Borrowing a trick from nature, researchers have switched off the extra chromosome that causes Down syndrome in cells taken from patients with the condition.
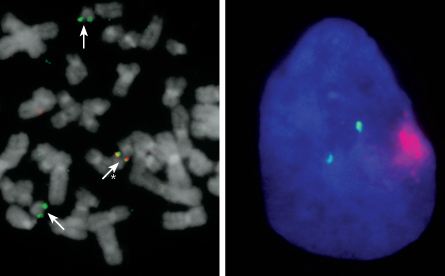
Though not a cure, the technique, reported July 17 in Nature, has already produced insights into the disorder. In the long run it might even make the flaw that causes Down syndrome correctable through gene therapy.
“Gene therapy is now on the horizon,” says Elizabeth Fisher, a molecular geneticist at University College London. “But that horizon is very far away.”
Down syndrome, also called trisomy 21, occurs when people inherit three copies of chromosome 21 instead of the usual two. It is the most common chromosomal condition, affecting around one in every 700 babies born in the United States. People with the disorder typically have both physical and cognitive complications of having an extra chromosome.
“Down syndrome has been one of those disorders where people say, ‘Oh, there’s nothing you can do about it,’ ” says Jeanne Lawrence, a chromosome biologist and genetic counselor at the University of Massachusetts Medical School in Worcester, who led the study with colleagues Lisa Hall and Jun Jiang.
The researchers decided to see whether they could shut down the extra chromosome by drawing on a biological process called X inactivation. Women have two X chromosomes and men have only one X and a Y. To halve the amount of X chromosome products, female cells shut down one copy. Cells do that using a chunk of RNA called XIST, which is made by one X chromosome but not the other. The RNA works by pulling in proteins that essentially board up the chromosome like an abandoned building. The other X stays on by making a different RNA.
Lawrence and Hall thought that if they put XIST on another chromosome, it might shut that one down too. So Jiang put the gene for XIST onto one of the three copies of chromosome 21 carried by stem cells grown from a man with Down syndrome. That copy of the chromosome got switched off.
“It’s kind of surprising that it wasn’t done before. I’m smacking my own forehead and saying, ‘duh,’ ” says Roger Reeves, a geneticist at Johns Hopkins University.
One idea about why an extra chromosome 21 causes cognitive problems is that it may slow down the growth of brain cells. Jiang grew nerve cells from the Down patient’s stem cells to see how cells with one shut-down chromosome developed compared with cells bearing three active copies. The cells with only two working chromosomes grew faster, forming clusters of nerve cell precursors within two weeks, while the uncorrected cells needed four or five additional days.
The work is an enormous step forward in Down syndrome research, Fisher says, and “may take us much closer to understanding the molecular basis of the disorder.” The technique could allow researchers to figure out which genes are involved in Down syndrome and how extra copies affect cells and ultimately the body, she says.
Reeves wants to use the technology in animal experiments, a critical step in determining whether it could find use as gene therapy for people with Down syndrome. He plans to work with Lawrence’s group to switch off the extra chromosome in mice engineered to have a disorder that simulates some features of Down syndrome.
But Reeves doubts that scientists could use the method to switch off the extra chromosome in every cell in the body. Doing so would probably require gene therapy at a very early stage of pregnancy, something scientists don’t know how to do. “I just don’t see how we would get there from where we are today,” Reeves says.
Such universal silencing of the extra chromosome may be necessary to forestall developmental problems. But other problems associated with Down syndrome might be prevented or reversed by shutting down the extra chromosome after birth. For instance, people with Down syndrome are at high risk of developing childhood leukemia and of getting Alzheimer’s disease. Gene therapy to turn off the extra chromosome in the bone marrow or the brain might prevent those problems.
Therapeutic possibilities are still far in the future and may never pan out, says William Mobley, a neurologist and neuroscientist at the University of California, San Diego. “We have to move cautiously and deliberately and not say that a cure for Down syndrome is on the horizon,” he says. “It’s not true, but gosh is there excitement that progress is being made.”
Back Story | A DNA Challenge of Stupendous Size
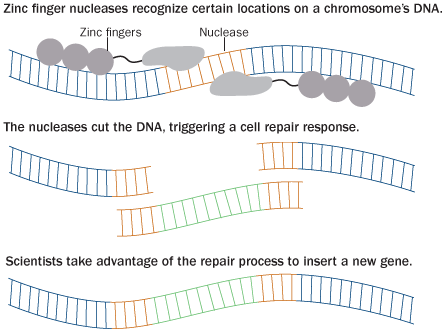
The idea came easily, says Jeanne Lawrence: Mimic nature by using RNA to shut down chromosome 21. But executing the vision was a feat that has left other scientists in awe.
Part of the problem is that the piece of RNA, called XIST, is huge. Previously, researchers had spliced into a chromosome pieces of genetic material shorter than about 8,000 DNA units, or nucleotides, says Jun Jiang, the University of Massachusetts Medical School colleague Lawrence says “worked night and day for five years” to pull off the project. At about 21,000 nucleotides, XIST dwarfed the previous record holder. “Getting something that big onto a chromosome had never been done before,” Lawrence says.
But Jiang did it. She customized proteins called zinc finger nucleases (SN: 7/30/11, p. 9) to accomplish the goal. Zinc fingers are structures that recognize and grab onto a specific sequence of DNA letters. Jiang engineered fingers to seek out a particular spot on chromosome 21. She tethered nucleases, enzymes that work in pairs to cut DNA, to the zinc fingers.
Cells repair breaks in DNA using a process called homologous recombination. The process normally uses the matching chromosome as a guide for sealing the breach. Instead, Jiang supplied a template containing the enormous XIST gene sandwiched between two bits of DNA that matched sequences on either side of the gap. So when cells healed the wounded copy of chromosome 21, they also incorporated the XIST gene.
If the technique works for chromosome 21, it might apply to lethal conditions that are caused by having three copies of other chromosomes. The research really goes beyond gene therapy to chromosome therapy, says Montserrat Anguera, a biologist at the University of Pennsylvania. — Tina Hesman Saey
Editor’s Note: This story was updated on July 17 to add researcher Jun Jiang’s name. It was further updated July 24 to correct the number of days it took the cells to form neural precursors.







