A CUNNING ADVERSARY You’d be wrong to call this black-legged tick an insect. Ticks have eight legs, like spiders and scorpions, not an insect’s six. Only a few of the world’s 900 or so tick species carry diseases, but those illnesses can be nasty.
DAVID M. PHILLIPS AND ERIC COHEN/SCIENCE SOURCE
Thanks, Holly Gaff. Soon, anyone straining to tweeze off a mid-back tick can find answers to the obvious question: What if humankind just went after the little bloodsuckers with killer robots?
Gaff, who calls herself a mathematical ecoepidemiologist, at Old Dominion University in Norfolk, Va., is one of the few people collecting real field data on the efficacy of tick-slaying robots. This summer, she’s been supervising a field test of a terminator named TickBot deployed to try making mowed grass safe for children. Researchers will start analyzing results in early fall.
Ticks make formidable enemies. “Almost every control measure that has been tried has failed, and has failed miserably,” Gaff says. “We are slowly coming to embrace the fact that you cannot eradicate ticks.” What human ingenuity might do, however, is manage the risks and — dream big! — make ticks irrelevant.
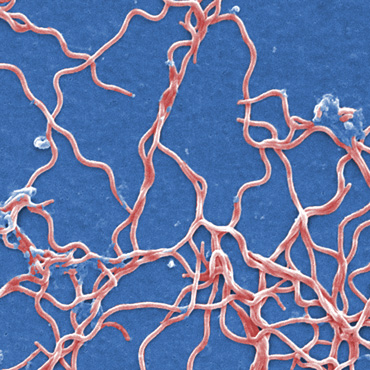
That’s an urgent hope. Data from the Cary Institute of Ecosystem Studies in Millbrook, N.Y., have for two years suggested 2017 will be a high risk one for Lyme disease in the Northeastern United States. Of the various illnesses that North America’s ticks pass along, Lyme is the most common, caused by a squiggle of a parasite called Borrelia burgdorferi. The disease can bring on an eerie red bull’s-eye rash, flulike misery and risks of long-term neurological and joint troubles if not treated early. In 2015, the U.S. Centers for Disease Control and Prevention tallied about 30,000 confirmed cases. Considering gaps in case reporting, some estimates put the number closer to several hundred thousand.
So bring on the robots and other science revenge fantasies. It’s time to rethink humankind’s defenses against ticks. Pesticides and tick checks just aren’t doing the trick.
There may be ways to attack ticks without touching a single molecule of their die-hard little bodies. Ecologists have made progress in tracing what ticks need from the woods and lawns where they lurk. For instance, researchers believe that it was a bumper crop of acorns in 2015 that, through a Rube Goldberg series of consequences, created conditions for a perfect tick storm two years later. Breaking key ecological connections could knock back the tick menace in the future.
Molecular biologists are focusing on tick survival tricks. Researchers are looking for weak spots inside tick guts and trying to take advantage of ticks’ reckless abandon in mating. Biology is proving as important as electronics in the robot line of defense.
Though, Gaff warns, the top design is not the laser-blazing Armageddon that a recently tick-bitten human might crave.
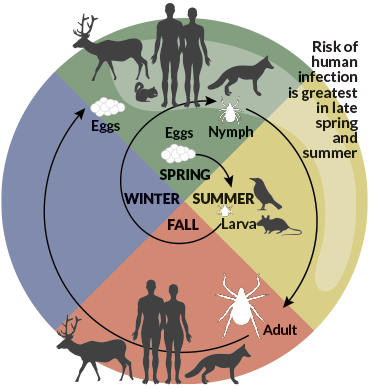
Ticks attack
First, a quick intro to ticks.
Unlike mosquitoes, ticks are pure vampires, consuming nothing but blood. Mosquitoes get colloquially called vampires, but blood is just their version of a pregnancy craving, a female-only nutrient gorge to aid reproduction in an adult life of sipping flower nectar.
For most of the troublesome tick species in North America, including the black-legged ticks that spread Lyme, blood is the elixir that lets them transition to the next life stage — from larva to nymph to adult. And after a single meal, an adult female can lay 1,000 or even 15,000 eggs without anything else to eat for the rest of her life. Hard ticks, the Ixodidae family, which includes the black-legged variety, typically have only two or three meals of any kind during the entire two or three years they live.
Soft ticks are gluttons, relatively speaking. Many move into mammal dens for a bedbug lifestyle. These ticks hide and, whenever they get hungry, just crawl over to the resident dinner.
For ticks without live-in prey, many “quest,” as the ambush is called. Ticks climb to some promising spot like the top of a grass blade, raise their front legs and just wait until something brushes by. But there are also ticks that hunt vigorously, even pursuing human prey.
A visit to Dennis Bente at the University of Texas Medical Branch in Galveston is unforgettable, in part because of a video of a Hyalomma tick chasing down one of Bente’s collaborators. The tiny brown creature scurries like a frantic ant in an almost-straight line over bare dirt, onto a boot and finally into a hand reaching down to grab it. This hunter doesn’t live in North America.
Ticks can spread a wide variety of diseases. Despite its name, Rocky Mountain spotted fever, which brings a higher risk of fatality than Lyme, is more common in the central United States and the South than in the Rockies. Other tickborne diseases are lately getting attention: A tick-bitten baby in Connecticut in April became the state’s first reported victim of the rare, but potentially fatal Powassan virus, thought to enter the bloodstream in just 15 minutes after a tick starts feeding. And medical journals are publishing discussions of whether a tick bite might lead to a sudden, deadly allergy to red meat. With a possible threat even to our beloved hamburger, new approaches to fending off ticks can’t come soon enough.
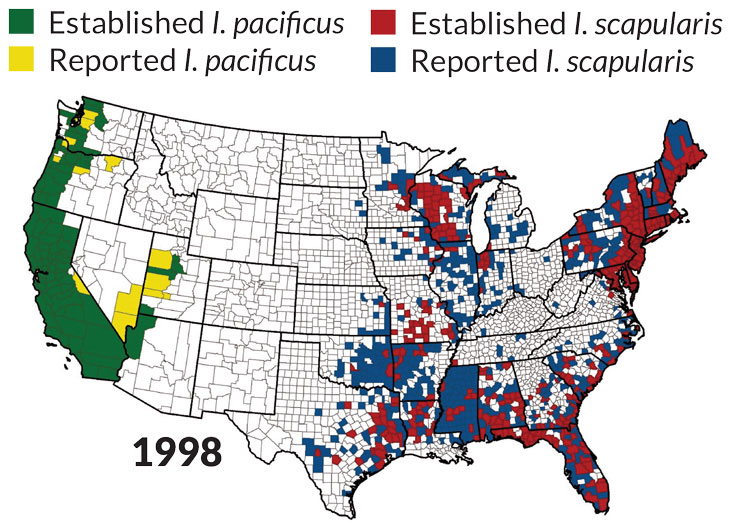
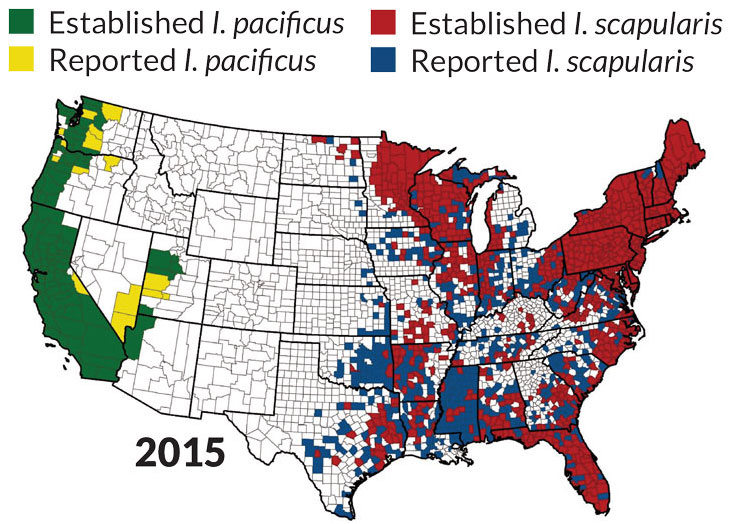
Source: R.J. Eisen, L. Eisen and C.B. Beard/J. Med. Entomol. 2016Left: Source: R.J. Eisen, L. Eisen and C.B. Beard/J. Med. Entomol. 2016Right: Source: R.J. Eisen, L. Eisen and C.B. Beard/J. Med. Entomol. 2016
Super reactors
The most dramatic way of rendering a disease irrelevant is a vaccine. One company raised hopes for this approach in April in Washington, D.C., at the World Vaccine Congress by announcing the start of human safety tests of a new Lyme disease formulation. The only Lyme vaccine for humans in the United States was withdrawn voluntarily in 2002 when controversy stalled sales. (Dogs can still get a Lyme vaccination.)
The strategy for the new Lyme vaccine isn’t like the familiar flu or tetanus vaccines because the pathogens get killed outside the human body. The company, Valneva, based in Lyon, France, has redesigned a protein, OspA, used in previous Lyme vaccines. The vaccine trains the human immune system to fight OspA, found on the surface of B. burgdorferi. When a black-legged tick starts sucking human blood, human immune cells get slurped in too and kill the Lyme-causing pathogens before they leave the tick’s gut. “The idea of this vaccine … is vaccinating the tick,” says CEO Thomas Lingelbach.
Even if the new vaccine proves to be safe and effective, its first shot in a doctor’s office, in the most optimistic view, is five to 10 years away.
There may be a bigger-picture way to imagine vaccines, however, than targeting each disease with its own shot. Ecologist Richard Ostfeld of the Cary Institute is one of the people hoping for a vaccine that stops the tick itself, and thus all the diseases it may pass along. By the luck of the great lottery of genetics, Ostfeld has a hyperactive immune response to tick saliva. Think of it as a natural version of what a tick vaccine might achieve.
Despite “many, many dozens of tick bites” over his career monitoring Lyme disease risk, Ostfeld has not gotten sick. He often wakes in the middle of the night with a “burning sensation” somewhere on his body. “I … put on my glasses and, sure enough, there’s a little dark spot surrounded by what’s already turned kind of red.” Warned by his vigilant immune system, he pulls off the dark bit of tick, which is usually dead or dying.
Maybe it’s a thing among tick scientists. Sam Telford of Tufts University’s veterinary school in North Grafton, Mass., who also studies the ecology of Lyme disease, has a similar reaction. Bites, he says, “itch like crazy.” A vaccine that makes people itch doesn’t sound very marketable, but blood that somehow poisons ticks sounds good.
A vaccine to protect cattle against debilitating blood loss from bites already targets the tick itself. Newer ways of targeting ticks are being developed for livestock, and for humans, though protecting our species poses extra challenges.
Ticks east and west
Of the nine or so tick species that spread diseases in North America, the three highlighted below cause the most trouble. Maps show each tick’s U.S. habitat.



Fix the landscape
“It would be lovely if we could get a vaccine,” says biochemist Kevin Esvelt of MIT. “But there’s a certain elegance to tackling the heart of the problem, which is ecological.” In several papers posted online at bioRxiv.org in 2016 and this year, Esvelt and colleagues laid out an approach that could slash Lyme risks by causing genetic changes in one of the mammals that ticks feed on, changes that could spread across whole landscapes.
The view of Lyme as an ecological disease blames much of the rise in cases on the suburbanization and fragmentation of once-wild countryside in North America. The shifts have fueled population booms in mammals such as white-footed mice that easily become great scurrying reservoirs of Lyme parasites. Ticks gorge on the mouse blood and a high percentage ingest the parasites.
Deer pick up a lot of the Lyme-spreading black-legged ticks. Yet deer aren’t biologically friendly to B. burgdorferi. The mice make a much better parasite paradise than deer do and are more likely to transmit those parasites.
The common name, “deer tick,” was a fluke of early misidentification, Ostfeld says. The tick was easy to collect on deer. But taxonomists realized the hard tick is just a northern form of the long-known black-legged tick, which dines on many mammals. In fact, just how deer abundance affects tick abundance was the No. 1 outstanding uncertainty about Lyme listed in a 12-person consensus published in the June 5 Philosophical Transactions of the Royal Society B.

Fighting Lyme disease appeals to Esvelt, who, like his pediatrician wife, grew up in the low-tick landscapes of the West Coast where Lyme is rare. In Massachusetts now, he says, “to both of us, it’s just horrific that a) there are that many ticks out there, and b) that they give you horrific diseases.” He especially regrets that neither of his two kids, nor anyone else’s, can tromp around outdoors, like he used to, carefree.
Esvelt calls the work of his lab, which plans to engineer a Lyme-resistant mouse, “sculpting evolution.” He and colleagues aim to tackle big biological problems like Lyme spread by using the insights of evolutionary biology plus the powerful gene-editing tool known as CRISPR/Cas9 (SN: 9/3/16, p. 22). But Esvelt wants to use that power with a startling openness and extreme public oversight.
“Right now, people don’t trust scientists to ensure that technologies are well understood before throwing them out there,” he says. “We have to fix that somehow.”
Before he even started to create a Lyme-resistant mouse in the lab, he asked for public meetings on the two Massachusetts islands where he hopes to test mice: Martha’s Vineyard and Nantucket. He got the green light to begin from citizen steering committees on both islands. But they still have the power to shut down the tests at milestones in the project. If the citizens nix the idea, he will walk away.
Originally Esvelt planned to sculpt Lyme disease into insignificance by acting on the ticks directly, driving down their numbers or changing them to be less dangerous. “But I talked to a lot of tick biologists who said, ‘Look, it’s not gonna happen.’ ” The black-legged ticks take so long to reproduce that the plan would only succeed “if you’re willing to wait about 50 years,” he says.
It’s actually faster to work with a mammal, the white-footed mouse. For the first tests, on islands, he plans great caution. He won’t even use a gene drive, the powerful way of deploying CRISPR/Cas9 so it overrides chancy natural inheritance and passes the desired genes to all offspring (SN: 12/12/15, p. 16). Instead he’ll just release mice genetically tweaked to be bad transmitters of Lyme and let natural mouse powers spread the genes.
Those mice won’t even be transgenic: They won’t carry genes from any other species. He’ll vaccinate island-captured mice in the lab, with an anti-Lyme vaccine or one that should confer an active immune response to tick bites. Then he’ll identify genes that produce the most protective reaction and put a large selection of them into what should be a safer animal that’s still “100 percent mouse,” he says.
While he’s tailoring safer mice for the island, however, he’s imagining new gene drives for a larger, mainland campaign. The way forward may require making gene drives less powerful, so they sputter out after a certain number of generations — “daisy chains,” he calls them, with loosely linked elements that fall apart easily.

Going for the gut
Ticks themselves probably have weaknesses that people haven’t yet exploited. The study of microbes in human guts has revolutionized ideas about human health and physiology. So Yale University’s Sukanya Narasimhan and Erol Fikrig are looking deep into the microbiome of the tick gut. Narasimhan describes the gut as a many-branched thing, “like a glove.” Ticks do have consistent bacterial residents, which could perhaps be exploited, but interactions look complex.
Along with Lyme, black-legged ticks can deliver other unpleasantries, such as human granulocytic anaplasmosis. When Anaplasma pathogens first tumble into a tick gut, invasion isn’t easy because some resident microbes form a biofilm along the gut lining that may be hard to breach. The pathogen, however, makes the tick secrete what’s essentially antifreeze, Fikrig, Narasimhan and colleagues reported in the Jan. 31 Proceedings of the National Academy of Sciences. The secretions can prevent biofilms from forming and ease the way for pathogen infection.
The sex lives of ticks could offer opportunities for completely different kinds of defenses, says longtime tick specialist Daniel Sonenshine of Old Dominion, author of Biology of Ticks.
He imagines, for instance, protecting livestock or dogs with decoys, “little bits of plastic” treated with a chemical cocktail that includes 2,6-dichlorophenol. That’s the come-hither substance female lone star and some other ticks release when they grab a mammal for a blood-feed. Like drinking venues for our species, mammals provide ticks with hot spots for finding mates. “These little plastic devices mimic a female tick,” Sonenshine says. And believe it or not, plastic fooled males long enough for a pesticide on the decoy to kill the ticks. (Tick sex on humans is possible but not likely, Gaff says. Humans rarely carry enough ticks at once to generate much of a scene.)
Robot vs. tick
Tick biology is also important in designing a robot army. The concept behind TickBot came out of a collision of two very different visions of pest-fighter robotics.
As Gaff tells the story, engineers at the Virginia Military Institute in Lexington, “were under this mistaken idea … that ticks live in trees and they fall on your head.” The engineers’ solution: Use lasers to shoot ticks out of trees.
When they called to enlist Sonenshine in the project, he had to break the bad news: no blasting into shrubbery; ticks are on the ground. His advice: Don’t build a robot to attack ticks at all. Get the ticks to attack the robot.
Instead of a laser-shooting, macho terminator, the concept morphed into a panting raccoon-sized machine that kills with dragged strips of pesticide-carrying cloth. The engineers’ four-wheeled buggy chugs slowly along following a looped guideline set up along a trail or in an open area.
As the bot works its way along, its motion, in some cases the passing shadow, will trick a tick into jumping at the cloth as it would the fur of a mouse or the sock of a hiker. But the big pull is carbon dioxide. Pest ticks “act like very lazy teenagers who don’t move unless they’re prodded,” says James Squire, one of the Military Institute engineer designers. Adding CO2 gets ticks’ attention and strengthens the illusion of a breathing, warm-blooded something.
The engineers — Squire, David Livingston and Gerald Sullivan — “came up with this amazing system of tubing and carbon dioxide canisters” for releasing CO2 from the bot, Gaff says. However, a student last year put a bit of dry ice in a cup with holes to mimic mammal breath. It worked and made the bot far easier to lug around.
Gaff remembers when she was first pulled in to do the field testing. “I was a huge skeptic coming in,” she says. To test TickBot outdoors, she chose as the first site a path through a wooded park with what she calls “infinite ticks.” There weren’t many Lyme ticks there to test, but the creeping cart tricked so many of the abundant lone star ticks that — high praise from a tick scientist in summer — she sat on the ground and had lunch.
Within a day, however, more ticks moved onto the cleared path from the nearby woods, she and colleagues reported in 2015 in Ticks and Tick-borne Diseases. Still, the notion of robotic tick catchers may be catching on. Gregory Gray, who teaches at Duke University’s Global Health Institute, worked with students from the local robotics club to design their own creeping, cloth-dragging cart.
And the TickBot team is now planning a bigger and faster robot that might ease the uncomfortable business of monitoring for cattle ticks on grazing land. Usually, people “dress up in woolly pajamas” as Squire puts it, and move through brush with an eye out for rattlesnakes in the Texas summer heat. The ticks grab the suit and are later counted. There’s got to be a better way.
For the TickBot itself, summer 2017 brought testing on grass around a playground. Like a little Roomba vacuum cleaner (but with guide wires), it set out twice a week to carve a safe zone by whisking away ticks, Gaff says.
“I’m giving them their space, and I’m asking them to respect my space,” she says. It’s all part of the mind-set of surrendering to the notion that there will always be ticks. But someday maybe we won’t care as much.