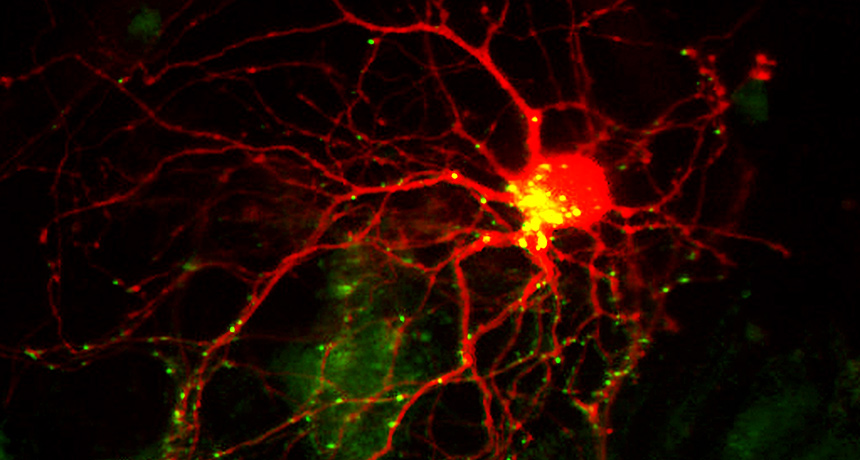
To keep things tidy, nerve cells (red) nibble away at themselves via Pac-Man–like structures (green and yellow).
Andrey Tsvetkov, Steven Finkbeiner, UCSF
There’s a little Hannibal Lecter in all of us.
But while the famous cannibal dined on chunks of his enemies and friends, most people stick to gnawing on themselves at a microscopic level. In fact, the cells of organisms from yeast to humans regularly engage in self-cannibalism. Cells chew on bits of their cytoplasm — the jellylike substance that fills their bellies — and dine on their own internal organs, although usually without the fava beans and Chianti.
It may sound macabre, but gorging on one’s own innards, a process called autophagy, is a means of self-preservation, cleansing and stress management.
“It has become evident that it is really an essential or vital function,” says Fulvio Reggiori, a cell biologist at the University Medical Center Utrecht in the Netherlands.
A munch here gets rid of garbage that might otherwise clog the system. A nibble there rids cells of malfunctioning parts. One chomp disposes of invading microbes. In lean times, all that stands between a cell and starvation may be the ability to bite off and recycle bits of itself. And in the last decade or so it has become clear that self-eating can also make the difference between health and disease.
“Too much or too little autophagy is a problem,” says Daniel Klionsky, a cell biologist at the University of Michigan in Ann Arbor.
A cell that bites off more than it can chew can kill itself, Klionsky says. A few rare genetic diseases are linked to an excess of unsuccessful autophagy: The muscles of people with Danon disease, Pompe disease and X-linked myopathy can become weak after filling up with Pac-Man–like structures that put the bite on the cell’s insides but can’t finish digesting.
Not enough autophagy can also lead to disease. Most notably, a cell that can’t clean itself up can turn cancerous.
Article continues after graphic
Hungry, hungry cell

A lack of nutrients, among other factors, can trigger cells to dine on themselves. The process begins with inhibition of a key protein. Then a curved double-membraned structure begins to form. It grows into what’s called an autophagosome and envelops cell innards in need of digestion. After an autophagosome fuses with a bag of enzymes called a lysosome, the contents get broken down and expelled for future use. Illustration: Nicolle Rager Fuller
With their sights on fighting disease, scientists are now uncovering the mechanics that keep autophagy in balance. Recent work, for example, shows that faulty versions of a gene that helps spark autophagy can make people more susceptible to tuberculosis. While boosting autophagy can help prevent cancer, researchers have found that once cancerous, cells can hijack the self-eating mechanism, a twist that suggests reining in autophagy may keep tumors at bay.
And for a number of neurodegenerative diseases — including Parkinson’s and Alzheimer’s — new studies show that cells can’t quite get their toxic innards down. In some cases, treating these diseases may be a matter of encouraging autophagy, while others may require a tightening of the self-cannibalizing belt.
Cellular dinnertime
Scientists have known since the early 1960s that autophagy happens. But until a decade ago, when yeast geneticists found genes that control the process, no one could do more than look through a microscope and describe how cells eat themselves. In the first recognizable step of the process, a small, curved, double-membraned structure appears around the parts of the cell that are on the menu. That little bit of membrane grows to encase whatever the cell plans to swallow. “Imagine a teacup expanding and growing and eventually becoming a football” (a soccer ball for Americans), says Sharon Tooze, a cell biologist at Cancer Research UK’s London Research Institute.
That soccer ball structure is the Pac-Man, known technically as an autophagosome. It fuses with another intracellular structure called a lysosome, basically a big bag of enzymes that break down proteins and other molecules into their basic building blocks, much the way stomach acid digests food. The building blocks can then be recycled to build more proteins, membranes or other things the cell needs.
Article continues after graphic
Feast or famine
Finding the right balance of autophagy may be crucial for preventing and treating a range of diseases. In some cases, more autophagy is better; in others, less is more.
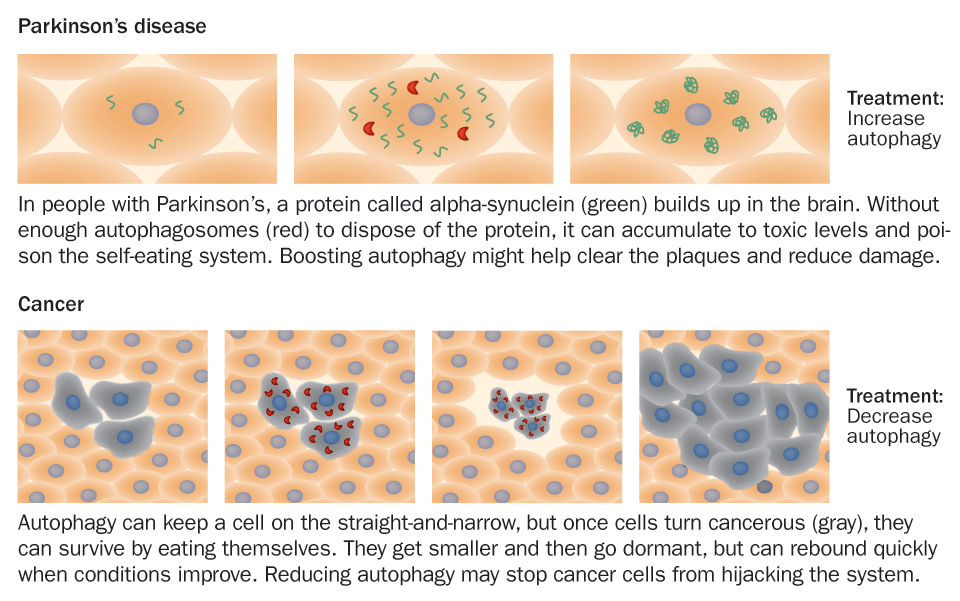
Illustration: T. Dubé
Since biologists working with yeast discovered the first genes involved in autophagy in the 1990s, more than 20 different genes have been found to play a role in the process in yeast — and many of those genes have counterparts in humans and other animals. But autophagy gets more complex in these other organisms: Several of the genes found in yeast, for example, have multiple copies in human cells. One species of yeast is known to have one ATG8 gene, while humans have six versions of the gene.
Each of those genes encodes proteins that interact with other proteins, further complicating things. Researchers at Harvard Medical School reported last year in Nature that it takes 409 different proteins interacting in 751 different ways to carry out autophagy in human cells. With so many moving parts, perhaps it’s no surprise that things sometimes go awry.
Unfortunately, interfering with self-eating can do more than just give a cell indigestion. Messed-up autophagy can ruin a cell’s whole life, not to mention the health and life of the organism in which the cell lives. That’s because autophagy is one of a cell’s best methods of coping with stress.
Though a low level of autophagy is maintained constantly, during times of stress the body ramps it up. This boost helps save cells from starvation. “It’s like carrying around your own refrigerator or a cooler with your sandwich,” says Vojo Deretic, a cell biologist at the University of New Mexico Health Sciences Center in Albuquerque.
Too much trash, such as broken cell batteries, misfolded or damaged proteins and inoperative organelles can also cause stress, so self-eating serves as an important cellular garbage disposal.
But perhaps the most important stress-reducing function of autophagy is defending against pathogens, Deretic says. He and his colleagues have found that without autophagy to digest invading microbes, a cell is headed for disaster.
Eating invaders
Autophagy probably arose as a pathogen-defense mechanism very early in the evolution of cells that sequester DNA in a compartment called the nucleus. These “eukaryotic” cells differ from bacteria and archaea, the other two major groups of life, which don’t have nuclei.
When the first cells were swimming around the primordial soup, bacteria and archaea ruled. “The eukaryotic cell was an intruder in the mix, feeding on bacteria,” Deretic says.
But bacteria can be a dangerous food. Once gobbled up by the eukaryotic cell, the microbes could start eating the cell from the inside out, giving the eukaryotes the microscopic equivalent of food poisoning. Autophagy was a eukaryote’s recourse, allowing the cell to recognize invading parasites, bacteria and viruses, then engulf and destroy them. “The cell learned how to defend itself,” Deretic says.
Reggiori agrees that autophagy may have been the first immune system, and he and other scientists are interested in studying pathogens that have learned to evade the autophagy system, in hope that the loopholes may point to ways to cure disease.
One microbe that has found such a loophole is the bacterium that causes tuberculosis, which can sometimes hide out in cells without getting digested. Some people are more prone to tuberculosis infections than others, but the reason why hasn’t been clear. A version of a gene called IRGM may be to blame, Deretic and colleagues reported in the December Nature Cell Biology.
His team found that the IRGM protein plays a role in a series of cellular events that trigger autophagy. When tuberculosis or other bacteria invade a cell, the protein helps autophagy go to work to eat the intruders. But in people with a defective version of the gene, cells can’t work up enough of an appetite to keep tuberculosis bacteria in check.
Another variant of the same gene may be linked to a seemingly unrelated dis-order, the inflammatory bowel condition known as Crohn’s disease. This IRGM version can’t be properly regulated by small RNA molecules known as micro-RNAs, which help control how much of the protein gets made.
With this version of IRGM, there is an overabundance of the protein and an increase in formation of autophagosomes. To figure out the consequences of this overproduction, researchers infected intestinal cells with a pathogenic form of E. coli, one possible trigger that could cause a flare-up of Crohn’s disease. Autophagosomes swallowed the bacteria but seemed to have a problem breaking down the invading microbes, Patrick Brest of the French National Institute for Health and Medical Research at the University of Nice Sophia Antipolis and collaborators reported online January 30 in Nature Genetics. That improper digestion meant more of the bad bacteria stuck around, possibly triggering inflammation and disease.
Hijacked digestion
While microbial invasion is one way to spur a cell to snack on itself, there are other dinner bells. One thing that gives a cell a taste for its own flesh is a reduction in outside nutrients getting to the cell.
Starvation inhibits an important biological signaling system, known as the mTOR pathway — named for a key protein involved in regulating cell growth and survival, cell movement and protein production. The inhibition of mTOR sets off a cascade of reactions inside the cell that end in autophagy and may be crucial to prolonging cell life and ultimately fending off cancer.
A drug that inhibits mTOR, called rapamycin, has been shown to extend life span in mice. It and calorie restriction are the only methods proven to prolong longevity, suggesting both may work through autophagy to make cells live longer.
Scientists think self-cannibalism may keep cells healthy by clearing away junk that might otherwise poison or damage the cell and its DNA. Or as Eileen White, a cancer biologist at the Cancer Institute of New Jersey and Rutgers University in New Brunswick, puts it: “The cleaning crew is out full blast mopping up all the garbage and keeping cells pristine.”
When the cleaning crew goes on strike or otherwise falls down on the job, a buildup of toxic cellular garbage can either kill cells or damage them in ways that lead to cancer.
But once a person has cancer, starvation and the resulting autophagy can become part of the problem. Cancer cells at the center of a tumor can be constantly hungry if they are cut off from nutrients in the blood. Those starving cancer cells inside tumors use autophagy as a survival mechanism, White says. The cells become very tough to kill when they dine on themselves, and they can start growing like crazy when more food becomes available, such as when tumors grow blood vessels or when cancer cells escape from the main tumor.
To get some clues about how tough a hungry cancer call can be, White and her colleagues made cells unable to commit suicide, a hallmark of cancer cells. Then the team starved the cells.
Even cells that can’t kill themselves would die eventually if they didn’t get enough nutrients, the researchers thought. But in the lab, these cancerlike cells seemed to do just fine without food. “When we looked at them closely, we realized they were eating themselves,” White says.
As these starving cells munched away on themselves, becoming smaller and smaller, they eventually went dormant, existing but not doing much else. That dormant state can last a week or a month, depending on the type of cell, White says. But when the cells were no longer under so much stress, they rebounded and began growing again.
Cancer cells in the body may use a similar strategy to hide and ride out tough times, then cause a relapse later. “These are what we call the oncology horror movies,” White says.
Although scenes of hungry zombie tumors rising from the dead are terrible, scientists aren’t throwing their hands over their faces and cowering. Instead, researchers are experimenting with therapies that may ruin cancer cells’ self-eating picnics. Clinical trials are currently under way to determine if drugs that block autophagy, such as hydroxychloroquine, can make cancer cells more vulnerable to chemotherapy.
Balance for the brain
Controlling autophagy may also be the key to quashing neurodegenerative diseases. Though researchers have known that autophagy is messed up in several neuron-killing diseases, only recently have the details come to light.
“In many of these diseases we’re getting some quite good clues about what is going wrong when,” says David Rubinsztein of the Cambridge Institute for Medical Research and Cambridge University in England.
For instance, in Parkinson’s disease globs of a protein called alpha-synuclein build up in the cell and stop autophagosomes from swallowing, Rubinsztein and his colleagues reported last year in the Journal of Cell Biology. With the cell’s chomping abilities lost, the toxic protein continues to build up, eventually leading to the death of the cell. The same is true of Huntington’s disease: Autophagy can’t do its part because of toxic levels of the huntingtin protein, Rubinsztein and others have found.
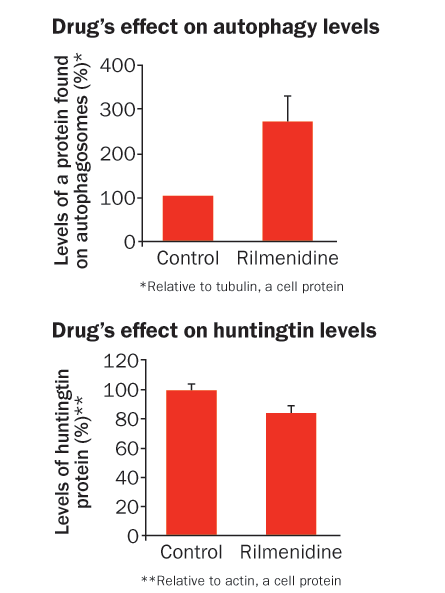
Boosting the amount of autophagy could overcome these defects and possibly prevent or slow the disease. In fact, researchers are beginning clinical trials to see if autophagy boosters, such as a drug for high blood pressure called rilmenidine, can head off Huntington’s disease. Last year Rubinsztein and his colleagues reported in Human Molecular Genetics that rilmenidine successfully treated Huntington’s in mice. If clinical trials in people also work, the drugs may be tested on Parkinson’s disease patients too.
Alzheimer’s patients might need to do just the opposite — inhibit autophagy. As people age, autophagy in their brain cells slows down, Marta Lipinski of Harvard Medical School and colleagues showed in a study published last year in the Proceedings of the National Academy of Sciences. But people with Alzheimer’s disease had much more autophagy activity in their brain cells, the team found. The increased activity could be the cells’ attempt to dispose of a cell-killing protein that has been linked to the disease, known as amyloid-beta (SN: 8/16/08, p. 20; SN: 3/12/11, p. 24).
Although the A-beta protein stimulates cells to make autophagosomes, it also prevents the autophagosomes from properly fusing with lysosomes, the cellular equivalent of choking on food, meaning it never reaches the stomach to get digested. Having too many auto-phagosomes sitting around may interfere with other important cellular functions.
“We call this the clogged toilet model,” Lipinski says. “Flushing a toilet is generally a good thing … but if the toilet is clogged, flushing is not a good idea anymore.”
In her team’s study, some Alzheimer’s drugs decreased autophagy in the cells. This may have reduced stress by preventing indigestible autophagosomes from building up, Lipinski speculates. The findings are still preliminary, she says, and much more work is needed before scientists can say exactly how A-beta leads to cellular indigestion and whether reducing levels of self-cannibalism might be helpful.
A full understanding of why some cells become overly cannibalistic — and why some don’t eat enough of themselves — still requires more research into exactly how autophagy works. Unanswered questions still linger, such as how cells know when it’s mealtime and where the membranes that form the autophagosomes come from. Scientists also aren’t sure how cells decide when they’ve eaten enough and the details behind laying down the self-eating fork.
But progress is being made, and soon enough, scientists may have a more complete profile of the inner Hannibal Lecter — and a better idea of when the cannibal should be unleashed.