I, Mold
Conquering the rising tide of infection is hindered by the many similarities between humans and fungi
In the germ world, fungi usually lack the flair of viruses or bacteria. To people with normal, healthy immune systems, a fungus will rarely show itself — even though you carry around a microscopic film of fungus on your hair and skin, and take in invisible clouds of fungal spores with each breath. While many other microbes prefer to make a living through disease and death, a fungus is often content to wait for its host to die of something else.

In fact, throughout the history of civilization fungi have mostly been humans’ friends, providing the bounty of bread and beer, recycling trash and enabling plants to extract nutrients from the soil. Scientists estimate that roughly 1.5 million species of fungus inhabit the Earth, but only a handful are capable of causing human disease.
Problem is, when they do, fungi can be remarkably lethal: For example, about half the patients who develop serious infections from the fungus Aspergillus will not survive. The mortality rate for the most common fungal infection in hospitals, candidiasis, has been reported to be just as high — and though numbers are hard to come by, reports suggest overall fungal infection rates have been on the rise. Doctors have also recently become concerned about a once-rare infection from the Cryptococcus fungus spreading in the Pacific Northwest (SN Online: 4/22/10).
Understanding what transforms a fungus from pal to pathogen has occupied researchers for more than a century. Yet scientists have only recently discovered some key principles that govern how fungi operate, and that allow a normally peaceful fungus to turn against people. In trying to decode the molecular conversation between microbe and human host, fungus explorers have also found some surprising secrets about the human immune system.
“We’re now starting to see studies that rival anything else done before them,” says William E. Goldman, a microbiologist at the University of North Carolina at Chapel Hill. Since 2004, when the complete genetic blueprint of Candida albicans was published, researchers have cataloged the genomes of about a dozen species of fungus that cause disease. Studies of these genomes may soon reveal how fungi survive in people’s bodies — and suggest new ways to extinguish the germs. Scientists have also recently discovered families of molecules on human immune cells that alert the body to the presence of fungi and other invaders, as well as mechanisms that allow a fungus to evade those cells.
Taken together, these findings may soon solve one of the most challenging aspects of treating fungal infection: how to get rid of the germ that is your closest relative.
In old biology classes, fungi were lumped with plants, presumably because both forms of life could sprout from dirt. Today, fungi are recognized as their own kingdom, a diverse group of organisms that live in the inky depths of the ocean, the sub-zero snows of Antarctica and the forgotten apple in the refrigerator.
Fungi include mold, yeast, mushrooms and other growths that don’t make energy from chlorophyll and light. To reproduce, many fungi shed microscopic spores, each one capable of propagating. Even if you don’t see fungi, you live with them daily. A 2005 study found that about a million spores are nestled in your pillow alone.
Fungi are usually the vultures of the ecosystem, preferring food that is almost or already dead. (Aspergillus, for example, usually hangs around rotting leaves and compost piles, feasting on decaying matter.) But sometimes, when conditions are right, a fungus starts to germinate while its host is still among the living. In people, this generally leads to troublesome, but not fatal, infections of the skin and nails.
“Most fungal pathogens are pretty wimpy,” Goldman says. “They are not very good at causing disease in normal hosts with normal immune systems.”
But a growing population of people have not-so normal immune systems. Fungal infections are so deadly in part because most patients who become seriously ill are already weakened by AIDS, cancer, transplants or medications that handicap the body’s ability to mount a strong defense. More and more of these patients have taken high doses of antibiotics to prevent other infections, fundamentally changing the body’s ecology and allowing unnatural fungal growths to take over. More patients are also undergoing medical procedures that breach normal immune barriers with catheters and other devices.
While relatively rare a generation ago, candidiasis — a blood infection from the fungal genus Candida, which normally lives on the skin — has become the fourth most common infection acquired in hospitals. Although infections from Aspergillus are not carefully tracked, studies suggest that the number of deaths quadrupled during the 1980s and ’90s.
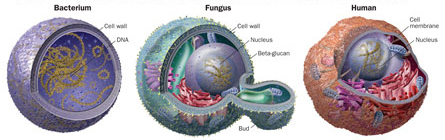
Drugs to treat fungal infections have been difficult to develop because fungi share many properties with people; from an evolutionary standpoint, fungi are closer to the animal kingdom than any other form of life. If you put mushrooms on your pizza, the mushrooms have more in common with you than with the tomato sauce. Fungi are also much more closely related to humans than are viruses and bacteria, which makes attacking fungal infection a tricky business.
Unlike other kinds of germs, both people and fungi are eukaryotes — among other commonalities, their cells have a nucleus, and the nucleus has its own membrane. In fact, fungal cells are so much like animal cells that much about the basics of human life has been gleaned from studies of baker’s yeast.
To fight infection, antimicrobial drugs often exploit some molecular difference between an invading organism’s cells and human cells. But with fungal treatment, human tissue is more likely to find itself in the line of fire. Although more modern antifungal drugs are less harsh than their predecessors, one of the first widely used antifungals — amphotericin B — had a reputation for being highly toxic.
“When we are evolutionarily so similar, it’s hard to get drugs that target fungi alone,” says Bruce Klein of the University of Wisconsin–Madison. Drugs that treat bacterial infections often aim for molecules in the bacterial membrane. However, if drugs attack fungal membranes, the treatments often hit human cells too.
Fungal targets
There are, however, distinctions between human and mold. Most notably, fungal cells enclose themselves in a tough outer wall that shields them from abrupt changes in moisture and temperature.
“The fungal cell wall is the major difference between us and them,” says Stuart Levitz of the University of Massachusetts Medical School in Worcester. “But it can be their Achilles’ heel. It’s what protects them in the environment, but also what flags them as being different.”
The most important building blocks of this wall, at least to the immune system, are large glucose-based molecules called beta-glucans. In recent years, researchers have begun to compile a laundry list of receptor molecules on the surfaces of human immune cells that recognize and interact with the beta-glucan molecules from fungi. Receptors act as gatekeepers, linking the outside of a cell to its internal workings. By examining these receptors, researchers can eavesdrop on the molecular crosstalk between fungi and people.
Studies have found that just after fungi enter the body, defense relies mostly on “innate immunity” — a general, shotgun-like immune response that enlists certain types of white blood cells to find and destroy invaders. The other type of immunity, “adaptive immunity,” takes longer to kick in, involving specialized infection-fighting white blood cells known as T cells and the production of antibodies that confer long-lasting protection against a specific target.
While humans produce plenty of antifungal antibodies, innate immunity is thought to be the first responder against a fungus. This basic defense mechanism is found throughout the animal kingdom; even horseshoe crabs protect themselves from fungi using innate immunity.
Scientists speculate that one reason fungi don’t cause as much human disease as other microbes is because “our innate immunity has evolved very, very well so we’re able to recognize and respond to fungi by a variety of different mechanisms,” says Levitz. “Possibly as a consequence of that, the fungi have not evolved to become significant human pathogens to the extent that bacteria, parasites and viruses have.” (Plants have not been so fortunate; despite plants and fungi’s long and close interaction, fungi are significant plant pathogens that spoil about 10 percent of the world’s harvests each year.)
Among the most important type of proteins that recognize fungi are the toll-like receptors, so named because they resemble a similar fruit fly molecule called toll. As receptors, they switch on when they encounter proteins from fungi and bacteria, setting off other reactions inside the cell. A team of French researchers reported in 1996 in the journal Cell that flies with mutations in the gene for a toll receptor were unusually vulnerable to infection with Aspergillus. In human white blood cells, two toll-like receptors in particular — TLR2 and TLR4 — appear to be involved in the body’s fungus-fighting ability.
In 2008, scientists from the Fred Hutchinson Cancer Research Center in Seattle helped demonstrate the fungus-fighting role of TLR4 in a study in the New England Journal of Medicine. The researchers examined patients who had received bone marrow transplants and later developed Aspergillus infections. In general, about 10 to 15 percent of transplant patients will develop the life-threatening condition aspergillosis, but it’s not clear why the other 85 to 90 percent of patients escape unscathed.
The researchers discovered an inherited gene that causes a malfunctioning TLR4 in the patients who had become ill. Without a normal TLR4, the scientists proposed, these patients’ immune responses may have been weakened. Genetic testing for this mutation among blood stem cell donors may one day identify patients who need special care or attention following a transplant, the authors pointed out.
Two other reports in the New England Journal of Medicine last year described genetic flaws that caused increased susceptibility to fungal disease, confirming the role of other receptors in fungal protection. One involved dectin-1, a receptor first recognized as key to fungal defense in 2001. Dectin-1 partners with the TLR receptors to produce substances that both attack fungi and deploy other white blood cells to help fight infection.
Last October, an international team of researchers described genetic studies of one family in which otherwise healthy women seemed particularly prone to chronic Candida (“yeast”) infections of the vagina, fingers and toes. The researchers found an inherited genetic alteration that led to a defect in dectin-1.
A second team investigated another family whose members were prone to recurrent, and sometimes fatal, infections of Candida. A team led by University College London researchers found a different inherited mutation that made a person vulnerable to fungal infection. When dectin-1 detects the fungus, it sets off a chain reaction that gets immune cells in battle mode. A mutation can interfere with one link in that chain, a molecule called CARD9, the researchers found. In this case, dectin-1 was triggered correctly, but the mechanism jammed farther down the line.
While these and other discoveries have brought new understanding to how immunity works, much about how the human body handles its relationship with fungi remains unclear.
“The thing that’s most on my mind is how these organisms can manage to survive and proliferate in such a close relationship with host cells without triggering alarms,” Goldman says.
Stealth spores
Goldman’s work focuses largely on Histoplasma, which can cause lung infections. These fungal spores grow inside white blood cells called macrophages — innate immunity cells assigned the job of destroying invading organisms like fungi. “Here is an organism that gets inside the very cell that’s supposed to be destroying them,” he says.
While the stealth tactics deployed by Histoplasma remain largely a mystery, scientists recently reported that Aspergillus may dodge the immune system by borrowing a tool from Harry Potter: a cloak of invisibility.
Though each cubic meter of inhaled air may contain a thousand or more Aspergillus spores, the immune system doesn’t seem to notice. Scientists had been unclear why. Then, writing in August 2009 in Nature, researchers from the Pasteur Institute and elsewhere offered an explanation: The body’s immune system can’t react to the spores because the immune system doesn’t know they are there. Normally, the spores are coated with a thatching of small fibers called the “rodlet layer.” In experiments with mice, the researchers found that the fibers alone do not excite the immune system. However, when researchers stripped the rodlets from the outside of the cells, the exposed spores invoked a robust immune response.
It appears that the rodlet layer may allow the fungal spores to hide in the body, waiting until conditions are favorable to germinate (such as death). The researchers also noted that when Aspergillus spores start to grow, the outer coating disintegrates and the immune system kicks in.
Scientists have recently discovered other deceptive feats. In the March 18 Nature, researchers revealed that members of a fungus genus that attacks plants are capable of passing off genes to one another — a lateral handoff thought to occur almost exclusively in bacteria. The discovery means fungi that develop genetic resistance to a drug treatment could theoretically share that secret with neighboring organisms.
The shocking thing, says study coauthor Michael Freitag of Oregon State University in Corvallis, was the ease with which fungi traded genes. Freitag and his colleagues simply put genetically distinct samples of the fungi side by side on a petri dish, incubated them together and tracked genetic movement. “It’s not that far removed from what would occur in natural conditions,” Freitag says. “I was surprised it would work that well.”
While no one knows whether other species of fungus are capable of sharing genes so readily, the findings reinforce the idea that nothing about fungi should be underestimated. To succeed in conquering infections, the next generation of treatment will need to hit several targets at once.
Freitag likens current antifungal treatments to the cancer treatments of the ’60s and ’70s, most of which were designed to target cells that grew rapidly, and not cancer cells specifically. Today, doctors have a number of drugs that can zero in on the specific defects of a malignant cell, and physicians prescribe drug cocktails that try to disable several mechanisms simultaneously. Fungal infections appear to have similar complexity, including sharing properties with nontargeted cells, and will require treatment just as sophisticated, Freitag says. “Just like now we can attack very specific targets in cancer,” he says, “we are going to have to do that with fungi.”