LOOKING FOR LITHIUM Flamingos feast on tiny shrimp in the saline lagoons of Chile’s Salar de Atacama. Lithium and copper mining operations compete with the protected birds for the region’s scant water resources.
saxlerb/iStock/Getty Images Plus
The future of lithium is electrifying. Cars and trucks powered by lithium batteries rather than fossil fuels are, to many people, the future of transportation. Rechargeable lithium batteries are also crucial for storing energy produced by solar and wind power, clean energy sources that are a beacon of hope for a world worried about the rapidly changing global climate.
Prospecting for new sources of lithium is booming, fueled by expectations that demand for lightweight, rechargeable lithium batteries — to power electric vehicles, cell phones, laptops and renewable energy storage facilities — is about to skyrocket.
Even before electric cars, lithium was a hot commodity, mined for decades for reasons that had nothing to do with batteries. Thanks to lithium’s physical properties, it is bizarrely useful, popping up in all sorts of products, from shock-resistant glass to medications. In 2018, those products accounted for nearly half of the global lithium demand, according to analyses by the Frankfurt-based Deutsche Bank. Batteries for consumer electronics, such as cell phones or laptops, accounted for another 25 percent or so of the demand. Electric vehicles accounted for most of the rest.
300
percent
Global estimated increase in demand for lithium in the next 10 to 15 years
That breakdown will soon be turned on its head: By 2025, as much as half of the demand for lithium will be from the electric vehicle industry, some projections suggest. Global demand for the metal is expected to rise at least 300 percent in the next 10 to 15 years, in large part because sales of electric vehicles are expected to increase dramatically. Right now, there are about 2 million electric vehicles on the road worldwide; by 2030, that number is projected to grow to over 24 million, according to the industry research firm Bloomberg New Energy Finance. Electric vehicle giant Tesla has been on a worldwide quest for lithium, inking deals to obtain lithium supplies from mining operations in the United States, Mexico, Canada and Australia.
As a result, lithium prices in global markets have been on a roller coaster in the last few years, with a sharp spike in 2018 due to fears that there just might not be enough of the metal to go around. But those doomsday scenarios are probably a bit overwrought, says geologist Lisa Stillings of the U.S. Geological Survey in Reno, Nev. Lithium makes up about 0.002 percent of Earth’s crust, but in geologic terms, it isn’t particularly rare, Stillings says. The key, she adds, is knowing where it is concentrated enough to mine economically.

To answer that question, researchers are studying how and where the forces of wind, water, heat and time combine to create rich deposits of the metal. Such places include the flat desert basins of the “lithium triangle” of Chile, Argentina and Bolivia; volcanic rocks called pegmatites in Australia, the United States and Canada; and lithium-bearing clays in the United States.
The hunt to find and extract this “white gold” is also spurring new basic geology, geochemistry and hydrology research. Stillings and other scientists are examining how clays and brines form, how lithium might move between the two deposits when both occur in the same basin and how lithium atoms tend to position themselves within the chemical structure of the clay.
Seeking simpler sources
Lithium, in its elemental form, is soft and silvery and light, with a density about half that of water. It’s the lightest metal on the periodic table. The element was discovered in 1817 by Swedish chemist Johan August Arfwedson, who was analyzing a grayish mineral called petalite. Arfwedson identified aluminum, silicon and oxygen in the mineral, which together made up 96 percent of the mineral’s mass.
The rest of the petalite, he determined, was made up of some sort of element that had chemical properties similar to potassium and sodium. All three elements are highly reactive with other charged particles, or ions, to form salts, are solid but soft at room temperature, have low melting points and tend to dissolve readily in water. Thanks to their similarities, these elements, along with rubidium, cesium and francium, were later grouped together as “alkali metals,” forming most of the periodic table’s Group 1 (SN: 1/19/19, p. 18). Lithium’s affinity for water helps explain how it moves through Earth’s crust and how it can become concentrated enough to mine.
The basic recipe for any kind of lithium-rich deposit includes volcanic rocks plus a lot of water and heat, mixed well by active tectonics. Worldwide, there are three main sources of lithium: pegmatites, brines and clays.

Most pegmatites are a type of granite formed out of molten magma. What makes pegmatites interesting is that they tend to contain a lot of incompatible elements, which resist forming solid crystals for as long as possible. The rocks form as the magma beneath a volcano cools very slowly. The magma’s chemical composition evolves over time. As elements drop out of the liquid to form solid crystals, other elements, like lithium, tend to linger in the liquid, becoming more and more concentrated. But eventually, even that magma cools and crystallizes, and the incompatibles are locked into the pegmatite.
Before the 1990s, pegmatites in the United States were the primary source of mined lithium. But extracting lithium ore, primarily a mineral called spodumene, from the rock is costly. On top of the cost of actual mining, the rock has to be crushed and treated with acid and heat to extract the lithium in a commercially useful form.
In the 1990s, a much cheaper source of lithium became an option. Just beneath the arid salt flats spanning large swaths of Chile, Argentina and Bolivia circulates salty, lithium-enriched groundwater. Miners pump the salty water to the surface, sequestering it into ponds and letting it evaporate in the sun. “Mother Nature does most of the work, so it’s really cheap,” Stillings says.
What’s left behind after the evaporation is a sludgy, yellowish brine. To extract battery-grade lithium in commercially useful forms, particularly lithium carbonate and lithium hydroxide, the miners add different minerals to the brine, such as sodium carbonate and calcium hydroxide. Reactions with those minerals cause different types of salts to precipitate out of the solution, ultimately producing lithium minerals.

Compared with pegmatite extraction, the process for extracting lithium from the brine is extremely cheap; as a result, brine mining currently dominates the lithium market. But in the hunt for more lithium, the next generation of prospectors are looking to a third type of deposit: clay.
Clays are the hardened remnants of ancient mud, produced by the slow settling of tiny grains of sediment, such as within a lake bed. To get lithium-enriched clay requires the right starting ingredients, particularly lithium-bearing rocks such as pegmatite and circulating groundwater. The groundwater leaches the lithium from the rocks and transports it to a lake where it becomes concentrated in the sediments.
The western United States, it turns out, has all the right ingredients to make lithium-rich clay. In fact, in 2017 in Nature Communications, researchers suggested that some ancient supervolcano craters that became lakes, such as the Yellowstone caldera, would be excellent sources of lithium.
Beneath North America lies a shallow pool of magma that feeds the Yellowstone supervolcano. For the last 2 million years or so, Yellowstone volcanism has been located in northwestern Wyoming (and is the centerpiece of Yellowstone National Park). But the Yellowstone hot spot isn’t stationary. Over the last 16 million years, as the North American plate has slowly slid to the southwest, it has moved over the stationary, shallow magma body, leaving a pockmarked track of volcanic craters stretching from Nevada to Yellowstone. One of the oldest known Yellowstone craters, called McDermitt Caldera, filled with water, then later dried up, leaving behind a potential treasure trove of lithium-rich clay. Vancouver-based Lithium Americas Corp., which plans to begin mining operations at a site called Thacker Pass within the caldera in 2022, estimates that by 2025, the lake bed could provide as much as 25 percent of the world’s lithium.
In the United States, Stillings says, McDermitt is “one of the very large resources that we know exists.” But lithium clays have some hurdles to clear before they can compete with brines. Retrieving the lithium ore requires open-pit mining, which is more expensive than pumping up the brine. And processing the clay to extract lithium carbonate or other industry-ready minerals is also pricey. Lithium Americas and other companies that claim to have developed their own clean, inexpensive extraction processes haven’t yet demonstrated that they will be competitive with brine mining.
White gold
Most of the world’s lithium sources (orange) are pegmatite mines in Australia and China and brine mines in Chile and Argentina. But planned mining ventures (blue) mean that the lithium rush will soon spread to the United States, Canada and Mexico.
Known sources of lithium around the world
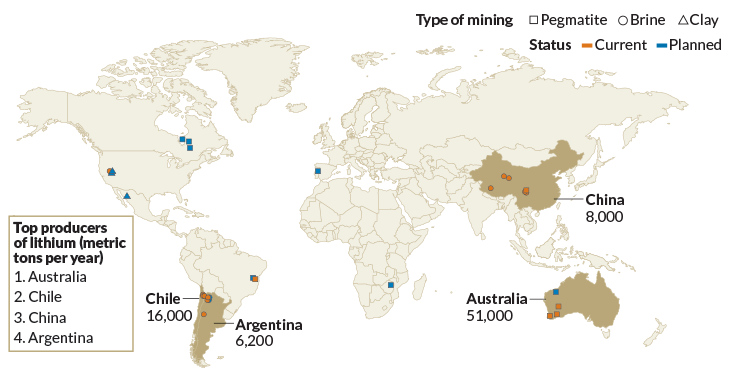

Source: USGS
Several other types of lithium extraction may be on the horizon, Stillings says. Lithium-rich brines can also form in tectonically active geothermal regions, where there is a lot of heat in the subsurface. Geothermal power plants already pump up the superheated water to generate energy, then inject it back into the subsurface. Some facilities are experimenting with extracting other commercially valuable elements from the brine, including lithium, manganese and zinc. Hydraulic fracturing, or fracking, also involves pumping up brines from the subsurface that may contain high levels of dissolved metals, possibly including lithium. Although the lithium may not be present in very high concentrations, the extraction could still be economically worthwhile, if it’s a by-product of mining already going on.
Revitalized research
In December 2017, the White House issued an executive order directing the U.S. Department of the Interior to ramp up research on new sources of certain “critical minerals,” including ores bearing lithium. Citing the economy and national security, the order instructed government scientists to analyze each link in the minerals’ supply chains, from exploration to mining to production, in hopes that new sources could be found within U.S. borders.
The United States isn’t alone in the rush to find lithium. China, the European Union and others are on the hunt for new sources. In January, a consortium of EU researchers launched a two-year initiative called the European Lithium Institute to become competitive in the lithium market.
To kick off this new phase in lithium research, Stillings helped convene a symposium at the American Geophysical Union’s annual meeting in Washington, D.C., last December. “We would like to understand how lithium cycles through Earth’s crust,” Stillings says. “Lithium is very soluble; it likes to be in solution. However, we’ve learned that as it moves through the crust, it does interact with clays.”
A multipurpose element
Lithium is useful for a lot more than batteries. Below are some common products and the lithium compounds they contain.
Mood stabilizer for bipolar disorder: Lithium has been used as a medication for conditions ranging from gout to mental disorders since the mid-19th century. Taken as lithium carbonate or lithium citrate, lithium has been in widespread use to treat acute mania, an aspect of bipolar disorder, since the 1970s.
However, scientists still aren’t sure why the treatment works. Due to their smaller size, charged particles, or ions, of lithium may substitute for potassium, sodium or calcium ions in certain enzymes and chemicals in the brain. Substituting lithium may reduce the sensitivity of certain receptors, making them less likely to connect to brain chemicals such as norepinephrine, which is known to be overabundant during mania.
Cosmetics: Lithium stearate acts as an emulsifier, keeping oils and liquids from separating in foundations, face powders, eye shadows and lipsticks. When added to face creams, a soft, greasy, lithium-bearing mineral called hectorite keeps the product smooth and spreadable.
Military, industrial, automotive, aircraft and marine applications: When added to petroleum, lithium stearate creates a thick lubricating grease that is waterproof and tolerant of high and low temperatures.
Shock-resistant cookware and aluminum foil: Compared with the other alkali metals, lithium atoms are small, particularly in their charged state. Lithium ions expand relatively little as they get hotter, so adding some lithium carbonate to glass or ceramics can make those products stronger and less likely to shatter when hot.
Lithium isotopes — it has two, lithium-6 and lithium-7 — are one way to track this exchange. “They are like a fingerprint,” says Romain Millot, a geologist with the French Geological Survey and the University of Orléans in France. The different masses of the two isotopes influence how they move between water and solid rock: Lithium-6 prefers to leave the water and bind into clay grains, compared with lithium-7. The isotopes are also proving useful at revealing the influences of weathering, water flow and heat on concentrating lithium, Millot says.
Because water is so important for concentrating lithium, researchers are shifting away from a classic “find the ore” framework, says Scott Hynek, a USGS geologist based in Salt Lake City. Instead, “we’re taking a more petroleum-like perspective,” he says. Scientists are tracking not just where deposits are, but how they might move: where the water flows, where the lithium-rich fluid could become trapped beneath a layer of hard, impermeable rock.
Lithium prospecting is also taking a page from the hydrology playbook, using some classic tools of that trade to track the circulation of groundwater through the subsurface to suss out where lithium-rich deposits might end up. Isotopes of hydrogen, oxygen and helium are used to track how long the groundwater has been traveling through the subsurface as well as the types of rocks that the water has been in contact with.
Faults, for example, can channel subsurface water, and therefore may play a big role in shaping where lithium deposits might form. “It’s an unresolved question,” Hynek says. “These are big-scale geologic controls on where high-lithium water goes.” He presented data at the AGU symposium suggesting that the highest lithium concentrations in a Chilean salt flat known as the Salar de Atacama occur near certain fault lines. That, he says, suggests the faults are helping to channel the groundwater and thereby concentrating the deposits.
Do no harm
One looming problem for lithium mining is that even “clean” energy isn’t completely clean. Extracting lithium from its ore and converting it into a commercially usable form such as lithium carbonate or lithium hydroxide can produce toxic waste, which can leak into the environment. Chemical leaks from a lithium mine in China’s Tibetan Plateau have repeatedly wreaked havoc on the environment since 2009, killing fish and livestock that drank from a nearby river.
Even when Mother Nature is doing much of the work, such as in evaporation ponds, there can be negative effects on the environment. In South America, for example, the problem is water supply. The lithium triangle, which includes Salar de Atacama, is one of the driest places on Earth — and mining consumes a lot of water. And that’s producing a worrisome confluence of events. Just at the edges of the Salar de Atacama salt flats is a flamingo nesting habitat: brackish lagoons filled with brine shrimp. “One of the major oppositions to this mining activity is the impact it has potentially on flamingo populations,” Hynek says. The same water source in the Andes that feeds the subsurface lithium brine reservoir also, ultimately, fills the lagoons.

In fact, the water table is already dropping in some places in the region, and indigenous communities, as well as both Chilean and Argentinian authorities, are on high alert, Hynek says. “Chilean authorities are worried that [miners] will pump so much that the lagoon water levels will also drop.” In February, Chile announced new restrictions on water rights for miners operating in Salar de Atacama.
Who’s to blame is the subject of a lot of debate. In addition to the lithium brine mining, copper mines high up in the Andes — where the groundwater originates — are extracting a substantial amount of water from the system. “The flamingos and the indigenous communities are literally stuck in the middle,” Hynek adds.
Such big environmental concerns could hamper future prospects for mining in the region. “You’re making the brine in the same area where you’re sustaining these important biodiversity habitats,” says David Boutt, a hydrologist at the University of Massachusetts Amherst.
There is so far little research on how water moves through the subsurface in dry areas with very low precipitation rates, such as South America’s lithium triangle, Boutt adds. “There are a lot of questions about where the water is coming from,” such as how variable the water flow rate is through the ground. “It can take a very long time for these systems to respond” to perturbations such as groundwater pumping.
The effects of withdrawing the briny waters now might not be felt for perhaps decades. “A concern,” Boutt says, “is whether we are going to be waiting 100 years before something bad happens.”
This article appears in the May 11, 2019 issue of Science News with the headline, “Looking for Lithium: The lightest metal on the periodic table is key to clean energy’s future.”