materials with the mix of properties needed.
-
Invisibility Uncloaked
Metamaterials are at the center of a race to make things disappear.
-
Carbon flatland
“Suddenly graphene came on the scene and it had a completely new physics to it…. That got everyone very excited.”
-
Beginnings of Bionic
Researchers are exploring how to “merge the rigid, brittle materials of conventional electronics with the soft, curving surfaces of human tissues.”
The Latest
-
‘Forever chemicals’ may pose a bigger risk to our health than scientists thought
PFAS are linked to obesity, cancers and more. Growing evidence of the chemicals’ risks has prompted new guidance for safe drinking water and consumer testing.
-
Microplastics are in our bodies. Here’s why we don’t know the health risks
Researchers are racing to try to understand how much humans are exposed and what levels are toxic.
-
Superconductor research surges forward despite controversy over stunning claims
After retractions from Ranga Dias’ group, high-pressure physicists are feeling the squeeze, fearing the controversy will tarnish other research.
-
Rare earth mining may be key to our renewable energy future. But at what cost?
We take you inside Mountain Pass, the only rare earth mine in the United States.