Gastric bypass surgery causes sugar-burning gut growth in rats
Intestinal changes could explain rapid improvements in diabetes
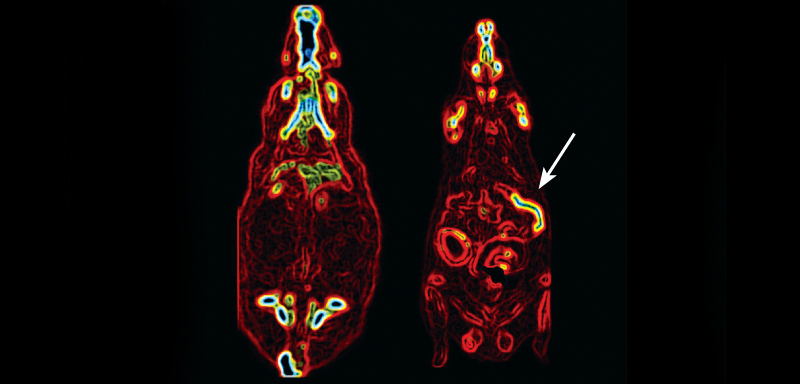
SUGAR HIGH After gastric bypass surgery, the intestines of an obese rat (Roux limb indicated by arrow, right) burn more sugar (bright red and yellow) than before the surgery (left). This boost in sugar use could explain why surgery can rapidly improve diabetes.
Courtesy of Stylopoulos Laboratory
A beefed-up chunk of intestines might do the heavy lifting of gastric bypass surgery.
The surgery’s rapid diabetes-improving effects appear to stem from growth of new intestinal tissue. After having an operation to remodel the gut, obese rats build the new tissue by drawing sugar from the blood, researchers report in the July 26 Science. This energy drain could explain how the most popular type of gastric bypass lowers diabetic patients’ sugar levels surprisingly swiftly, says coauthor Nicholas Stylopoulos, an obesity researcher at Harvard Medical School.
It’s the first time researchers have linked the effects of gastric bypass surgery to the gut’s sugar use, says Blandine Laferrère, a clinical diabetes researcher at Columbia University. “It’s a fascinating new piece of the puzzle,” she says.
Currently, gastric bypass surgery is one of the best treatment options for morbid obesity and obesity-related diabetes, says Randy Seeley, an obesity and diabetes researcher at the University of Cincinnati. Patients typically lose weight and no longer need insulin shots to lower blood sugar. But it isn’t practical to use the procedure on millions of patients. Beyond the risks and complications of major surgery, Seeley says, “There aren’t enough surgeons or surgery tables to treat everyone.”
So researchers would like to engineer drugs or other scalpel-free treatments that mimic the effects of gastric bypass. But first, scientists have to figure out how the surgery works.
In the most popular type of bypass surgery, called Roux-en-Y, surgeons rearrange the gut’s plumbing by hooking the middle part of the small intestine to a golf ball–sized pouch that they cut away from the patient’s stomach. (Normal stomachs are roughly the size of a football, Stylopoulos says.) Food travels through the small pouch to the intestine section, which is nicknamed the Roux limb, bypassing most of the stomach and upper intestine.
For years, researchers assumed patients lost weight and reduced blood sugar after the surgery because they had to restrict food intake due to the smaller stomach pouch. Or perhaps the surgery reduced calorie absorption because of the intestinal bypass. “It seems so obvious,” says gastroenterologist Lee Kaplan, of Massachusetts General Hospital’s Weight Center. “But it’s not true.”
Several puzzling observations cued scientists to look for explanations. After surgery, patients’ blood sugar levels tend to return to normal in just a few days — before they’ve lost any weight, Stylopoulos says.
And after scientists perform the surgery in rats, the animals’ food preferences seem to change. Instead of eating fatty meals, the rats switch to foods with fewer calories, Seeley says. “That’s exactly the opposite of what you would expect.” If the drastically smaller stomach were the main driver of the weight loss, he says, animals should chow down on fatty foods to pack in calories before their pouches get too full.
Researchers looking for explanations for the surgery’s effects have picked out key differences between the guts of gastric bypass patients and people who didn’t get the surgery. Gastric bypass flips several hormonal switches that within weeks seem to alter patients’ appetites and insulin levels (SN: 9/9/11, p. 26). Surgery might also nurture growth of gut microbes that help burn fat (SN: 5/4/13, p.10).
But scientists still can’t explain why blood sugar levels improve so quickly after surgery — often before patients have left the hospital.
To find out which organ was using up the sugar, Stylopoulos and team injected a radioactive form of glucose into the bloodstream of rats that had undergone bypass surgery. Compared with rats that underwent a sham surgery, rats with gastric bypasses shuttled nearly twice as much sugar to the intestine, especially to the Roux limb.
When the researchers dissected the animals, they noticed that the Roux limb looked big.
“You can easily see which section is the Roux limb,” says coauthor Nima Saeidi, a biomedical engineer at Harvard Medical School. “It’s very thick.” The Roux limb grows to a thickness at least 40 percent bigger than the intestine of normal rats, Stylopoulos says.
The burly new gut section cranks up metabolism and starts making a protein that grabs glucose from the blood, the researchers found. Then the gut burns through the sugar to build the new intestinal tissue.
Stylopoulos and his colleagues are trying to figure out why surgery makes the Roux limb get so big. The researchers suspect that undigested food dumped into the intestines from the stomach pouch might somehow trigger the growth.
The research helps quash the idea that gastric bypass surgery’s effects stem simply from limiting caloric intake.
“It’s one more nail in the coffin,” Seeley says.
Kaplan agrees: “That body is dead and buried.”







