The clockwork launch of bubbles from the wall of a beer glass mesmerizes a worker relaxing after a long day. Sparkling orbs rising in champagne brighten a party later that night.



Bubbles impart levity, animation, and highlights of flavor and feel to beers, some wines, and many other beverages (SN: 11/28/98, p. 344). Without them, beer is flat, champagne is unrecognizable. Is it any wonder that brewers and champagne makers tend to their bubbles with great care?
“We want to keep the bubbles happy,” says chemist Dave Thomas of Coors Brewing Co. in Golden, Colo. Many scientific studies have focused on the bubbly foam that makes up the head of a beer and the “collerette,” or collar, of champagne (SN: 7/29/89, p. 72; 3/30/91, p. 207). By contrast, the bubbles in the body of the drinks have received scant scrutiny. Now, researchers in France and Australia are taking a fresh look at the physical properties and behavior of the bubbles in the liquids.
The bubbles typically contain carbon dioxide gas and in both beverages form rapidly ascending, similar-looking bubble trains. Yet striking differences distinguish champagne bubbles from their beer counterparts, new research shows.
Those differences have emerged from an intimate portrait of champagne bubbles by a team of French experimenters. Among the team’s findings is a dissimilarity between champagne and beer in the microscopic features of their bubbles’ surfaces. Those variations appear to affect the visible behavior of the sparkly orbs, such as their speed of ascent.
Meanwhile, scientists from Down Under have blown bubbles in a computer model to settle a long-standing debate in pubs that serve the dark, flavorful Irish beer known as Guinness stout.
The question: Do the bubbles in Guinness always rise, or do they sometimes appear to defy the laws of physics and go down?
The researchers predict that with such computational tools, brewers and wine makers will gain new insights into the influence of bubbles on beverage chemistry.
Flat beer
For most of the 5,000 to 10,000 years that people have enjoyed beer, it was served the same way—flat. Brewers processed the beverage in open tanks that let the carbon dioxide escape. Around 1600, someone discovered that beer kept in corked bottles sometimes became bubbly. Later in the same century, bottlers made a similar discovery about certain wines.
With bubbles now the norm for beer and champagne, these drinks contain high concentrations of dissolved gas. Brewers and vintners enlist the help of tiny laborers, the microscopic fungi known as yeast, to generate both the gas and alcohol, although beverage makers sometimes also inject carbon dioxide directly into their products.
Wine makers mix the juice from ripe grapes in an open vat with yeast and, for red wine, the grape skins. The fungi convert the fruit’s sugars into alcohol and carbon dioxide, which escapes.
To make champagnes or other sparkling wines, manufacturers add sugar and a second dose of yeast and then seal the wine in a sturdy bottle to trap the carbon dioxide.
Beer makers also use yeast to convert sugars to alcohol and fizz. However, starting with grains instead of fruits, brewers must perform several preliminary steps in which they allow grain kernels to sprout and to produce enzymes that break down most of the grain starches into sugars.
The brew then typically undergoes two rounds of fermentation, the second in a closed vessel. By the end, the yeast has converted the sugars into alcohol and gas.
The final sparkling beverages also contain some proteins and other large organic molecules from both the yeast and the grapes or grain. Beer contains higher concentrations of such molecules than wine does and includes long-chain starches.
Scientists suspect that three properties of the beverages most strongly affect the qualities of their bubbles: the amount of dissolved gas, the percentage of alcohol, and the concentration of large molecules. Champagne boasts three times the dissolved carbon dioxide and more than twice the alcohol content of beer. In terms of large molecules, however, beer rules. One study indicates, for instance, that beer has 30 times champagne’s concentration of protein.
Trapping gases
Since learning how to trap dissolved gases reliably in their beverages, manufacturers haven’t worried too much about the exact character or behavior of the little bubbles that those gases form. Now, in part because a French physics student with a passion for bubbles recently took up graduate work in wine studies, researchers are beginning to examine how those three main properties of beverages shape the bubbles produced.
Beverage bubbles typically spring to life at the walls of a glass where dust or other deposits act as so-called nucleation sites, prompting gas to come out of solution (SN: 11/17/90, p. 319).
A bubble grows because carbon dioxide diffuses from the liquid into the tiny packet of gas. The higher pressure of the gas dissolved in the liquid compared with that inside the bubble drives bubble inflation. Once the bubble develops enough buoyancy, it detaches from the nucleation site and continues to grow as it rises (SN: 1/18/97, p. 36). In both champagne and beer, this growth makes the bubble increasingly buoyant, causing it to scurry more and more quickly toward the surface.
Gírard Liger-Belair is a student at the University of Reims, at the heart of France’s Champagne region. He’s teamed up with colleagues at the university and scientists in the champagne industry and French government to launch a campaign to thoroughly characterize champagne bubbles.
Champagne makers would like to better understand their bubbles and learn to control them because “in France, people make a connection between the quality of the champagne and the smallness of the bubbles,” Liger-Belair says. He argues that this is a misconception.
In the August 1999 issue of the quarterly American Journal of Enology & Viticulture, Liger-Belair’s team reported using a high-speed camera, strobe light, photographic enlarger, and ruler to measure sizes, speeds, and rates of formation of champagne bubbles.
No one has published comparable data on beer bubbles, the investigators say. However, they found that a pair of California chemists had dabbled in beer-bubble physics a decade ago and published some useful results.
“Once you begin to learn about the nature of beer bubbles, you will never again look at a glass of beer in quite the same way,” Neil E. Shafer-Ray, now of the University of Oklahoma in Norman, and Richard N. Zare of Stanford University said in a 1991 article in Physics Today.
The champagne researchers compared their more extensive measurements with the California observations on a single train of beer bubbles. The champagne bubbles expanded much more rapidly than their beer counterparts and seemed to form at higher rates.
An obvious reason for differences in bubble growth and formation rates is champagne’s threefold higher gas pressure, the French experimenters note. Alcohol content may also play a role in bubble differences, they suspect, but they haven’t yet conducted studies to explore it.
In the Feb. 22 Langmuir, however, the champagne scientists unveiled findings from newer experiments on both champagne and beer. These data show that the different concentrations of protein molecules in the two beverages strongly affect their bubble physics.
Rising bubbles often pick up hitchhikers—suspended or dissolved materials that stick, or adsorb, to bubble surfaces. The mining industry exploits that feature of bubbles to separate minerals with different affinities for certain gases.
Adsorbed molecules stiffen a bubble by forming a shield on its surface, Liger-Belair says. According to fluid mechanics, a stiffer sphere moving through a liquid runs into more resistance, or drag, than one with a more flexible skin. Consequently, adsorbed molecules tend to slow bubbles down. In the absence of other influences, such as a nearby glass wall, the most rigid bubbles show the greatest drag.
Since bubbles continue to grow as they rise, the term rigid applies in a fluid-mechanical sense only. “A bubble can grow even if it is completely covered” with adsorbed molecules, Liger-Belair says.
In analyzing their one train of bubbles, Shafer-Ray and Zare concluded that the beer bubbles were behaving as rigid spheres. Measurements of bubble height versus time closely resembled a standard graph of a rigid sphere’s movement.
Now, the champagne team has provided additional evidence that beer bubbles are rigid. Champagne bubbles, however, are not. It appears that the rich soup of proteins in beer causes the rigidity. Champagne, by contrast, has too little protein, and its bubbles grow too quickly to succumb to the same fate.
Scientists didn’t know how much protein it takes to stiffen a bubble fully until 2 years ago. Research published then showed that only 0.5 milligrams of protein per square meter of bubble surface would do the trick. That result came from a study of air bubbles injected into uncarbonated water containing a dissolved protein, bovine serum albumin. Christophe Ybert, now at the University of California, Los Angeles, and Jean-Marc di Meglio of Université Louis Pasteur in Strasbourg, France, carried out the study.
In the Langmuir report, Liger-Belair and his colleagues demonstrate that the fluid’s drag on rising champagne bubbles starts out high, probably because of the nearby glass wall. As the bubbles rise higher, drag drops below that expected on rigid spheres. By contrast, the drag on beer bubbles remains equal to or greater than the drag on rigid spheres throughout their ascent.
“The drag is higher in beer, so bubbles of the same size are rising slower than in champagne,” Liger-Belair explains.
The scientists also calculated how much of a protein coating typical champagne bubbles acquire in their journeys. On the basis of Ybert and di Meglio’s findings, the French team concludes that to become stiff, the bubbles would have to travel more than 10 centimeters, a higher ascent than bubbles make in a typical champagne flute. Moreover, the rapid bubble growth of champagne continuously dilutes the surface concentration of adhered molecules, thereby keeping the bubble surface partially flexible.
In unpublished work, Liger-Belair and his coworkers have examined bubble trains taller than 10 cm and found that the ever-increasing bubble growth overwhelms protein adsorption. Until those experiments were completed, “I was not sure that the bubble wouldn’t rigidify more,” Liger-Belair says. Now, he told Science News, it seems that during such long journeys, the concentration of surface protein actually drops because bubble surfaces grow so rapidly, making the bubble boundaries become increasingly flexible rather than more rigid.
Downward bubbles
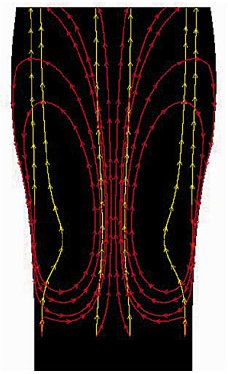
In champagne or beer, the bubbles that the French researchers observed headed one way only—up. Yet computer simulations at the University of New South Wales in Sydney, Australia, indicate that some bubbles in Guinness go down.
While that behavior may seem strange for ordinarily buoyant bubbles, “the actual mechanism can be explained in acceptable scientific terms,” says Clive A.J. Fletcher, director of the university’s Centre for Advanced Numerical Computation in Engineering and Science (CANCES), which developed the simulation.
To model fluid dynamics in Guinness, researchers tapped brewery scientists for numbers describing the beer’s viscosity as well as the sizes, abundance, and contents of its bubbles. In this extremely foamy brew, the bubbles encompass both carbon dioxide and some nitrogen gas that brewers add to the beverage.
Using the Guinness parameters in fluid-mechanics software, the researchers generated animations of bubble trajectories. The movies show that near the sides of the glass, some bubbles do go down, as keen-eyed pub crawlers have been claiming for years.
Fletcher explains that because Guinness is so viscous, the upward rush of bubbles in the center of the glass pulls the liquid with it, creating fountainlike currents that rise in the center and fall near the walls. The displacement of liquid from the bottom center of the glass causes fluid to flow in from the sides to replace it.
Slow-moving, small bubbles often linger near the walls. These freshly minted bubbles haven’t yet had time to grow and accelerate. Also, the nearby walls tend to slow bubbles’ rise. Fletcher says that the new bubbles, with diameters smaller than about 50 to 60 micrometers, have too little buoyancy and momentum to resist the downward currents and so get carried along with them.
Not every glass of Guinness will necessarily exhibit this effect, he notes. The simulations only apply to beer in the trademark, barrel-chested Guinness glass. “We had to do some field trips to find an appropriate specimen” to take measurements for the simulations, Fletcher says. “We think the [glass] shape is one of the factors that influence the phenomenon,” he adds.
Although the Australian team has conducted no wet experiments to confirm their findings, Fletcher says he is convinced that the simulations are accurate. He points out that for years aircraft makers have depended on fluid-dynamics modeling to design planes. “If Boeing can fly aircraft based on what computational fluid dynamics is telling them,” he explains, “we are confident that what [the computer] says takes place in a glass of Guinness really does occur.”






