Microbes can play games with the mind
The bacteria in our guts may help decide who gets anxiety and depression
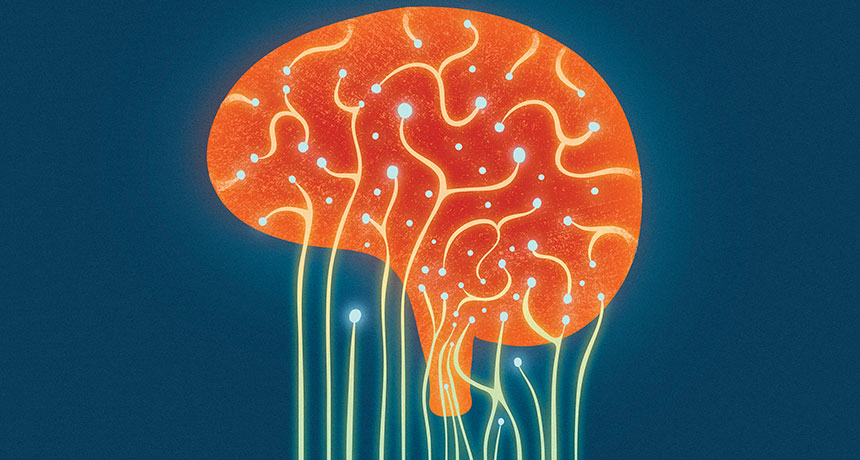
GUT FEELINGS Through several lines of communication, gut bacteria and the brain affect each other.
Tang Yau Hoong
The 22 men took the same pill for four weeks. When interviewed, they said they felt less daily stress and their memories were sharper. The brain benefits were subtle, but the results, reported at last year’s annual meeting of the Society for Neuroscience, got attention. That’s because the pills were not a precise chemical formula synthesized by the pharmaceutical industry.
The capsules were brimming with bacteria.
In the ultimate PR turnaround, once-dreaded bacteria are being welcomed as health heroes. People gobble them up in probiotic yogurts, swallow pills packed with billions of bugs and recoil from hand sanitizers. Helping us nurture the microbial gardens in and on our bodies has become big business, judging by grocery store shelves.
These bacteria are possibly working at more than just keeping our bodies healthy: They may be changing our minds. Recent studies have begun turning up tantalizing hints about how the bacteria living in the gut can alter the way the brain works. These findings raise a question with profound implications for mental health: Can we soothe our brains by cultivating our bacteria?
By tinkering with the gut’s bacterial residents, scientists have changed the behavior of lab animals and small numbers of people. Microbial meddling has turned anxious mice bold and shy mice social. Rats inoculated with bacteria from depressed people develop signs of depression themselves. And small studies of people suggest that eating specific kinds of bacteria may change brain activity and ease anxiety. Because gut bacteria can make the very chemicals that brain cells use to communicate, the idea makes a certain amount of sense.
Though preliminary, such results suggest that the right bacteria in your gut could brighten mood and perhaps even combat pernicious mental disorders including anxiety and depression. The wrong microbes, however, might lead in a darker direction.
This perspective might sound a little too much like our minds are being controlled by our bacterial overlords. But consider this: Microbes have been with us since even before we were humans. Human and bacterial cells evolved together, like a pair of entwined trees, growing and adapting into a (mostly) harmonious ecosystem.
Our microbes (known collectively as the microbiome) are “so innate in who we are,” says gastroenterologist Kirsten Tillisch of UCLA. It’s easy to imagine that “they’re controlling us, or we’re controlling them.” But it’s becoming increasingly clear that no one is in charge. Instead, “it’s a conversation that our bodies are having with our microbiome,” Tillisch says.
Figuring out what’s being said in this body-microbe exchange, and how to shift the tone in a way that improves mental health, won’t be easy. For starters, no one knows the exact ingredients for a healthy microbial community, and the recipe probably differs from person to person. And it’s not always simple to deliver microbes to the gut and persuade them to stay. Nor is it clear how messages travel between microbes and brain, though scientists have some ideas.
It’s early days, but so far, the results are compelling, says neuroscientist John Cryan of University College Cork in Ireland, who has been trying to clarify how microbes influence the brain. “It’s all slightly weird and it’s all fascinating,” he says.
Cryan and others are amassing evidence that they hope will lead to “psychobiotics” — bacteria-based drugs made of live organisms that could improve mental health.
We’re not alone
Ted Dinan, the psychiatrist who coined the term “psychobiotics,” was fascinated by a tragedy in Walkerton, Canada, in May 2000. Floods caused the small town’s water supply to be overrun with dangerous strains of two bacteria: Escherichia coli and Campylobacter. About half the town’s population got ill, and a handful of people died. For most residents, the illness was short-lived, about 10 days on average, says Dinan, who collaborates with Cryan at University College Cork. But years later, scientists who had been following the health of Walkerton residents noticed something surprising. “The rates of depression in Walkerton were clearly and significantly up,” Dinan says. That spike raised suspicion that the infection had caused the depression.
Other notorious bacteria have been tied to depression, such as those behind syphilis and the cattle-related brucellosis, and not just because ill people feel sad, Dinan says. He suspects there’s something specific about an off-kilter microbiome that can harm mental health.
This possibility, though it raises troubling questions about free will, is certainly true for lab animals. Mice born and raised without bacteria behave in all sorts of bizarre ways, exhibiting antisocial tendencies, memory troubles and recklessness, in some cases. Microbes in fruit flies can influence who mates with whom (SN: 1/11/14, p. 14), and bacteria in stinging wasps can interfere with reproduction in a way that prevents separate species from merging. Those findings, some by evolutionary biologist Seth Bordenstein of Vanderbilt University in Nashville, show that “there’s this potential for [microbes] to influence behavior in this complex and vast way,” he says.
By sheer numbers, human bodies are awash in bacteria. A recent study estimates there are just as many bacterial cells as human cells in our bodies (SN: 2/6/16, p. 6). Just how legions of bacteria get messages to the brain isn’t clear, though scientists have already found some likely communication channels. Chemically, gut microbes and the brain actually speak the same language. The microbiome churns out the mood-influencing neurotransmitters serotonin, norepinephrine and dopamine. Bacteria can also change how the central nervous system uses these chemicals. Cryan calls microbes in the gut “little factories for producing lots of different neuroactive substances.”
Signals between the gut and the brain may zip along the vagus nerve, a multilane highway that connects the two (SN: 11/28/15, p. 18). Although scientists don’t understand the details of how messages move along the vagus nerve, they do know that this highway is important. Snip the nerve in mice and the bacteria no longer have an effect on behavior, a 2011 study found. And when the gut-to-brain messages change, problems can arise.
New bacteria, new behavior
Wholesale microbe swaps can also influence behavior. In unpublished work, Dinan and his colleagues took stool samples from people with depression and put those bacteria (called “melancholic microbes” by Dinan in a 2013 review in Neurogastroenterology and Motility) into rats. The formerly carefree rodents soon began showing signs of depression and anxiety, forgoing a sweet water treat and showing more anxiety in a variety of tests. “Their behavior does quite dramatically change,” Dinan says. Rats that got a microbiome from a person without depression showed no changes in behavior.
Cryan and colleagues have found that the microbiomes of people with depression differ from those of people without depression, raising the possibility that a diseased microbiome could be to blame.
The fecal-transplant results suggest that depression — and perhaps other mental disorders — are contagious, in a sense. And a mental illness that could be caught from microbe swaps could pose problems. Fecal transplants have recently emerged as powerful ways to treat serious gut infections (SN Online: 10/16/14). Fecal donors ought to be screened for a history of mental illness along with other potentially communicable diseases, Dinan says.
“Gastroenterologists obviously check for HIV and hepatitis C. They don’t want to transmit an infection,” he says. The psychiatric characteristics of the donor should be taken into account as well, he says.
A fecal transplant is an extreme microbiome overhaul. But there are hints that introducing just one or several bacterial species can also change the way the brain works. One such example comes from Cryan, Dinan and colleagues. After taking a probiotic pill containing a bacterium called Bifidobacterium longum for a month, 22 healthy men reported feeling less stress than when they took a placebo. The men also had lower levels of the stress-related hormone cortisol while under duress, the researchers reported at the Society for Neuroscience meeting in Chicago last October. After taking the probiotic, the men also showed slight improvements on a test of visual memory, benefits that were reflected in the brain. EEG recordings revealed brain wave signatures that have been tied to memory skill, Cryan says.
The researchers had previously published similar effects in mice, but the new results move those findings into people. “What’s going to be important is to mechanistically find out why this specific bacteria is inducing these effects,” Cryan says. And whether there could be a benefit for people with heightened anxiety. “It’s a very exciting study, but it’s a small study,” Cryan cautions.
Bacteria in an even more palatable form — yogurt — affected brain activity in response to upsetting scenes in one study. After eating a carefully concocted yogurt every morning and evening for a month, 12 healthy women showed a blunted brain reaction to pictures of angry or scared faces compared with 11 women who had eaten a yogurtlike food without bacteria.
Brain response was gauged by functional MRI, which measures changes in blood flow as a proxy for neural activity. In particular, brain areas involved in processing emotions and sensations such as pain were calmed, says Tillisch, coauthor of the study, published in 2013 in Gastroenterology. “In this small group, we saw that the brain responded differently” when shown the pictures, she says. It’s not clear whether a blunted response would be good or bad, particularly since the study participants were all healthy women who didn’t suffer from anxiety. Nonetheless, Tillisch says, the results raise the questions: “Can probiotics change your mood? Can they make you feel better if you feel bad?”
So far, the human studies have been very small. But coupled with the increasing number of animal studies, the results are hard to ignore, Tillisch says. “Most of us in this field think there is something definitely happening,” she says. “But it’s pretty complicated and probably quite subtle…. Otherwise, we’d all be aware of this.” Anyone who has taken a course of antibiotics, or fallen ill from a bacterial infection, or even changed diets would have noticed an obvious change in mood, she says.
Two-way traffic
If it turns out that bacteria can influence our brains and behaviors, even if just in subtle ways, it doesn’t mean we are passive vessels at the mercy of our gut residents. Our behavior can influence the microbiome right back.
“We usually give up our power pretty quickly in this conversation,” Tillisch says. “We say, ‘Oh, we’re at the mercy of the bacteria that we got from our mothers when we were born and the antibiotics we got at the pediatrician’s office.’ ” But our microbes aren’t our destiny, she says. “We can mess with them too.”
One of the easiest ways to do so is through food: eating probiotics, such as yogurt or kefir, that contain bacteria and choosing a diet packed with “prebiotic” foods, such as fiber and garlic, onion and asparagus. Prebiotics nourish what are thought to be beneficial microbes, offering a simple way to cultivate the microbiome, and in turn, health.

That a good diet is a gateway to good health is not a new idea, Cryan says. Take the old adage: “Let food be thy medicine and let medicine be thy food.” He suspects that it’s our microbiome that makes this advice work.
Combating stress may be another way to change the microbiome, Tillisch and others suspect. Mouse studies have shown that stress, particularly early in life, can change microbial communities, and not in a good way.
She and her colleagues are testing a relaxation technique called mindfulness-based stress reduction to influence the microbiome. In people with gut pain and discomfort, the meditation-based practice reduced symptoms and changed their brains in clinically interesting ways, according to unpublished work. The researchers suspect that the microbiome was also altered by the meditation. They are testing that hypothesis now.
If the mind can affect the microbiome and the microbiome can affect the mind, it makes little sense to talk about who is in charge, Bordenstein says. In an essay in PLOS Biology last year, he and colleague Kevin Theis, of Wayne State University in Detroit, make the case that the definition of “I” should be expanded. An organism, Bordenstein and Theis argued, includes the microbes that live in and on it, a massive conglomerate of diverse parts called a holobiont. Giving a name to this complex and diverse consortium could shift scientists’ views of humans in a way that leads to deeper insights. “What we need to do,” Bordenstein says, “is add microbes to the ‘me, myself and I’ concept.”
This article appears in the April 2, 2016, issue of Science News with the headline, “Microbes and the Mind.”