special report
Proyecto Agua/Flickr (CC BY-NC-SA 2.0)
The scene was stranger than it looked, even by Las Vegas standards: Two young men pull up in a U-Haul truck to a motel outside the city. They check in and move a cooler into their room. They appear to be handling something of importance, and look to see if the ice needs replenishing. Inside the cooler is not the makings of epic hangovers but instead an experiment in eternal youth.
Tucked within, protected from the desert heat, are more than a hundred tiny pond invertebrates. One of the men, Daniel Martínez, with a Ph.D. in ecology and evolution a month or so old, is rearing these little organisms to test a claim that they somehow stay young all their lives, no more likely to die as years go by as they are early on. They can die, however, from high temperatures or starvation. Leaving the animals on their own for more than a day invites disaster, so if Martínez travels, even stopping for sightseeing with his brother in Las Vegas, all the animals in the aging experiment travel, too.
Their road trip was in 1993, when the “dogma,” as Martínez recalls, was that evolution would not allow any multicelled organism to escape aging. Just as humans age, the thinking was, other organisms also decline in health as time goes by, with death becoming more and more likely. Yet few people at the time were bothering to document aging in any creatures other than a few standard lab residents.
Biologists have long tracked aging in fruit flies and lab mice (SN: 7/23/16, p. 16), but a bloom of recent data from more diverse organisms is stirring up discussion about how aging could have evolved — and if it’s inevitable. The ongoing studies of Martínez’s pampered pond invertebrates and a massive effort to study aging in a roadside weed are good examples of these provocative approaches. They’re shaking up basic assumptions of a long-standing theory and inspiring new thinking to explain why there’s so much crazy variety in how life deteriorates — or maybe doesn’t.
Old ideas
Deciding whether an organism is aging can get tricky. For humans, the slowing and graying, the wrinkling and creaking are all too obvious. But what about plants? Or fungi? For a metric that applies across many species, evolutionary biologists often focus on how the number of deaths in a population changes over a particular period of time. If this death rate increases as time passes, the organism ages. (In this scheme, life span is irrelevant. A hypothetical species that lives for just a few months but keeps its death rate flat until the end would still be considered “biologically immortal.”)
Early evolutionary thinkers proposed that aging followed by death is a good thing, another marvel of the mindless force of natural selection. Built into individuals, this inevitable decline kept feeble parents from sapping resources from the young.
But the idea that aging evolved as a boon for the next generation “is really nonsense,” says Axel Kowald of Newcastle University in England, a biochemist who specializes in the bioinformatics of aging. Among the many objections: It’s hard to see why a lucky few that could live a bit longer and continue to reproduce wouldn’t overtake a population. With more offspring, they’d spread more of their genes. Over time, then, genes for aging should be few, fewer, gone.
One of the modern mainstream explanations of aging rests on the idea that evolutionary forces lose their power to edit as adulthood stretches on. As genes are copied generation after generation, mutations are made. Natural selection can remove from a population the typos that harm the young; disadvantaged carriers don’t pass those mistakes down to the next generation in much abundance.
Mistakes that cause trouble late in life, however, can be almost impossible to purge, argued the late zoologist Sir Peter Medawar, a Nobel laureate who titled his autobiography Memoir of a Thinking Radish. In a 1951 lecture, he explained this approach to aging by whimsically tracing the perilous lives of laboratory test tubes. The mortality rate of these hypothetical test tubes, which for the sake of explanation reproduced more than once in their lives, allowed few tubes to reach old age. Test tubes that don’t reach old age don’t reveal detrimental effects from mutations that act only late in life. Therefore natural selection didn’t have a chance to stop those mutations from being passed down to test tube babies. In a scenario now called mutation accumulation, the late-acting mutations could thus build up and cause the declines of aging, also known as senescence. Natural selection doesn’t weed out these mutations because, Medawar said, wild organisms “simply do not live that long.”
In a perverse twist on this idea, natural selection might not just allow genes that bring late-life decrepitude to accumulate but also might favor those genes. Evolutionary biologist George C. Williams, later eulogized as a quiet and deep thinker with the look of Abraham Lincoln, argued in 1957 that genes with split personalities, like Jekyll and Hyde, could help explain aging. The benefits of these genes appear early in life and the gene is thus passed to the next generation, with its downside revealed as frailty only late in life.
Story continues below graphs
Till death do us part
In the 1990s, as the theories were then understood, a widespread idea was that “nothing can escape aging,” Martínez says. Yet as a graduate student at Stony Brook University in New York, he read about some tiny hydra species that had extraordinary powers. These branching bodies can reproduce by budding off clone babies, and they can rebuild themselves after dismemberment. What’s more, they didn’t appear to deteriorate with passing time. Biological immortality was a grand claim for these distant jellyfish relatives, soft translucent stalks a few millimeters tall with a tuft of tentacles wiggling from the top. But no one had done a rigorous test collecting the hydra and tracking their death rates.
Martínez eventually set up 145 Hydra vulgaris in laboratory luxury, where no predator could reach them and they could enjoy catered food all their lives. “When I started doing the experiment, I thought that I was going to prove that hydra could not escape aging,” he says. “A year and a half later I got my Ph.D. — the hydra were still with me.” The expected rise in death rate that characterizes aging organisms still hadn’t started. “I got a postdoc at University of California, Irvine,” he says, “so I crossed the country in a U-Haul truck with the hydra and all my furniture.”
The truck was supposed to be air-conditioned but wasn’t, and with a hot engine right under the cab, driving a southern route pulling a trailer, Martínez had to keep careful track of ice for the hydra cooler. Plus, there was all the changing of water, the feeding, the raising brine shrimp so the hydra had live prey. This was when the whole party visited Las Vegas.
The hydra made it. (Martínez, however, no longer even considers a hydra project without a technician to manage their care.) In 1998 he published results of four years of hydra watching. His title was cautious: “Mortality patterns suggest lack of senescence in hydra.”
“I published the paper and forgot about it,” says Martínez, now at Pomona College in Claremont, Calif.
Opinions about the inevitability of aging continued to run strong — as demographer James Vaupel of the Max Planck Institute for Demographic Research in Rostock, Germany, discovered in 2002. At a workshop on aging in nonhuman species, Vaupel stood up to say that the paper he had found most interesting was one describing mortality rates in a roadside weed. The rates appeared to drop as time passed, leading Vaupel to propose it as a possible case of what he called “negative senescence.”
“My remark was met with ululations of horror, cries of derision, hisses and boos,” Vaupel says. Eminent biogerontologists said that theorist William Hamilton had proved decades earlier that mortality, at least in repeatedly reproductive adults, universally rises with age and “there was no need for the audience to listen to a demographer who didn’t understand biology.”
Further data on the weed showed a more complex story, but the meeting outcry had a meaningful effect. As soon as Vaupel got back to his Rostock lab, he asked Annette Baudisch, then a new Ph.D. student, to “figure out why Hamilton’s proof was wrong.”
Baudisch published her critique of Hamilton in the Proceedings of the National Academy of Sciences in 2005. Hamilton’s proof could not explain the full diversity of aging, she argued. Some species that keep growing throughout adulthood, tortoises and many plants, for example, might not be included.
Though some researchers believe there’s still much truth to Hamilton’s approach, Vaupel took this conclusion as a cue to go questing for examples of prolonged youth. He talked Martínez into redoing the hydra experiment — but bigger. Instead of four years, the test ran for eight. Instead of 145 animals, the team had multiyear data from 2,256.
The resulting paper came out last year in the Proceedings of the National Academy of Sciences. Two species of hydra, with their many representatives divided between Claremont and Rostock for raising, had continued their usual low-drama lives, feeding and budding off babies but not showing any upsweep in their mortality rates. In 10 of the 12 groups, the annual probability of death stayed around 0.6 percent, and two groups held steady with an even lower annual rate of 0.09 percent.
Continuing the tests until the whole study population died, which would be ideal for tracking the hydra’s entire life history, would take more than 1,000 years, researchers calculate. But eight years of data gave Martínez and Vaupel confidence. The old view that aging is inevitable, the paper declared, “is no longer tenable.”
The hydra results so far are “solid evidence” that not all species age, Kowald says. And there are other, less-studied candidates for what’s called negligible senescence, too: three-toed box turtles and bristlecone pines, for instance.
Into the wild
The lab, of course, isn’t where evolution shapes life. Biologists seeking to understand how aging evolved need to know if and how organisms age in the wild, research that is likewise challenging Medawar’s pronouncements.
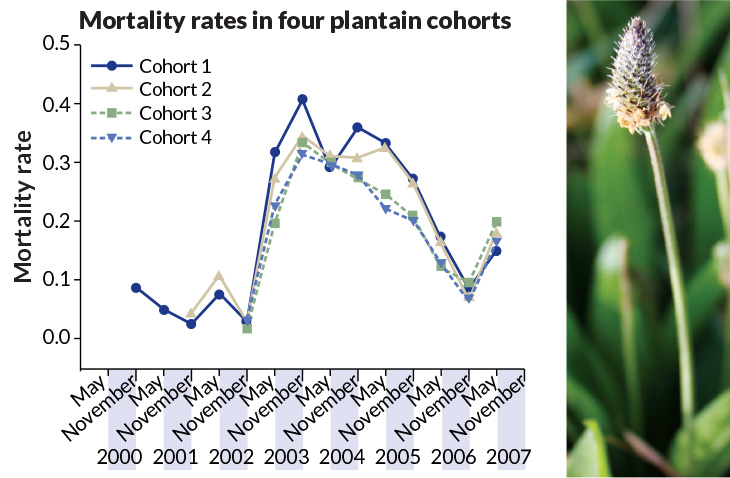
The plant study that caused a ruckus for Vaupel was an early version of a test by Deborah Roach, a plant evolutionary biologist now at the University of Virginia in Charlottesville. Her results from 4,476 ribwort plantains (Plantago lanceolata), set out at a long-term research site in Durham, N.C., had suggested that this common roadside weed was escaping the supposedly inevitable decline of aging. But construction of a new art museum wiped out those plots after less than five years.
After moving to Virginia and summoning the resolve to start the experiment again, Roach selected meadows at Thomas Jefferson’s birthplace, close to Charlottesville and under the protection of local historical preservationists. During the years 2000 through 2002, an army of undergraduates set out 30,000 plantains, all of known genetic heritage and marked for individual monitoring.
After collecting seven years of data on when plants died, Roach picked up a subtle signal she hadn’t observed in Durham. At first, plantains of different ages had about the same mortality rates, all relatively low, with six-month mortality rates at less than 10 percent. But during the three years that followed, the plantains clearly struggled. Roach suspects soggy winters plus competition from neighboring foliage were to blame. Death rates rose to around 30 percent and — this was the important bit — the death rates climbed higher for the older plants. A population that had looked as if it weren’t physically declining with age showed signs of senescence when the going got tough.
The results contradict Medawar’s fundamental assumption that life is so short and brutal in the wild that the possibility of seeing frail, aged organisms there would be exceedingly rare. “Now we have a great body of literature showing that in fact there are these old animals out there, these old plants,” Roach says.
Story continues below graphic
A 2013 tally by Dan Nussey of the University of Edinburgh and an international array of colleagues documented more examples of aged organisms in the wild — 175 species, in fact, including Dall sheep, antler flies and great tits, among others. A study of painted turtles published June 7 in the Proceedings of the National Academy of Sciences added that species to the list, showing that human impacts might inadvertently be nudging an Illinois population toward senescence.
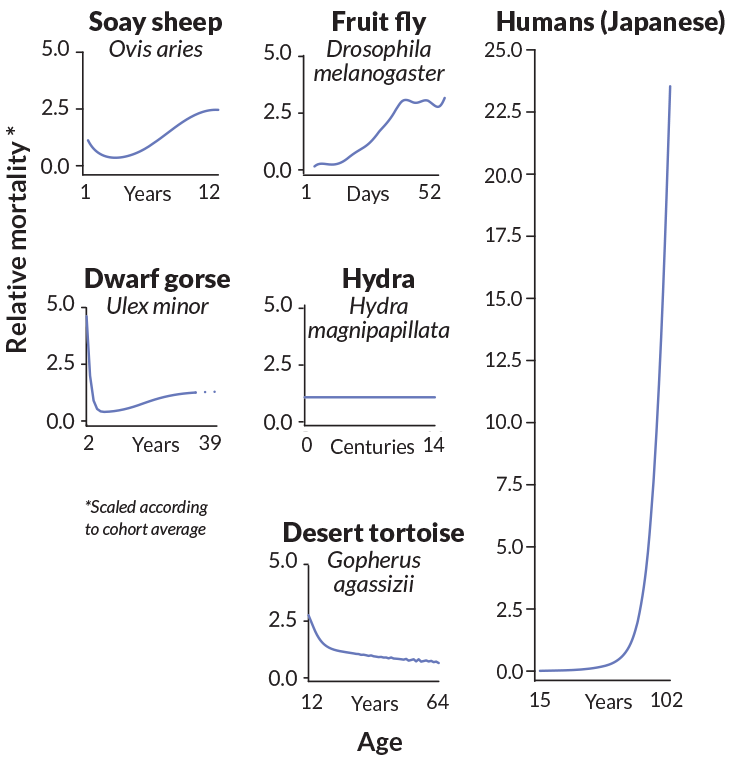
Across the tree of life, aging now looks more varied than old ideas predicted. Drawing on data for 46 species, Vaupel, Baudisch (now at the University of Southern Denmark in Odense) and 12 coauthors published a paper in 2014 featuring a full page of mortality curves, which track how the number of deaths in a given group changes over time. Many of the organisms show the expected upsweep with time, but other curves are idiosyncratic. The curve of Soay sheep curls concavely downward during early adulthood before rising again to a rounded hilltop in old age. Alpine swifts’ curve looks like a side view of a lawn chair relaxed way back for a summer snooze.
With mortality data on so few of the species on Earth, it’s too early to pronounce big trends. So far, body size and life span don’t appear to dictate the shape of the curve: The curves of water fleas and lions look remarkably similar. Organisms from different kingdoms can also have similar curves: The curves for desert tortoises and netleaf oaks both tilt downward.
“We’re going to have to figure out what it is about the biology of these species that explains the variety,” Roach says. The new reports, she adds, “are putting a big, bold spotlight on the theories and saying, ‘Hey guys, we need to update.’”
Think again
Last year in Experimental Gerontology, Kowald and Thomas Kirkwood of the Institute for Ageing at Newcastle University proclaimed that Medawar’s idea about natural selection losing its power appears to be “difficult to reconcile” with new research. The discussion on that point isn’t over yet, though.
Baudisch, for her part, would like theoretical frameworks that describe aging (or its lack) as part of the whole topography of change during a life. “Theories that just deal with the end of life don’t speak to all this diversity,” she says. “Physicists don’t make theories that only apply on Sundays.”
One long-standing approach does offer more of a whole-life framework. Kirkwood proposed the approach, called disposable soma theory, back in 1977. Wild organisms have to split their limited resources between reproduction and maintaining the soma, the nonreproducing parts of the body, he noted. In many cases, the best strategy demands such liberal spending on reproduction that there’s not enough left for full upkeep of the rest of the body. Aging is, in this interpretation, the sum of deferred maintenance.

The hydra species in the big lab test pursue a different strategy. Because they can regenerate and reproduce through budding, there is no distinction between reproductive cells and soma. Kirkwood says that the lack of senescence in hydra fits easily with the disposable soma approach. He would bet on their immortality.
He also thinks the ideas proposed by Medawar and Hamilton still have great value, with “central relevance to understanding aging.” To explain all the recently uncovered variety in aging, researchers may simply need more than these theories. They might need to know, for example, the particulars of a species’ home, be it pond, meadow or bug-rich forest.
Understanding these details may or may not allow humankind to do much about the process of aging. But creating better theories might finally reveal how some pale brainless squiggles of pond life may have achieved perpetual youth when humankind, despite all its apparent sophistication, has not.
This story appears in the July 23, 2016, Science News with the headline, “Aging’s Wild Side.”