Beneath that blazing facade
Researchers revamp ideas about what’s in the sun
In the pantheon of cosmic celebrities, the sun is one true superstar. It’s not only the Earth’s prime source of light and heat — it also fuels the greenery that makes breathing possible, keeps time by setting the body’s daily rhythms and spits out charged particles that create the beauty of the aurora borealis.



But for all its roles on life’s stage, the sun remains something of an inscrutable star. You might say it’s the Tiger Woods of the cosmos.
Behind its blazing facade, the sun turns out to be reluctant to give up its secrets. Most frustratingly, astronomers haven’t figured out one of the most basic facts about Earth’s nearest star: exactly what it’s made of.
“We really don’t know what the sun’s composition is,” says Carlos Allende Prieto, an astronomer at the Instituto de Astrofisica de Canarias in the Canary Islands. “It’s a big problem.”
Physicists do know a lot about the sun and how it works: Hydrogen atoms fuse in its core, forging helium and heavier elements and spewing out energy in the process. But over the past several years, scientists have dramatically overhauled estimates of the sun’s chemical makeup. In particular, they say there may be far less of key elements such as oxygen, carbon and nitrogen than previously thought. These changes are major enough to throw into question other basic assumptions about the sun, such as ideas about how sound waves travel through its interior, ringing it like a gong.
And because the sun is the yardstick by which many other astronomical phenomena are measured, if scientists change their ideas about solar chemistry, they must also modify their thoughts about the chemical composition of sunlike stars. Those changes, in turn, affect ideas about how galaxies evolve, such as the rate at which stars form over time, synthesizing and ejecting heavier elements out into the universe.
“People always compare stars of the same type to the sun, and now the sun has changed,” says astronomer Nicolas Grevesse of the University of Liège in Belgium. “Now we’re rechecking everything, restarting all the analyses from A to Z.”
Slowly, however, researchers are edging toward an answer. New, more sophisticated computer models have improved understanding of the sun’s atmosphere, permitting better estimates of chemical abundances. Deeper discussions of which data to include, and which to leave out, are helping smooth battle lines between research teams arguing over what the final numbers should be. Soon, stories about what Earth’s superstar is made of could read more like trusted newspaper copy than celebrity gossip.
Reading between the lines
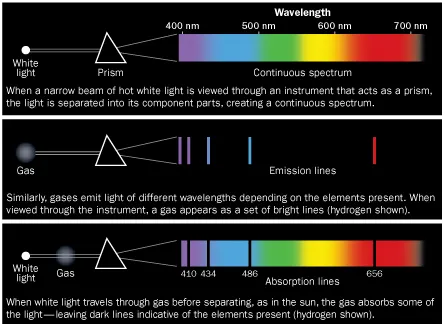
Researchers’ preferred way to study the sun’s chemical makeup, from nearly 150 million kilometers away, is to analyze the light flowing from it. Spread sunlight out into its spectrum of wavelengths, as a prism creates a rainbow, and the light appears riddled with dark lines. Since the late 1850s scientists have known that these dark lines correspond to chemical elements in the sun’s outer layers; the elements absorb radiation coursing outward from the core and blot out the light. The more light is absorbed, the more of that element is assumed to be present.
In 1929, astronomer Henry Norris Russell used this spectroscopic technique to publish solar abundances for 56 elements. Since then, astronomers have refined the numbers further, and generally believe that hydrogen, the lightest element, makes up around 70 percent of the sun by mass. Over millions of years, nuclear fusion in the solar core slowly converts the hydrogen to helium and subsequently to heavier elements, collectively known to astronomers as “metals” (though they include gases such as oxygen). The question has been exactly how much of the sun was made of metals.
An influential 1989 review paper, coauthored by Grevesse, reported that metals make up 2 percent of the solar photosphere, the lower level of the sun’s atmosphere where spectral lines are formed. But during the 1990s, new and more sophisticated analyses began to throw that estimate into doubt.
Though spectral lines alone offer basic clues to the sun’s innards, researchers need models to interpret those lines. Such models try to reproduce the churning and flowing of gases in the sun’s photosphere. Previous modeling had used one-dimensional computer simulations that divided the solar atmosphere into simple stratified layers. In contrast, the new models look at a small chunk of the atmosphere but simulate it in three dimensions and in much more detail — including, for instance, how mass and energy churn out from the convection zone, the outermost layer of the sun’s interior, into lower layers of the atmosphere.
The 3-D simulations had a profound implication: Astronomers’ earlier interpretations of spectral lines had to be changed. In some cases, the strength of an absorption line could signal an abundance that was different from what scientists had thought.
These recalculations seemed to improve things, by tightening estimates for the abundances of elements like iron and silicon. “It was only when we started applying it to more important elements like oxygen and carbon, the most abundant metals in the sun, that we quite quickly realized our results were going to be very different,” says Martin Asplund, an astronomer at the Max Planck Institute for Astrophysics in Garching, Germany.
Asplund’s team published estimates of solar photospheric oxygen abundance that were 30 to 40 percent lower than commonly accepted values, with similar changes for carbon, nitrogen and neon. The researchers also analyzed the other elements in the sun, most of which required only minor revisions. In all, the team found a solar metal content not of 2 percent, but of 1.4 percent. The researchers summarized their latest work last year in the Annual Review of Astronomy and Astrophysics.
Radiating puzzles
Lower metallicity doesn’t bode well for other aspects of solar study — particularly models of the solar interior based on helioseismology, the study of how waves, such as sound waves, travel through the sun.
Turbulence within the sun generates sound waves that oscillate back and forth within the star. Studies of this reverberation have yielded important insights into the solar interior, such as how its layers rotate at different speeds. The technique can even be used to study sunspots on the far side of the sun, since sunspots absorb the sound waves and dampen the reverberation.
Helioseismology studies have built up a certain picture of the solar interior, including such details as the depth of the convection zone. These models of the solar interior were reasonably successful, and physicists thought they understood the sun’s insides.
The work of Asplund’s team throws a wrench in that picture. If models of the solar interior are adjusted to fit the 30 to 40 percent lower oxygen abundance in the photosphere, then they no longer match up with helioseismologists’ observations. For instance, the models now calculate incorrect values for the speed of sound and the density within the sun, as compared with those actually measured.
Assuming Asplund’s abundance measurements are correct, “what this tells us is our models are not good enough,” says Sarbani Basu, a helioseismologist at Yale University.
In other respects, though, the lower abundances solve a different astronomical dilemma. “It was a problem for a long time that the sun appeared to be unusually abundant in heavy elements compared to the solar neighborhood,” Asplund says. Now, the new work “makes the sun completely normal” relative to nearby stars, he says.
Some astronomers are already incorporating the new, lower metal abundances into studies of the evolution of stars in galaxies other than the Milky Way. Because stars become more metal-rich over time, measuring the metallicity of galaxies provides a look at how far those stars have evolved and a clue to when they formed.
But Lisa Kewley, an astronomer at the University of Hawaii at Manoa, says that some researchers — herself included — haven’t started using the new abundances yet, because it is not clear whether Asplund’s numbers or others offered up by a competing team are correct. It’s possible to study metal content in other galaxies without having a solar reference to pin it to, she notes. For instance, astronomers can compare the abundance of oxygen with the abundance of hydrogen in a particular galaxy. Using this same measure over many galaxies allows researchers to build a comparative picture of galaxy metallicity without ever having to worry about what the sun’s true oxygen abundance is.
Still, arguments over the true solar numbers are making a lot of researchers uncomfortable. Several years ago at the Paris Observatory in France, scientists led by Hans-Günter Ludwig decided to use their own 3-D simulations of the solar atmosphere to see if they could confirm Asplund’s work. And they found that they couldn’t, at least not exactly. Ludwig’s team, including then graduate student Elisabetta Caffau, has reported its own abundances of certain elements — most of which were higher than Asplund’s but not as high as the earlier, 1980s-era estimates.
This spring, the two teams converged in Garching for a weeklong discussion to try to hammer out their differences. Both groups agree that their 3-D models confirm lower metallicity. That finding is also broadly supported by another line of evidence: primordial meteorites. The space rocks known as “CI carbonaceous chondrites” are thought to be the most chemically primitive rocks known, providing clues about what the chemistry of the early solar system, including the newborn sun, was like. Analyses of such meteorites suggest that oxygen and other elements may be less common than the 1980s solar estimates reported.
The problem, though, comes in a number of smaller choices made by each team in determining the abundance estimates. For instance, Ludwig, now at the University of Heidelberg in Germany, and his colleagues use a larger number of spectral lines to derive abundances, but Asplund’s team rejects a number of those lines.
Grevesse, who works with Asplund, says that Ludwig’s group is using spectral lines that are blended, or contaminated, by lines from other elements in the solar atmosphere. “We discarded a lot of lines that had been used in the past,” Grevesse says. Ludwig counters that educated guesses have to be made: “One starts to have to make subjective decisions, like which kind of lines one should include.”
Other subtle effects also contribute to differences between the two groups, such as whether to include only signatures from atoms — such as oxygen atoms — or also lines from molecules — such as carbon and oxygen bound together as CO. (Asplund includes molecular data, whereas Ludwig does not.) The two teams also use different assumptions to calculate details of how the spectral lines form in the first place. “It’s a fairly tricky business,” Asplund says.
Over the past few months, the two teams have edged closer together on the abundances for some elements. Oxygen, however, remains the major battleground. Ludwig’s team comes in with an abundance some 15 percent higher than Asplund’s.
Reconciling the two groups will either require one to accept the other’s line-selection and calibration calculations or will require entirely new types of relevant data. Grevesse says he’d like to see better data from atomic spectroscopists (scientists who study the spectral lines created by different elements) in order to better calibrate observations of the sun. But such studies are expensive and time-consuming.
Ludwig, too, would like better observations — a better catalog of spectral lines as they appear coming from across the entire face of the sun. And Asplund would prefer new and more sophisticated calculations of solar opacity, to see if that might resolve the discrepancies with helioseismology.
None of these approaches are being actively pursued, so a resolution is not likely any time soon. “I think it’s going to take a while to get a definitive answer,” says helioseismologist Basu. The sun may hold onto its secrets as long as it can.
Hunting for exoplanets
Some astronomers are hoping that pinning down the sun’s chemical composition will tell them not only about Earth’s parent star, but also about other planetary systems.
By looking at the chemical makeup of nearby stars, some researchers say, they may one day pinpoint which neighbors are likely to have Earthlike planets. If so, scientists would be able to identify the best candidate systems for hosting extraterrestrial life — without the time and expense of building satellites to hunt for such planets.
Stars that resemble the sun in temperature, gravity and metal content are known as “solar twins.” The key in planet-hunting would be to look for solar twins that have reduced levels of elements such as iron and aluminum that easily condense into dust particles, says astronomer Martin Asplund of the Max Planck Institute for Astrophysics in Garching, Germany. In theory, those elements may have been caught up and incorporated into Earthlike planets that coalesced from the swirling disk of gas and dust around the newborn star. The stuff that ends up in the planets doesn’t end up in the star.
Astronomers have discovered more than 460 planets around stars other than the sun, but most of those planets are far more like the gas giants Jupiter and Saturn than like small, rocky Earth. Scientists look for planets through either a slow method (laboriously watching the movement of a star over many years for the tiny gravitational perturbation caused by a nearby small planet ) or an expensive method (sending spacecraft into orbit to look for the dimming of light caused by a planet passing in front of its star). The French-led mission COROT and the NASA mission Kepler are currently looking for terrestrial planets in this way. None have yet been confirmed.
The chemical analysis method could streamline such hunts, Asplund argues. “We propose that you can identify stars likely to have terrestrial planets just by looking at their chemical composition,” he says.
Limited surveys suggest that about 10 to 20 percent of solar twins seem to be depleted in elements like iron and thus, like the sun, may also host Earthlike planets, Asplund says.