Pacemaker treats sleep apnea
Experimental device works for many patients who can’t use breathing machines
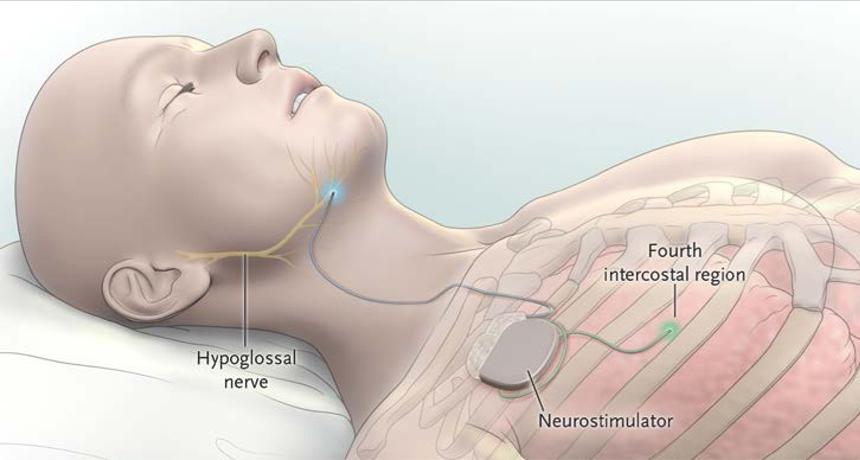
BREATHE IN, BREATHE OUT An electronic pacemaker (neurostimulator) implanted just beneath the skin of the chest gets signals from a sensor between ribs (fourth intercostal region) whenever the chest expands. The pacemaker shoots an impulse to a lead attached to the hypoglossal nerve, which controls the muscle at the base of the tongue. This causes the tongue to protrude, opening the throat just in time for the person to inhale — all while asleep.
P.J. Strollo et al/NEJM 2014







