Young’uns adrift on the sea
Scientists try to identify and track elusive larvae in a boundless ocean
Steve Simpson is gearing up his lab for research he dismissed as loony just two years ago: studying how coral larvae, mere squiggles of still-developing tissue, respond to sound.

He hadn’t expected corals — that is, the simple, tiny animals that build coral skeletons — to even have a way of detecting noises. “They’re kind of lecture two in invertebrate biology,” says Simpson, of the University of Bristol in England.
Most of his earlier work had focused on larval reef fishes, which unlike corals grow up to have ear bones and brains. But a few years ago when he deployed students to Australia’s Great Barrier Reef to study the fish, a minor mutiny gave him a new perspective.
His fish team fell in with a crew of coral biologists and developed an urge to broadcast sounds to corals so young they still bobbed along in open water. Perhaps the sounds of a reef would clue in larvae about where to settle down, the biologists speculated. “You guys have got to be crazy,” Simpson remembers telling them.
In May 2010, however, Simpson and his colleagues reported in PLoS ONE the first evidence that coral larvae (and larvae of any cnidarian, the group that includes corals, sea anemones and jellyfish) respond to sound. He has thus joined a growing circle of biologists who are rethinking the capabilities of elusive marine larvae.
Plenty of sea species, including many shellfish, crabs and worms, as well as corals and fish, go through a larval stage as they develop from fertilized eggs to full-grown adults. Instead of immediately settling down where mom and dad live, the very young sea creatures venture off. They’re tiny; the oceans are vast.
Where larvae go, whether they control their travels or just wash along with the currents, and how much the specks of life sense their environment remain largely open questions for most species. Conservationists, fisheries managers, marine ecologists and evolutionary biologists would dearly love to find the answers, and they are beginning to develop the tools to do so.
Researchers have observed that juveniles eventually settle down to replenish populations on rocks, sand or reefs and the waters around them. Whether these are the offspring spawned in those spots days or weeks before, or are counterparts swept in from elsewhere, has perplexed generations of biologists. As answers start to emerge, though, biologists are discovering that treating these larvae as helpless blobs floating in the water would indeed be lunacy.
The whatsit phase
To try to convey the challenges of studying marine larvae, Tracey Sutton stares at the computer in his shipshape office at the Virginia Institute of Marine Science in Gloucester Point and clicks through baby pictures.
These very young fish, mostly days or weeks old, are of interest to fishing fleets, ecologists and aquatic enthusiasts, but are fiendishly hard to identify. The larvae often look nothing like their parents, Sutton explains.
Many fish eggs hatch into similar, pale, simplified larvae that don’t carry a lot of diagnostic characteristics. Sutton calls them nondescript, with much the same intonation that a polite person might use to admit that looking at a dozen cell phone photos from a friend’s recent marathon got a little repetitive. Other larvae have distinct traits, but look more like cartoon aliens than adult fish.
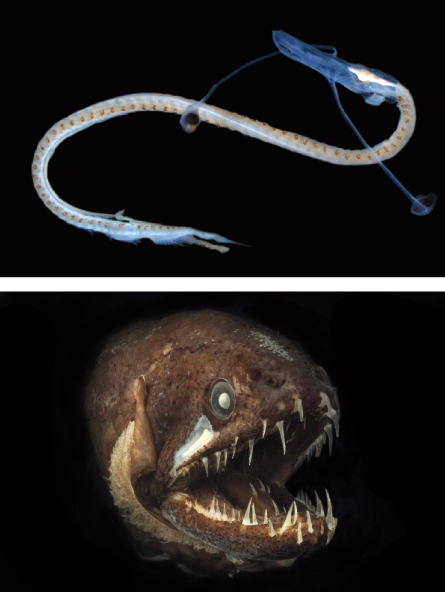
“This one has eyes on stalks,” Sutton says. On the screen he shows a long, pale Y-shaped thingy with a plump bulge of an eye atop each of the Y’s arms. Whatever this creature looks like, a mutant ninja gummy worm or a doodle from the dawn of slingshot design, it does not resemble the dragonfish it will grow up to be.
Marine biologists thus struggle with problems comparable to that of a mammal biologist finding some newborn feline but not knowing whether it’s a house cat or a tiger. In some cases, it’s as bad as not knowing whether a mewling little cutie is a kitten, a puppy or a bear cub. And any worry over identification assumes that scientists can find the creatures in the first place. Larvae don’t nestle under a cozy rock outcropping but are more like dandelion fluff blowing, or maybe flying, in the wind.
During a research cruise sampling life in deep water, Sutton managed to spot an elusive youngster that resolved a long-standing muddle of the kitten-puppy-bear type. The youngster was developed enough to be recognized as a whalefish but hadn’t lost all of its baby features yet.
The transitional youngster allowed Sutton and colleagues, including G. David Johnson of the Smithsonian Institution in Washington, D.C., to outline the whole sequence from larva to adult for the species in 2009. Once called tapetails, the larvae drag a long tattered streamer behind them as if they’ve just emerged from a tangle of seaweed. The larval form, the adult female and the adult male had previously each been identified not only as separate species, but also as species in different families.
Some of the misclassifications are side effects of the power of evolutionary adaptation to environments. Larvae have to survive as bite-sized nuggets in open water, coping with perils different from those faced by bigger adults flitting around reefs or nudging along the seafloor. Outrageously long spines and prickles on larvae may poke and choke a predator. Johnson speculates that larval streamers like those on the tapetails may create a helpful ambiguity about whether a larva is edible.
To human eyes, ambiguous characteristics can bedevil the study of even some common fish. Sutton says that the 300-plus known species of rattails mature from larvae that basically look alike. Rattails may not be familiar menu items, and probably never will be under that name, but the elongated, slinky fish nicknamed for a stretched-out anal fin play an important role on the abyssal plains of the seas. “Drop some dead meat down there, and they’re usually the first things that show up,” Sutton says. By volume, the deep sea amounts to a lot of habitat on a blue planet. Thus, he points out, biologists can’t yet distinguish reliably among the larval forms of species of one of the most dominant fish families on Earth.
Building boundaries
As difficult as larvae are to identify, their actions are of interest for conservation as well as basic biology.
A recent surge of larval research addresses the challenges of drawing borders for protected areas in the seas. Declaring a teeming stretch of coastal waters off-limits to fishing, for example, may not do much good if the new reserve’s residents start out as larvae somewhere with no protection. To complicate matters, the politics of protection often play out differently at sea than they do on land.
For instance, people protect Yellowstone and Yosemite to preserve gems of terrestrial wilderness. But a marine reserve where people aren’t allowed to fish often has a lot to do with saving the livelihood of people fishing nearby, says marine ecologist Robin Pelc of the Monterey Bay Aquarium in California.
“Fisheries are in crisis,” she says. One big hope for rebuilding and sustaining them is to create fishing-free sanctuaries with marine populations that grow large enough to spill over the boundaries and replenish fisheries beyond. Designing such a reserve requires tricky balancing acts, Pelc says. Borders have to embrace enough area for populations to thrive, yet the area can’t be so big that the riot of life inside the borders stays within those lines.
A modeling analysis confirms the idea that reserves designed to enhance fisheries should be no larger than roughly twice the target species’ larval dispersal scale, Pelc and her colleagues report in the Oct. 26 Proceedings of the National Academy of Sciences. Proving that the sacrifices made for a reserve really have generated spillover has been tricky, though. Most of the success stories feature shellfish, Pelc says. That trend may reflect the tendency of shellfish larvae not to travel particularly far and thus to show up readily during monitoring just beyond park borders.
In contrast, larvae that disperse widely may indeed spill over the borders of the park but continue on to scatter over a broad swath of ocean. Far-flung larvae may fulfill the dreams for going forth and replenishing, but they may end up so thinly distributed that typical monitoring programs won’t notice any uptick against naturally hiccuping population changes.
It’s the catastrophic natural variations that interest Lauren Mullineaux of the Woods Hole Oceanographic Institution in Massachusetts, who studies larvae at deep-ocean vents.
As underwater volcanoes, these vents offer a nutrient-rich but precarious home, where thriving communities often disappear in a Pompeii minute. They’re great spots for ecologists to study how ecosystems rebuild after a disaster, a process that all depends on those traveling larvae.
In late 2005 or early 2006, a catastrophic eruption occurred along the East Pacific Rise off the coast of Central America, eradicating a swath of the tube worms and mussels and other vent dwellers that Mullineaux and her team had been studying for a decade or more. Though the interruption was bad in some ways, Mullineaux says, “most of us knew it was going to happen eventually and were pretty excited at the chance to take advantage of a natural experiment.”
After four years of monitoring the new settlers at the site, she and her colleagues say the emerging vent community seems to be on a trajectory different from that of the previous community. Species are mixing in a different blend, the researchers report in the April 27 Proceedings of the National Academy of Sciences.
“The most astonishing thing about this discovery is that we found a species recolonizing our site that appears to have come from over 300 kilometers away,” Mullineaux says. Larvae of the limpet Ctenopelta porifera have arrived. The adults, shaped roughly like a lima bean with rows of prominent bumps, are thriving even though Mullineaux has never seen them at the site before. Another small limpet, predicted to survive only during the earliest days of rebuilding, remains a dominant player at the site.
Where, oh where have they gone?
Despite the importance of larval travel, researchers are still figuring out where in the world the youngsters go. Tracking has proved difficult, and researchers have often made do with rules of thumb for estimating travels.
In a heroic research effort about a decade ago, scientists combined information about water movements around deep-sea vents with studies of larval development done in chambers pressurized at 250 atmospheres to simulate the deep ocean. The study’s conclusion: The larvae of an iconic Riftia tube worm probably settle within 100 kilometers of their parents.
For shallower species, molecular tagging has yielded more direct evidence. The results are shaking up a long-standing assumption that since many species occur widely, their larvae must also travel widely. Some larval damselfish dosed with tetracycline, which leaves a mark on their ear bones, later set up housekeeping in their native home reef off of Australia’s Lizard Island. And a bunch of clownfish settling in a stretch of Kimbe Bay in Papua New Guinea were identified as local kids.
Considering human impacts on the ocean, one important question is what larvae can and can’t travel through. Larvae of a common starfish can’t readily disperse around water outflows from river mouths or water-treatment plants, says Jonathan Puritz of the University of Hawaii’s Hawaii Institute of Marine Biology in Kaneohe Bay. He and Robert Toonen, also of HIMB, used genetic markers to see where starfish larvae settled in the waters off southern California’s coast. The populations on either side of water outflows differed more than populations from opposite ends of the United States’ Pacific coast, he reported at the Evolution 2010 meeting in June in Portland, Ore.
Water movements also play a role in larval dispersal, Lisa Levin of the Scripps Institution of Oceanography in La Jolla, Calif., and colleagues report in the Nov. 9 Current Biology. Two closely related mussel species growing in roughly the same place along the California coast tend to send larvae off in opposite directions, the scientists found, because one spawns during the spring and the other spawns in fall, when nearshore currents move in a different direction.
Masters of their fate
But current isn’t everything. Certain species are turning out to have some say in where they end up.
The fastest fish larvae of the tropical reef can challenge a human scientist fitted with fins and swimming earnestly, says Jeff Leis of the Australian Museum in Sydney. Even larvae that don’t swim much at all can still change their fate by moving into another layer of water that’s flowing in a different direction.
Considering larvae as possible captains of their fate, or at least contributors to it, opens the question of how the youngsters might choose that fate. Experiments offering various water samples to larval clownfish at the settlement stage have found that water collected near an island, for example, holds more appeal than a sample from the open ocean, and water that has flowed over vegetation proves more attractive than water that has not.
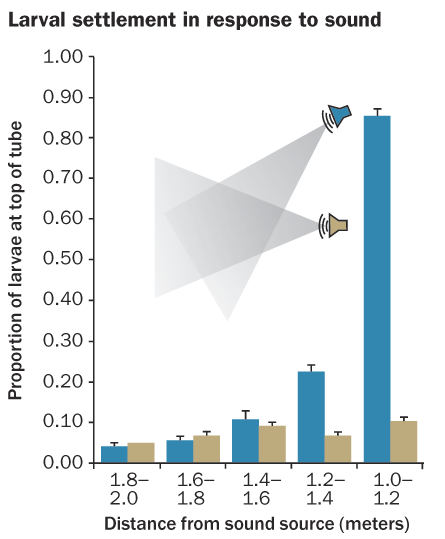
Now it turns out that corals may have their preferences too. Simpson’s wayward fish team set up three underwater speakers to broadcast sounds recorded on reefs outward in different directions. In front of each of the speakers, the researchers positioned a long tube holding coral larvae, with mesh ends to keep the larvae inside.
Each tube was positioned at a different angle relative to possible environmental cues such as water currents and the reef itself. Regardless of the orientation, though, the majority of larvae crowded into the end near the sound. In a tougher test, the researchers repeated the experiment with speakers raised above the tubes of larvae. Even though tiny corals normally sink downward to settle, the majority of larvae in tubes reversed the normal preference and clustered on the tube’s ceiling. Sound appears to be an overlooked cue for determining where corals settle, Simpson and his colleagues report.
Coral researcher Valerie Paul says she doesn’t know about sound, but she has seen that coral larvae show strong preferences for, or against, certain surfaces. Paul, of the Smithsonian Marine Station at Fort Pierce in Florida, and her colleagues offered larvae of staghorn and elkhorn coral what looked like swatches of pink paint (actually four species of crustose coralline algae) as possible landing pads. Biologists have known that corals often settle on such algae. But as the researchers reported in the March 2010 issue of Coral Reefs, larvae responded differently to the algal species.
In the emerging view of larvae, marine youngsters are not only elusive. As Paul puts it, “they’re very picky.”
Matching game: Then and now
Many marine youths look nothing like their adult forms, which makes it difficult for scientists to identify the larvae. In some cases, larvae of different but related species look so much alike that few, if any, biologists can tell them apart. Try your hand at matching the larval creatures below (on the left) with their adult counterparts (on the right). Answers can be found below the images, upside down.

Credit: Clockwise from top left: Wim van Egmond/Visuals Unlimited/Corbis; Fred Hsu/Wikimedia Commons; MikeMurphy/Wikimedia Commons; © Hans Hillewaert/Wikimedia Commons; © Hans Hillewaert/Wikimedia Commons; Wim van Egmond/Visuals Unlimited; G. David Johnson/NMNH, Smithsonian Institution; Otto Larink/Wikimedia Commons