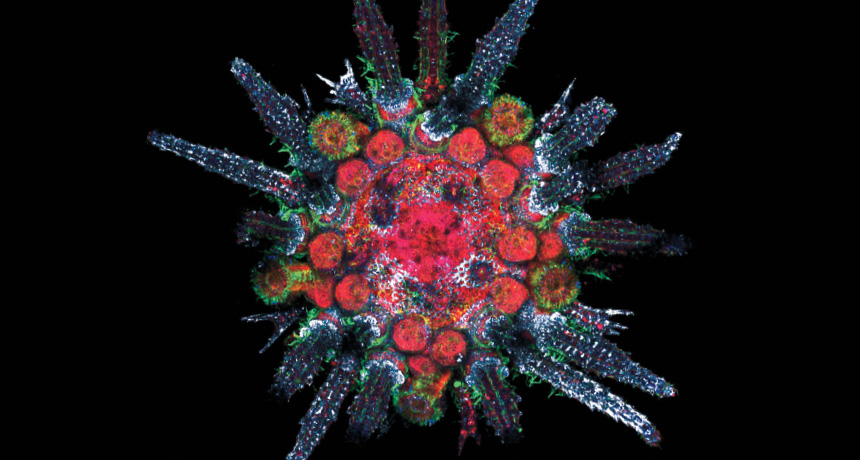
STRANGE VISIONS A lab image of a juvenile purple sea urchin (Strongylocentrotus purpuratus) — without obvious eyes — shows an abundance of light-detecting proteins, such as c-opsins (red).
E.M. Ullrich-Lüter Et Al/Integr. Comp. Biol. 2013
It sounds like a riddling trick: How can an animal with no eyes still see? But it’s a serious scientific question — the trickiest kind of riddle.