Carbon flatland
Graphene’s two dimensions offer new physics, novel electronics
Some physicists spend their days exploring the three dimensions of space, the four dimensions of spacetime or even the 11 dimensions of something called M-theory. Other researchers are content with just two.

But fewer dimensions doesn’t mean less science. For seven years, researchers have been enjoying a two-dimensional playground of new physics provided by a superflat material called graphene.
This deceptively simple substance — nothing more than a sheet of honeycombed carbon atoms, which you can find within flakes from pencil lead — contains head-slappingly bizarre physics. Unlike almost any other common material, graphene sometimes behaves according to the weird rules of quantum mechanics. And electrons within it assume an otherworldly identity, zipping along as if they have no mass.
“Suddenly graphene came on the scene and it had a completely new physics to it,” says Joseph Stroscio, a physicist at the National Institute of Standards and Technology in Gaithersburg, Md. “That got everyone very excited” — even scientists who ordinarily like lots of dimensions.
Discovered in 2004, graphene was quickly recognized as cool enough to warrant a Nobel Prize in physics, awarded in 2010. Now, researchers are shifting from simply being excited about graphene (SN: 9/27/07, p. 200) to more deeply understanding and even harnessing the physics at play.
For one thing, scientists now understand how stacking one sheet of graphene atop another in just the right way can change the way electrons flow between the layers. Other researchers have found that putting graphene atop a slab of boron nitride lets them manipulate the electron flow far better than before. Some groups are already designing devices for a new graphene age; this spring, IBM researchers reported building the first electronic circuit entirely out of graphene.
Graphene, the two-dimensional wonder material, seems ready to deliver on some of its early promises for the three-dimensional world.
A simple sheet
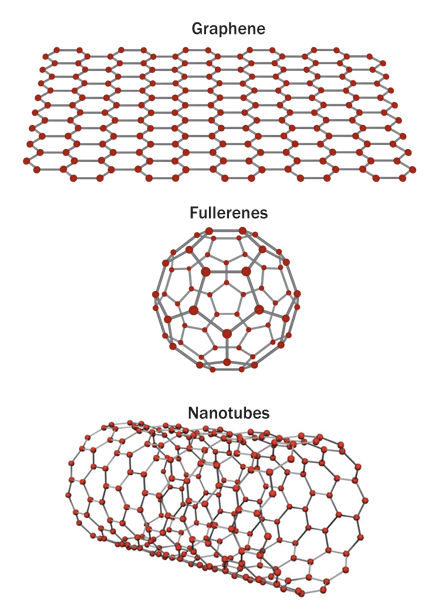
Not all of graphene’s predecessors have lived up to their original hype. In 1985, Texas and British chemists discovered cages of carbon atoms including the famous “buckyballs,” 60-carbon conglomerates that look like miniature soccer balls. These molecules, part of a class called fullerenes, were touted as the next big thing in electronics; yet after a quarter century, nobody has a buckyball running an iPhone. Then, in 1991, a Japanese scientist discovered another carbon curiosity, tiny “nanotubes” made of rolled-up atoms. Although some scientists have developed new electronics based on carbon nanotubes (SN: 12/4/10, p. 20), the tubes turned out to be hard to make and arrange cleanly.
Now graphene is having its try at a technology revolution, and many argue it will fare better than the other carbon protégés. “The thing about graphene is that it’s a truly two-dimensional crystal,” says Antonio Castro Neto, a theoretical physicist at Boston University who is setting up a new graphene research center at the National University of Singapore. “We never had something like that before.”
In graphene, electrons can flow far more freely than they can in either buckyballs or nanotubes, in part because it is the simplest of these forms of carbon. The bonds between the atoms also make graphene superstrong and superflat; in theory, a 1-meter-square hammock of graphene could support the weight of a cat despite being lighter than the cat’s whisker.
Rather than forming as individual sheets, graphene forms as layer after layer within graphite, the stuff of pencil lead. One millimeter of graphite contains roughly 3 million layers of stacked graphene. “If you write very carefully, it’s likely you’ll get a few layers of graphene from your pencil,” says Sankar Das Sarma, a physicist at the University of Maryland in College Park.
In keeping with the office-supplies theme, the scientists who won the Nobel for graphene used Scotch tape to pull apart flakes of graphite. By repeatedly folding and then opening up a piece of tape with graphite stuck on, Andre Geim and Konstantin Novoselov of the University of Manchester in England managed to peel off single graphene layers.
Each single layer, the scientists later found, behaves in extraordinary ways. In most materials, the speed of electrons changes with their energy. In graphene, though, electrons behave as if they have no mass; they move at a constant speed no matter their energy, and they cannot be stopped. Only particles in atom smashers and cosmic rays behave this way, and the math that describes graphene electrons is very much like the math that describes neutrinos, those elusive, nearly massless particles that zip through space.
“Boom — all of a sudden we have a system of quasi-neutrinos,” says Eva Andrei, a physicist at Rutgers University’s campus in Piscataway, N.J. The only difference is that graphene electrons travel at roughly a million meters per second; neutrinos (and light) travel 300 times that fast.
Discovery after discovery has revealed the bizarre things these graphene electrons can do. In April in Science, Geim and his colleagues reported that under certain conditions, electrons in graphene can adopt a split personality in which one of their properties (electric charge) behaves according to the rules of the everyday world but another property (spin) behaves according to the otherworld of quantum mechanics. “We are not used to quantum mechanical effects happening in our normal life,” says Castro Neto. “When you find a material like that, it’s really a treasure.”
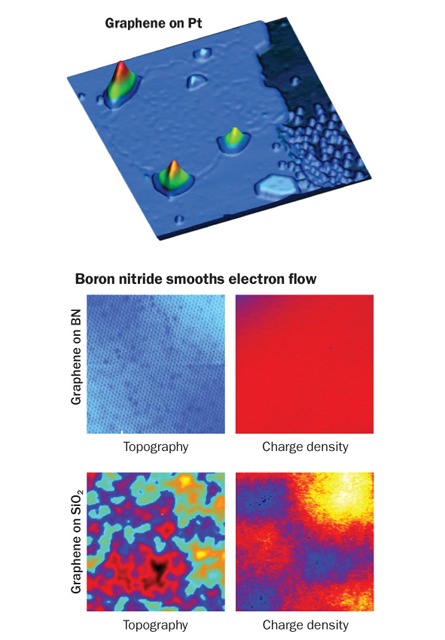
Stretching graphene also makes its electrons do funny things. At the University of California, Berkeley and the Lawrence Berkeley National Laboratory, scientists accidentally found that if they grew graphene atop platinum, the graphene sheet could sprout tiny bubbles on its surface. Within those bubbles, electrons act as if they are under the influence of a strong magnetic field. Nobody is really sure what this means, says team member Castro Neto, but researchers in Singapore have managed to create similar bubbles at will. “Now we can control at the nanoscale the nature of the electronic states,” Castro Neto says. “I think this is going to really generate a revolution in the way in which we deal with graphene.”
And all that in just a single layer of graphene. For even more new tricks, scientists are turning to two-layer, or bilayer, graphene.
Doubled up
When it comes to building new electronic devices, single-layer graphene suffers from one huge drawback: It doesn’t have a “band gap,” or break in the energy levels that its electrons can occupy. Without a band gap, scientists can’t turn the flow of electrons on and off — a crucial part of any electronic gadget. But adding a second layer of graphene creates such a band gap, making the bilayer structure more like a semiconductor in which the flow of electrons can be controlled instead of zooming along willy-nilly.
“Unlike single-layer graphene, bilayer has the possibility of shifting charge from one layer to another,” says Amir Yacoby, a physicist at Harvard University. And interactions among the electrons cause other weird and wonderful physics, Yacoby says, such as the breaking of fundamental symmetries in how the electrons spin and move. “Several experiments indicate that interesting things are happening, but there is really no good understanding of what is going on there as of yet,” he says.
As intriguing as bilayer graphene is, making it isn’t as simple as slapping one graphene layer atop another. How the two layers are stacked relative to one another is crucial for electronic applications, Andrei and her colleagues reported in March in Physical Review Letters. If the carbon honeycombs of each layer are rotated less than 5 degrees relative to each other, Andrei’s team found, then they behave as a true bilayer, and can create the electronic band gap. But if the honeycombs are offset by about 20 degrees or more, then the graphene layers continue to behave electronically as two separate layers.
Such research shows how graphene electrons can be coaxed into acting however scientists want them to, Andrei says. “Here we have an external knob to control the electronic properties,” she says. “That’s quite exciting.”
Where two layers are good, three might be even better, and so some researchers are pushing to make trilayer graphene. But as with the bilayer, researchers can’t just throw three graphene sheets in a pile; the carbon honeycombs have to line up just so. Usually trilayer graphene comes in what’s called the ABA form, in which the honeycombs of the top and bottom layer mirror each other. The ABC form, in contrast, slides that topmost layer over to one side so that the honeycombs climb like stairsteps.
Nobody has ever gotten the ABA version of graphene to do anything very exciting, but a research team led by Tony Heinz of Columbia University has been playing around with the ABCs. In a paper appearing online in May at arXiv.org, Heinz and his colleagues report making an electronic band gap appear in ABC graphene. It’s the first time anyone has been able to do this, and opens a new class of materials that scientists can work with.
Graphene building
Just as in the construction industry, foundation is everything when it comes to building with graphene. Left to its own devices, a graphene sheet will wrinkle like poorly torn plastic wrap. Some researchers try to get around this by suspending graphene on clips in air, like a piece of laundry hanging out to dry. Even then, though, the edges will ripple or roll themselves up, or the sheet itself will tear. So scientists are investigating new ways to lay graphene flat and keep it that way.
Early on, most research teams plopped some graphene on a slab of silicon dioxide, the stuff of everyday computer chips. But the silicon and oxygen atoms interfered with the way the graphene’s electrons zipped along. Instead, scientists have now switched their foundations to boron nitride, the stuff used to add glitter to cosmetics. Like graphene, boron nitride also has its atoms arranged in a single hexagonal sheet, with boron and nitrogen alternating in the spaces where carbon atoms sit in graphene. It’s an almost perfect match.
“Once you use a boron nitride substrate and stack it with graphene, a number of things change,” says Philip Kim, a physicist at Columbia. Compared with silicon dioxide, boron nitride lets the electrons zip along without interference, Kim and his colleagues showed last year.
To see why boron nitride works so well, scientists at UC Berkeley and the Lawrence Berkeley lab recently took a closer look at what happens when graphene and boron nitride meet. Using a scanning tunneling microscope, which can see at the level of individual atoms, the team compared graphene mounted on silicon dioxide with graphene mounted on boron nitride. The silicon dioxide version turned out to be strewn with “charge puddles,” or spots where the electron flow got hung up. In contrast, the boron nitride samples were practically puddle-free. Michael Crommie, Alex Zettl and colleagues reported the findings this year in Nano Letters.
With this solid foundation and new ways of stacking, researchers can now act as architects, designing devices that take advantage of graphene’s protean qualities. Though it probably won’t ever replace the industry standard of silicon, graphene could lead to new kinds of gadgets. “It’s really about what we can gain by using graphene in different applications,” Castro Neto says.
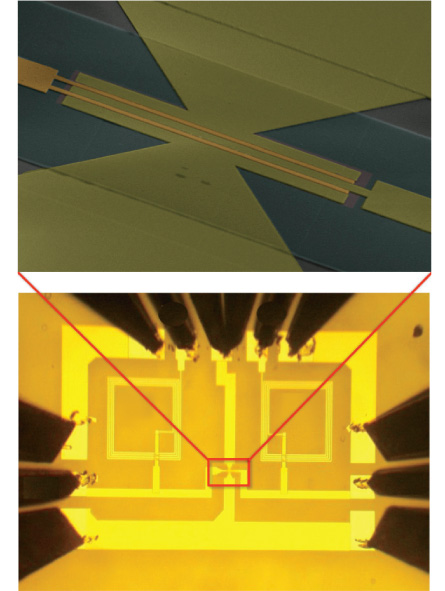
Already, the wonder sheets are making inroads into silicon’s traditional territory. In June in Science, IBM scientists reported making an integrated circuit whose components, including a transistor, are made completely out of graphene. Other teams have made individual graphene components before, and even linked hundreds of transistors together on a single chip. But the IBM group, led by Phaedon Avouris at IBM’s Watson Research Center in Yorktown Heights, N.Y., is the first to make a complete circuit entirely out of graphene. Because graphene is so cheap, these kinds of circuits could prove popular for use in smartphones and other portable devices.
Unlike today’s rigid computers, graphene can also be molded over surfaces, like a dust sheet protecting furniture from debris. Last year, for instance, researchers at Samsung in South Korea showed off the world’s first graphene touch screen, a flexible sheet that uses the carbon atoms as see-through electrodes. Other industry scientists are developing graphene-based biological sensors and solar cells as cheap, bendable alternatives to ones already on the market. And in a paper published in June in Physical Review B, the Berkeley collaborators describe how they gathered up graphene like a bedsheet into folds, ruffles and pleats, creating a never-before-seen material they dub “grafold.”
At another research frontier, scientists are taking advantage of graphene’s weird electronic properties by interleaving stacks of it with different materials, such as insulators. “It’s like making a big sandwich with tomato, lettuce, meat, bread and so on,” says Castro Neto. “Each material gives a different taste, and at the end of the day you can have a very juicy sandwich that doesn’t look at all like the piece of bread you started with.”
Scientists are just starting to test the various combinations available, and it’s clear the graphene delicatessen won’t run out of possibilities anytime soon. In early 2005, at the first major American Physical Society meeting after Geim and Novoselov’s Nobel-winning discovery, attendees at the only graphene session didn’t even fill a small conference room. In spring 2011, Novoselov headlined the same physical society meeting while dozens of breakout sessions delved into the new possibilities offered by graphene.
With this amount of intellectual firepower, the discovery that started with some Scotch tape and pencil lead is sure to meet a much higher-tech end.







