Hepatitis E vaccine shows strong coverage
Large trial in China indicates 87 percent protection against the virus
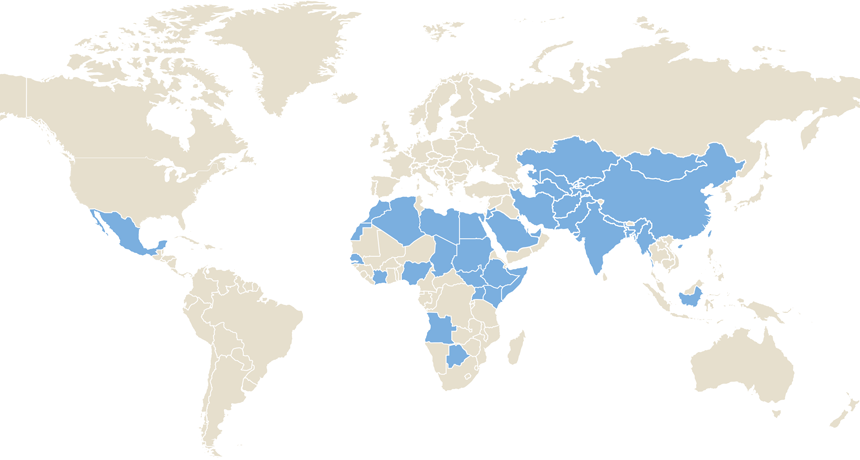
TRANSMISSION RISK A vaccine offers long-lasting protection against hepatitis E, new research shows. The virus can spread when drinking water gets contaminated due to poor sanitation. Shaded areas denote countries where waterborne hepatitis E remains a health problem.
redmal/iStockphoto, adapted by E. Otwell; source: WHO