Better Than Pap: Virus test detects cervical cancer
For more than 50 years, doctors have used Papanicolaou tests—better known as Pap smears—to screen women for cervical cancer. But researchers now report that a newer test gives a more accurate diagnosis.
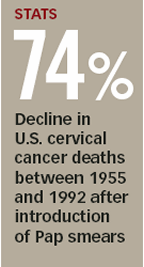
In a Pap smear, cells scraped from the cervix are analyzed under a microscope for physical abnormalities. Tests developed in the past decade, however, can detect the presence of human papillomavirus (HPV) in cervical cells. This sexually transmitted disease is the primary cause of cervical cancer.
In two studies in the Oct. 18 New England Journal of Medicine, researchers report that HPV tests caught many more cases of cervical cancer than Pap smears did.
“When something is visible in the microscope, that’s when the Pap test is positive. But the HPV test can go far before that and give us a window of safety,” explains Eduardo Franco of McGill University in Montreal, who led a study of more than 10,000 Canadian women.
Participants in the study got both a Pap smear and an HPV test. Women with abnormal results in either test, along with a random sampling of those with normal results, had cervical biopsies, a surefire way of diagnosing cancer. Franco’s team found that the HPV tests detected almost 95 percent of the cancers, whereas the Pap smear caught only 55 percent.
Franco adds, though, that the Pap test still serves a purpose. It’s far less likely to give a false negative, he says: If you can see something wrong on the Pap smear, it’s almost as good as a diagnosis by biopsy.
“So it makes more sense to do the Pap test when the HPV one [has shown up] positive,” says Franco.
The second study, led by Joakim Dillner of Lund University in Malmö, Sweden, included more than 12,000 Swedish women. It showed that giving a woman both tests rather than a Pap smear alone increased the chance of early cancer detection.
Gynecologic oncologist Carolyn Runowicz of the University of Connecticut Health Center in Farmington says that more research is needed, however, before any policy change. The two studies, she points out, compare HPV tests with a kind of Pap smear that isn’t widely used in the United States. In conventional Pap smears, cell samples are held between glass microscope slides. The cells tend to clump, making them hard to examine under a microscope. In the United States, 85 percent of smears are now done using a liquid that spreads the cells into a single layer.
“It’s exciting to think about molecular screening,” says Runowicz, “but we’re just not there yet.”







