Blind Bet
Despite uncertain odds, many horse owners gamble on stem cell therapy
No animal has shaped the course of civilization more than the horse. Horses have pulled plows, herded cattle, and brought riders into battlefields and to the edges of continents. Today, horses are carrying their human companions to another frontier—the uncharted territory of stem cell medicine.


Throughout the rich history of horses, their legs have been the source of both strength and weakness. For all their size and brawn, horses move on limbs that are relatively long and slender. That arrangement provides agility and speed to 1,000 pounds of body weight, but leaves the animal at risk of injury. At a gallop, a horse places only one hoof on the ground at a time. That means at any given moment, the animal’s great weight depends on a joint about the size of a baseball. No wonder then that roughly half of all performance horses end up retiring because their legs finally fail them.
So far, no remedy for injuries or arthritis can claim stellar results. Newer treatments exist, but rest and controlled exercise remain mainstays. But now, many equine experts believe that stem cells could supply what medicine so far has not: replacement parts.
“It’s very exciting and incredibly promising,” says researcher Lisa Fortier of Cornell University’s College of Veterinary Medicine.
Not only for horses. Researchers are exploring the potential of stem cells in human orthopedics also, and if the approach works in the equine world, it will probably work in the human one. Two-legged patients have only a fraction of the joint stress. “A horse is essentially walking around on your middle finger,” Fortier says.
Recent studies of stem cell therapies have produced encouraging results, but Fortier also points out that “there is zero peer-reviewed published evidence that this actually works.”
Investigations of stem cells for veterinary medicine began in earnest just this decade, as scientists throughout the world began to understand stem cells. Stem cells have the potential to become many different types of cells. Cells that have already committed to becoming muscle, bone, or any other tissue are unchangeable. Stem cells, by contrast, retain possibility.
Much controversy has embroiled research into stem cells derived from human embryos. But stem cells of a different variety—called adult-derived stem cells—reside in fat, bone marrow, and other tissues. Scientists are still sorting out the potential of these two types of stem cells, and a recent study even announced the ability to transform adult skin cells into an embryoniclike state.
Almost all veterinary work has focused on adult-derived cells, using a technique that involves removing a stem cell from fat or bone marrow and turning it into a new tendon or another component of a joint in an injured horse. Currently, injured tendons become interwoven with scar tissue after healing. Scar tissue is strong but lacks the spring of the original tendon. Elasticity is a key to withstanding force: A palm tree can weather hurricane-strength winds largely because of its ability to bend.
In “most tendon injuries, when they happen, the tendon will never go back to its original mechanical flexibility,” says Allison Stewart of the University of Illinois at Urbana-Champaign’s College of Veterinary Medicine.
Stem cells are appealing because they could potentially grow into something that looks less like scar tissue and more like tendon—without surgery or drugs. And the price, usually only a few thousand dollars, is a small gamble for owners with high economic and emotional commitment to their horses.
Roger Smith, of the Royal Veterinary College in England, helped pioneer the movement by developing a technique in 2002 for extracting stem cells from the horse’s own bone marrow, growing them in a laboratory for about 3 weeks, and then injecting them into the injured limb. Once the stem cells were surrounded by mature tendon tissue, researchers hoped that local chemical cues would induce the bone marrow stem cells to develop into tendon cells. Smith partnered with an entrepreneur to form a company, VetCell Bioscience, and began offering the treatment to horse owners.
Meanwhile, in the United States, veterinarian Robert Harman was also developing a therapy for equine orthopedics, in this case using stem cells extracted from fat tissue on a horse’s rump. His Poway, Calif.–based company, Vet-Stem, has been offering the treatment since 2003.
Most researchers, company founders among them, acknowledge that the instant commercial availability of the treatment has been a major barrier to clinical research. To gauge the effectiveness of any treatment—in people or animals—scientists commonly compare a group that receives treatment with a group that gets nothing or only an elaborately disguised sham. Neither patient nor clinician knows who gets what. But horse owners, in a virtual stampede to stem cell treatment, are loath to consent to a study where their horses might get a placebo of injected saline solution.
“We made the mistake of actually getting this out commercially, so nobody will participate in a study where they might get saline,” says Harman, who is chief executive officer of Vet-Stem, which has treated around 3,000 horses.
So far, the only clinical data are the company’s follow-ups of treated animals. In unpublished data, Harman and his colleagues have compiled a retrospective look at 66 horses treated for tendon injuries in 2004 and 2005. More than 90 percent of the horses were injury-free a year later, and 77 percent had returned to their previous level of activity. Historically, studies suggest that only about half of horses treated for tendon injuries return to their full level of activity. Of these, around 40 to 50 percent injure themselves again.
Smith of VetCell Bioscience, which has also boasted a booming business, discussed data for his horses in December at the annual meeting of the American Association of Equine Practitioners. In a follow-up 3 years after treatment, he reported, horses treated with the VetCell product experienced about half as many reinjuries as did horses treated by conventional methods. Among 82 horses followed for more than a year, 78 percent had returned to full training.
While intriguing, these types of studies are fraught with uncertainties, says Stewart. How severe were the initial problems in these horses? How strenuous was their activity before the injury? What was their level of performance when they returned to competition?
“There’s a lot of weakly supportive evidence,” she says. “You really need a controlled study where you have horses with similar injuries and they get equal amounts of time off.”
She would like to conduct a more rigorously designed study, but is unsure whether she can obtain the funding or the participation from owners. “There’s a huge need to just start simply and sort things out,” she says. Vet-Stem officials report they are conducting a randomized study of 30 horses, half of which will get only saline. They expect results to be available later this year.
Given the practical hurdles to conducting controlled clinical trials, veterinary researchers are also relying on laboratory studies to help the science behind stem cell therapy catch up with its popularity, says Wayne McIlwraith, director of the Orthopedic Research Center at Colorado State University’s College of Veterinary Medicine and Biomedical Sciences in Fort Collins.
Even the basics are still under investigation. “There’s still argument as to what constitutes a true stem cell,” McIlwraith says. In addition, researchers currently have a difficult time examining the tissue after treatment, tracing the origin of the growth, and comparing the new to the original.
In one new study, to be published in the American Journal of Veterinary Research, Alan Nixon of Cornell’s College of Veterinary Medicine and Linda Dahlgren will report the results of an investigation comparing eight horses with induced tendon injuries. Four of the horses received treatment with fat-derived stem cells, and four received saline. The animals were allowed to heal for 6 weeks, and the tendon tissue was then examined at autopsy.
In their article, the scientists will report a significant improvement in the internal architecture and organization of the tissue in the horses that received injected stem cells. The mechanism behind the tendon regeneration, however, remains unclear.
“One of the big questions is whether these cells we inject are actually surviving,” says Dahlgren, formerly of Cornell and now with the Virginia-Maryland Regional College of Veterinary Medicine in Blacksburg, Va. The cells could be releasing proteins that recruit other cells in the tendon repair. And a few studies have characterized the makeup of injected cells—they could be a mishmash of stem cells and other cells influencing growth. Researchers are likewise uncertain about the best way to isolate and concentrate stem cells.
Also controversial is whether the two main reservoirs of horse stem cells—fat and bone—differ in their ability to repair damage. In one of the few direct comparisons, which involved animals with early osteoarthritis, researchers from Colorado State found that the bone marrow stem cells appeared to possess anti-inflammatory properties not seen in the fat stem cells. Neither type of cells, the researchers reported at a recent meeting of the American College of Veterinary Surgeons, “demonstrated overwhelming effectiveness” for the disease.
During that same meeting, the Colorado scientists also described 15 horses with injured joints that had exhausted all other treatment options—their lameness appeared irreparable—before receiving treatment with bone marrow–derived stem cells. With follow-up times ranging from 6 months to 18 months, 10 of the animals were able to return to their previous level of work. The researchers are now following a much larger group of treated horses.
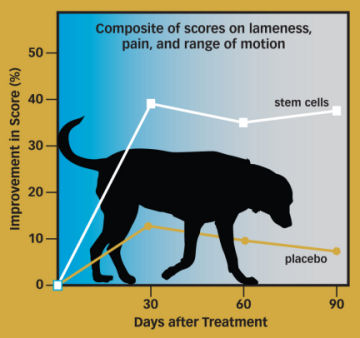
Even as investigations continue, the business side is expanding into new markets. VetCell began offering its bone marrow product in the United States a year ago. And Vet-Stem announced in August that it will start making the treatment available to dogs. Unlike their experience with horses, the company conducted a trial before making the canine treatment available. The first randomized trial of stem cell therapy in dogs appears in the winter 2007 issue of Veterinary Therapeutics.
A team led by Vet-Stem researchers evaluated dogs using a tool for rating the degree of movement, lameness, and pain. Nine dogs receiving a placebo for osteoarthritis showed about a 10 percent improvement after 3 months. However, nine stem cell–treated dogs experienced about a 35 percent improvement.
“Three of these dogs had owners who were considering euthanasia because of their animals’ pain and functional disability,” say the authors, who include veterinarians from five U.S. clinics. “These dogs are now living relatively pain free.”
Smith predicts that the large, randomized trials that could answer key questions probably won’t be conducted until scientists have a similar product to test in humans. In human medicine, government regulators require studies before approval. In 2007, VetCell Bioscience Ltd. changed its name to MedCell Bioscience Ltd. to reflect its aim to develop the approach for people. Such products are already under development by several other U.S. companies.
Time may be the biggest test of whether stem cells live up to their expectations in equine orthopedics, says Stewart. The same horse owners that have driven the excitement for stem cells will spread the word if the track record starts to look disappointing. “Whether it stays,” she says, “will in part be determined by how effective it is.”
Laura Beil is a freelance writer in Texas.