How trans fats oozed into our diet and out again

The advent of mass-produced trans fat helped make the doughnuts, cookies and crackers we love inexpensive and shelf-stable. But now companies have to get along without them, while we still want to have our cake and eat it, too.
JMiu/Flickr (CC BY-NC-SA 2.0)
On June 16 the Food and Drug Administration made the final call: Trans fats are no longer “generally recognized as safe” for use in food. That means that food manufacturers have three years to ooze these cheap and useful fats out of their processed foods.
In fact, most of them already have. Trans fat —a big source of which is partially hydrogenated vegetable oils — has been the food villain of choice since 2006, when the FDA required companies to include trans fat content on food labels. Since then, the oily fats that used to lurk in everything from crackers to frosting have largely vanished — with a few exceptions, such as ice cream sprinkles and some doughnuts.
As trans fats went in and then out of style, some people blamed food companies for embracing the fats, which offered a cheaper alternative to animal products. But what trans fats brought to the world of microwave popcorn and Oreos was a bit more complicated than cost savings. And their fall from grace shows that nutrition — like any other scientific area — is subject to self-correction over time.
I can’t believe it’s not…
Before trans fats came around, the delicious circles of pie crusts and doughnuts revolved around butter, lard (pork fat) and tallow (beef fat). These fats have two features in common: They are solid at room temperature, and they don’t go rancid in baked goods as fast as liquid oils. They are also all saturated fats, chains of single-bonded carbon atoms with hydrogen atoms neatly lined up on either side of each carbon. They are called saturated because hydrogens fill all the bonds in the carbons, soaking the molecule in hydrogen.
Unsaturated fats, in contrast, have fewer hydrogen atoms, leaving carbons free to double bond to each other. These double bonds mean that the fat molecules don’t line up, making the fat (usually) an oil at room temperature. The bonds in saturated fats are also more stable, making them less likely to go rogue and bond to oxygen and end up rancid (store an open jar of natural peanut butter at the back of the shelf for a few months, sniff it, and you’ll see what I mean).
But saturated fats are all animal fats, making them more expensive than oils from soybeans or corn. And so scientists began to wonder how they might alter vegetable oils to get some of the benefits of saturated fats, without the animal by-product.
The French chemist Paul Sabatier first discovered how to add hydrogens to fat chains around the turn of the 20th century, using nickel as a catalyst. He was followed closely by the German chemist Wilhelm Normann, who in 1901 developed a hydrogenation process using hydrogen gas.
The process eliminates some of the double carbon bonds and saturates the fat molecules with hydrogen, says Sarah Ash, a nutritionist at North Carolina State University in Raleigh. “But it’s kind of like musical chairs.” The hydrogens attached to the carbons are all on their “chairs.” When the music starts and hydrogenation begins, she says, “hydrogen atoms get up and run around.” As they do, some of the double bonds between carbons break, leaving free slots for hydrogens to come and saturate the bond. But when the music stops, the hydrogens go back to bind again to the carbons, and they don’t always come down in the same positions on the remaining double bonds. Instead of ending up on the same side of two carbons in a double bond — the cis side — a few end up on opposite sides — the trans side. Hello, trans fats.
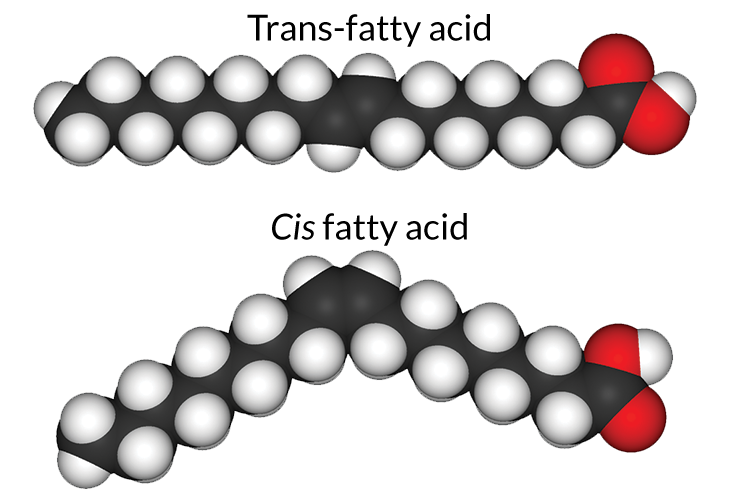
When hydrogen atoms are in a cis position, they cause a kink in the chain of carbons. But when the hydrogen atoms are in the trans position, the whole fat molecule ends up a little straighter than a fat molecule with all cis bonds. This straighter position allows the fats to behave more like straight saturated fat chains — that is, solid at room temperature and less prone to going rancid. “It was an opportunity from the food industry perspective to do what animal fats could do — make the oils harder and get rid of the double bonds,” Ash says, using vegetable oils instead of animal products. (Dairy fat also has small amounts of trans-fatty acids, but they are not necessarily the same chemicals as those in partially hydrogenated vegetable oils. When people say “trans fat,” they are almost always talking about the ones derived from vegetable oil.)
It helped that the trans-fatty acids made from vegetable oils never had the aftertaste of lard. “They were nice, bland white fats,” says Tom Brenna, a food scientist at Cornell University. “If you use lard or butter to make a frosting, it will taste like lard or butter, but partially hydrogenated vegetable oil doesn’t taste like anything. It just gives you mouthfeel.” There seemed to be no real downside. Trans fats were cheap, easy and — as far as anyone knew — healthier than animal fats. Fudgy brownies and Apple Jacks could have a pleasant texture and sit on the shelf for longer, all for far less than it would cost to use butter or tallow. And so in the 1980s, companies began to hydrogenate cottonseed oil, soybean oil and other oils on a large scale, and food reformulation began.
A short-lived relationship
Trans fats gained popularity as public health officials worried about the association between high levels of saturated fat and cardiovascular disease. Companies began to look for ways to get saturated fat content off of their nutrition labels, and unsaturated trans fats fit the bill. “Your [nutrition] label would look OK because they weren’t saturated fats and they didn’t have to be labeled,” Ash explains.
But as soon as trans fat began to take over the market, more research began to show that, instead of being better than saturated fat, trans fats might actually be worse. “Trans-fatty acids increase LDL cholesterol [our “bad” cholesterol], and they don’t have much of an effect on HDL [our “good” cholesterol],” contributing to cardiovascular disease risk, says Alice Lichtenstein, a nutritional biochemist at Tufts University in Boston, who published a review on the topic in Current Atherosclerosis Reports in June 2014.
The data, she says, are “very consistent” that increased trans-fatty acid consumption increases the risk of cardiovascular disease. But as yet, scientists remain unsure as to how exactly this happens. Lichtenstein notes that trans-fatty acids slow down the breakdown of LDL particles. They also decrease large LDL particles and increase the presence of small, dense LDL particles — which are associated with higher risk of cardiovascular disease.
As the 1990s progressed and the evidence mounted that trans fats have adverse effects on health, the FDA began to take notice. In 2003 the agency issued a statement that trans fats would get a separate line on the nutritional label by 2006. “I think the change the FDA made requiring [trans-fatty acids] to be listed had a big effect,” Lichtenstein says. “I don’t think customers use the information, but I think the companies realized that it looked better for their products to be listed as zero trans fat.” And so began the re-reformulation.
“Now, we’re back to more naturally saturated products like palm oil,” Ash says. “Many folks are back to butter and lard.” Coconut oil is also back on the scene, its high saturated fatty acid content now a feature instead of a bug.
Back to the future, and back to butter
At first, public health officials worried that all fat was bad. Then they realized saturated fat might be bad, but unsaturated fat is good. Now we know that not all unsaturated fats are created equal, and trans fats have their own problems. With each dietary recommendation and processed food reformulation, it’s easy to think that maybe nutrition, as a field, doesn’t know what it’s doing. “I think in general there’s been a squandering of our credibility,” Brenna says. “Having to reformulate from one thing to another thing, people are making louder and louder noises wondering what the story is. Is there really science going on?”
It doesn’t help that reformulations do take time. Food products have reputations to keep up, after all. “The public wants the doughnut the way it’s always tasted, just without the trans fats,” says Ash. “We want to have our cake and eat it, too.”
Not only does the public demand great taste that’s less filling, Ash explains, “there’s [also] this pressure to make a dietary recommendation.” People just want to know if they can eat it and how much they should eat. New knowledge that makes eggs a villain one day and a hero the next leaves people feeling in the lurch. People are more tolerant of sweeping changes in scientific knowledge when it’s not a part of their plates.
But nutrition is a science, just like every other. New findings will frequently change and overturn old ones. “The challenge in nutrition — as in all areas of scientific inquiry — is there’s a constant progression of knowledge,” Ash explains. People are complex, and so is the food we eat. A few years from now, we might be facing a new — as yet unknown — dietary villain. And then it might be time for a re-re-reformulation.
If you’re truly worried about what to eat, the oldest advice might still be the best. Everything in moderation. “If you eat all things in moderation and variety … you likely won’t need to worry about the next ‘new finding,’” Ash says. It’s not so hard to cut something from your diet or eat less of it if you never ate that much of it in the first place.
So maybe I will take a doughnut once in a while, as long as it’s trans fat–free.







