Earth’s stratosphere is a mysterious superhighway for microbes
Some organisms found on Earth’s surface lead a double life riding high in the atmosphere

Seen from a scientific balloon 30 kilometers above New Mexico, the stratosphere is a gray zone between Earth and space. The air pressure at this height is one percent that at sea level and the ultraviolet radiation is intense. But scientists are learning that some microbes survive here, at least temporarily.
Elvis Andrés Ayala/Trinidad System
The stratosphere is both beautiful and hostile — a purgatory between Earth and space, between life and death.
Hover 30 kilometers above the ground, and you are nearly twice as high as any raincloud on Earth. The surface of the planet curves beneath you. A diaphanous film of blue stretches over that horizon, representing the disconcertingly thin layer of atmosphere that envelops all life as we know it. Above that, the sky resembles black interplanetary space.
You would quickly die here. The air pressure is one percent what it is at sea level. As you gasped for oxygen, your blood would boil inside you, causing your skin to welt like bubble wrap.
But the stratosphere holds plenty of life — tiny single-celled microbes that somehow navigate extreme dehydration, temperatures as low as −60° Celsius and intense DNA-damaging ultraviolet radiation that would kill most life on Earth’s surface.
“If you took a microbe from those altitudes and you put them on the surface of Mars, they wouldn’t even know the difference,” says microbiologist Brent Christner of the University of Florida in Gainesville.
When Christner and his team started looking for life high in the atmosphere over a decade ago, they intended to find the upper limits of Earth’s habitable zone. They hoped that this, in turn, might show whether life could persist on the cold, radiation-pummeled surface of Mars, where the atmosphere is just as thin. But when Christner’s graduate student Noelle Bryan sent sampling balloons to 38 kilometers above Earth’s surface, she was utterly surprised by what they found: “We did not hit an altitude where we couldn’t find something [alive],” says Bryan, who is now a senior research manager at Mass General Brigham in Boston.

In 2025, Christner’s team revealed something even more surprising. The microbes the researchers found weren’t the kind of extremophiles that you’d expect at the edge of space. They looked nothing like the exotic organisms that inhabit extreme habitats on Earth, such as boiling hot springs, deep subterranean rocks or pools of concentrated acid.
Instead, they turned out to be some of the same humdrum bugs that grow on our crops, our gardens, even our skin. These well-known critters may live a secret double life that few people imagined: flying around the world at two to three times the height of a commercial jetliner.
The atmosphere “is like a highway system,” Christner says. It allows microbes “to move globally in periods of weeks,” crossing oceans and settling in new habitats. This realization expands our view of the biosphere and evolution. It could reshape our understanding of how pathogens spread around Earth. And it could transform how we look for life on other worlds — on Mars, in the clouds of Venus and even on exoplanets light-years away.
Hunting for microbes in the sky
Scientists have wondered for a century what sort of life, if any, might exist high in the atmosphere. For decades, their explorations yielded only tantalizing hints.

In 1935, the U.S. Army Air Corps launched a helium balloon to a height of more than 22 kilometers over the Black Hills in South Dakota, setting a world record. Two pilots in a pressurized gondola recorded air temperatures, magnetic fields, radiation levels and dozens of other measurements. They also dropped a sterile sampling device designed to collect airborne microbes on its way down. After falling halfway, it closed and parachuted back to Earth with its sample sealed inside.
Scientists managed to grow 10 types of bacteria and fungi from the sample that were ostensibly collected during its fall through the lower stratosphere. But in this era before scientists knew about DNA, they never learned much more than the shapes of the cells and the temperatures and nutrients that they needed to grow. Much later, in 1974, Soviet scientists launched several rockets equipped with sticky microbe collectors 48 to 77 kilometers into the mesosphere — the atmospheric layer above the stratosphere, where conditions are even more extreme. The collectors parachuted back to the ground and arrived with a few living cells embedded in them, which scientists again managed to grow in the lab.
That Soviet work is widely cited, Christner says, “but there’s just no way to know what they sampled” and whether it came from contamination at ground level. He doesn’t believe that they actually found something alive at that altitude.
A new generation of high-altitude studies started in the early 2000s.
NASA was flying its ER-2 aircraft (a civilian version of the U-2 spy plane) 20 kilometers up in the stratosphere to collect cosmic dust that filters down from space and use it to study the history of the solar system. Microbiologist Dale Griffin, then with the U.S. Geological Survey in St. Petersburg, Fla., arranged to have sticky microbe catchers taken on several of those flights from 2003 to 2008.
Griffin grew several kinds of bacteria from those samples. And unlike his predecessors, he subjected them to a simple DNA analysis called genetic barcoding that used a short sequence from a single gene to get a general idea of which family or genus they belonged to. These bacteria were related to several that were known from remote islands and volcanic soils, Griffin found. He speculated that eruptions might have lofted them into the stratosphere.

Taken together, the U.S. and Soviet observations didn’t reveal much. They depended on growing microbes in the lab, but scientists can’t grow 99 percent of the microbes that exist in most environments because they can’t re-create the proper growing conditions. As such, the findings didn’t reveal how many living cells were present in the stratosphere. They often didn’t identify the species. The scientists didn’t test whether these microbes could survive the extreme cold, desiccation or UV radiation found in the stratosphere. And they never identified the lifestyles of these microbes on Earth’s surface — whether the creatures lived on plants, slurped raw sewage or grew in the armpits of NFL linebackers.
Christner was at Louisiana State University in Baton Rouge when he and Bryan started their search for life in the stratosphere in 2008. They planned to start low and sample progressively higher. They would try to grow the cells they collected, as other people had previously done.
But they would also count the total number of living cells, something no one had ever done before at that altitude.
They expected that at some altitude, the number of living cells would drop to zero. This would define the outer limits of life on Earth — and hint at the possible limits of life on Mars.
Bryan spent three years building a series of microbe collectors, using Styrofoam and balsa wood purchased at Hobby Lobby and Home Depot. She launched these contraptions into the air dangling from helium balloons. During a dozen launches in Texas and Louisiana, she attempted to sample airborne microbes from as high as 25 kilometers. Each device included a control chamber that didn’t open during the flight, allowing her to check whether the samples were contaminated with ground-level microbes. She gradually improved her design, bringing the levels of contamination down to almost zero.
By 2013, Bryan was using a more advanced system developed by LSU engineer T. Gregory Guzik. Its rugged circuits allowed it to reach even higher altitudes, where the thin air can cause electricity to arc from one wire to another, damaging a device. She and Guzik used this system in a pivotal series of balloon launches over Fort Sumner, N.M. These would be the last samples that she collected for her Ph.D. research, and they would transform the endeavor from a cool science project into a major discovery.
A scientific balloon bonanza
The town of Fort Sumner crouches amid an arid plain of grass and yucca in eastern New Mexico. The 19th-century gunslinger Billy the Kid is buried in a cemetery at the south end of town. Just north of town is the airport, consisting of a corrugated steel hangar and cracked tarmac. The facility is occasionally used for launching scientific balloons, which benefit from the area’s stable weather and flat, undeveloped terrain.
On the morning of August 21, 2013, Bryan, Guzik and several other people wearing hard hats emerged from the hangar grasping the tether of a spherical white helium balloon the size of a small elephant. It bobbed and tugged impatiently as they walked it into an open field and let go. It surged upward, pulling its payload into the sky.
A downward-pointing camera captured video as it rose. The payload swung lazily as the desert shrank into a hazy, wrinkled mosaic. A large jet — perhaps a Boeing 737 — streaked by far below, tiny and silent. Later, the balloon was tossed by violent winds, marking its passage into the stratosphere.
When the balloon reached 18 kilometers, an electric motor whirred, opening a compartment and exposing a sticky collector to snag airborne cells. Minutes later, as the balloon passed 23 kilometers, the compartment door closed. An electric heating coil severed the balloon tether, sending the payload parachuting back to Earth.
Meanwhile, Bryan drove west on Highway 60 across empty rangeland while a fellow student tracked the balloon via GPS. She eventually veered off the road onto the dusty plain and after several miles found her germ catcher nestled between a yucca and a prickly pear cactus.
This was one of six balloons that Bryan, Guzik and colleagues launched during a three-week stint in 2013; some reached as high as 29 kilometers. A seventh launch on a larger NASA balloon reached 38 kilometers during that same period.
But in retrospect, the August 21 launch would loom especially large. That afternoon in the airport hangar, Bryan tested the sample for ATP, an energy molecule found in living cells. “I really thought everybody was going to be dead,” she says. But test after test revealed the presence of ATP, prompting her to think, “Oh my God, they’re alive.”
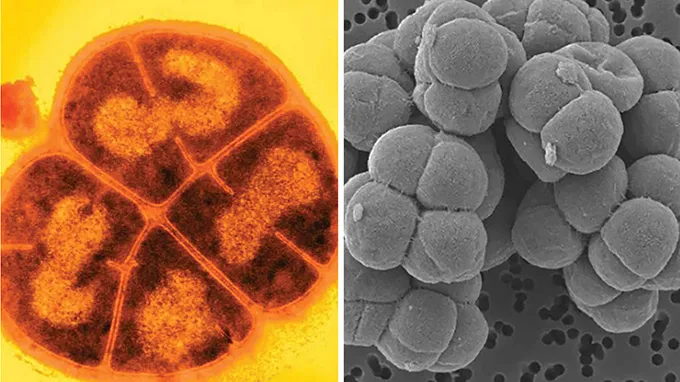
Back at LSU, Bryan spent several months incubating her samples. The one from August 21 spawned a glistening orange bacterial colony, which she named L6-1. Samples from other launches spawned yellow, pink and black colonies. The colors probably indicated well-known pigments that protected the cells by blocking some UV radiation.
Bryan assumed that these still-unidentified microbes would turn out to be tough cookies — maybe belonging to a group called Deinococcus that can survive intense radiation, or to another group such as Bacillus that can compact itself into inanimate spores able to survive boiling. But as she isolated and barcoded their DNA repeatedly in 2013 and 2014, a very different picture emerged.
Only one out of a dozen or so bacteria turned out to be the sort of spore-former that Bryan expected. L6-1 and several others belonged to a genus called Curtobacterium that often lives on plants. Others belonged to groups that inhabit soil.
It “should have been nothing but spore-formers,” Bryan says. It “should have been nothing but extremophiles.” The fact that these critters appeared to be neither presented a mystery that she desperately wanted to solve. Fully unraveling it would take a decade of work.
Plant pathogen superhighway
Bryan spent most of 2014 and 2015 doing work that was crucial to her Ph.D., including rigorous calculations and lab experiments to convert the cell counts from her balloons into estimates of how many live cells are floating around in the stratosphere.
These calculations would ultimately get published with Christner and Guzik in 2019 in the ISME Journal. The work suggested that at 24 kilometers high, the concentration of living cells was 100,000 per cubic meter. At 36 kilometers, the air still held nearly 8,000 cells per cubic meter.
In 2016, Bryan turned back to her mystery microbes for a new set of experiments. She tested their tolerance to UV-C radiation — an ultraviolet band that damages DNA and kills cells far more potently than the UV-A and UV-B bands that cause sunburns. UV-C is absent on Earth’s surface because it’s blocked by the ozone layer, but it’s abundant in the mid-stratosphere.
Bryan compared her stratospheric germs to a bacterium called Deinococcus radiodurans that can survive roughly 1,000 times the radiation dose lethal for a human. D. radiodurans was long considered the toughest organism on Earth, after its accidental discovery in 1956. Scientists working with the U.S. military were trying to sterilize canned meat with gamma radiation. When they later opened the cans, they found the meat putrid, spoiled by these bacteria that had somehow survived.
D. radiodurans is also uniquely resistant to UV-C radiation. But when Bryan compared L6-1 to the supermicrobe, she was shocked at the results. Her dainty little germ, accustomed to the cool, shady canopies of plants, was just as tolerant to UV-C as D. radiodurans. And some of her other plant and soil bacteria weren’t far behind.
“Clearly I [fudged] up,” she remembers thinking — though using a spicier verb. But even as she repeated the experiments over a period of months, looking for potential errors, the results stayed the same.
This moment in 2016 was when the team’s understanding “all sort of came together,” Christner says. Curtobacterium, the plant- and dirt-dwelling genus that L6-1 belonged to, was “pretty abundant in the atmosphere,” he says. “Almost every time we put up a balloon and collected samples, whether it’s in the high troposphere or in the stratosphere, we are collecting something that is highly similar to this.”
Discovering that L6-1 was resistant to radiation and drying hinted that it wasn’t up there by accident. It suggested that these plant-dwelling bacteria, closely related to species that were well known, had a secret talent that no one suspected: surviving, at least briefly, at the edge of space.
This was the state of knowledge when Bryan finished her Ph.D. in 2017. Her discoveries had followed a path no one expected. But the story wasn’t complete.
As Bryan began a postdoctoral fellowship at MIT, Christner moved from LSU to the University of Florida, where he began to examine another intriguing aspect of L6-1. Based on Bryan’s earlier DNA barcoding, L6-1 and several of the other stratospheric bacteria were closely related to a notorious crop pathogen called Curtobacterium flaccumfaciens. It had plagued Midwestern farms from the 1920s to the 1970s, causing entire bean fields to brown and wilt. It had gradually faded before re-emerging around 2003.
“Plant pathogens in the
Brent Christner
stratosphere — that was
the furthest thing that
we expected to run into.”
microbiologist
Hoping to find out whether the stratospheric bacteria were also pathogens, Christner reached out to two plant pathologists in 2020, Brian Kvitko at the University of Georgia in Athens and Robert Harveson at the University of Nebraska in Scottsbluff. Sure enough, they found that L6-1 caused bean plants to yellow and wilt. So did two other Curtobacterium microbes Bryan had collected from as high as 29 kilometers.
That same year, Christner’s new graduate student, Adam Ellington, sequenced the full genome of L6-1, revealing it as a new species, which they named Curtobacterium aetherium. It matched the DNA sequence of another unidentified pathogen that Harveson isolated from a diseased millet crop in Nebraska years before.
The discovery that C. aetherium was a plant pathogen was finally published in 2025 in Microbiology Spectrum. “Plant pathogens in the stratosphere — that was the furthest thing that we expected to run into,” Christner says.
Harveson, a worldwide authority on Curtobacterium, had long believed that these pathogens spread on infected seeds. He was stunned to learn that at least some members of its genus could survive so high in the atmosphere. “I don’t doubt that it can move that way,” he says. “I’m just amazed that so many of these things survived.”
“Brent [Christner] and his group are kind of the pioneers for this,” Harveson now says. What they found could “change our thought processes about movement of diseases.”
Life finds a way
The advantages of traveling long distances through the atmosphere could be huge. By surviving those journeys, a microbe could cross oceans and sprinkle down on new ecosystems, diversifying into countless niches. A pathogen like C. aetherium could escape regions where plants develop resistance and land on new hosts. Evidence has been building for some time that plant- and soil-dwelling microbes are well suited for making the jump.
Bacteria that live on leaves are known to experience frequent drying, which can damage their DNA in a manner similar to radiation. So some of them can already repair their DNA reasonably well. Leaf bacteria can also loft into the air with surprising ease. One landmark study published in 1982 found that during a warm day in a bean field, microbes levitated into the air — nearly 20 billion cells per hectare per hour — lifted by breezes, thermal updrafts or electric fields that coalesce around the drying leaves. Bacteria may even encourage this by clumping onto tiny leaf hairs where air currents can more easily lift them.
Once airborne, microbes probably have several ways of getting 10 or 20 kilometers into the air. Major thunderstorms, dust storms, hurricanes and volcanic eruptions can rapidly lift them. Smoke plumes from large wildfires can lift billions of live cells into the air, and in some cases can reach the stratosphere.
Ellington did work that suggests C. aetherium could have originated from a leaf-squatting, levitating ancestor, which then evolved new adaptations for surviving extreme altitudes as it got lofted higher.
In one experiment, Ellington looked at C. flaccumfaciens, the crop pathogen that’s closely related to C. aetherium but has not been found high in the atmosphere. He found that it is already moderately tolerant to UV-C radiation. When he exposed it to 12 rounds of UV-C, its survival at high doses increased by another 20- to 80-fold. This bolsters the idea that some plant-dwelling bacteria have a natural flair for evolving extreme radiation protection if they’re frequently lofted high into the atmosphere.
When Ellington examined the genes that C. aetherium turns on during UV-C exposure, he found three especially notable DNA repair enzymes — suggesting that its particular style of UV-C resistance may be well-suited for the high atmosphere.
The “most intriguing” was an enzyme called spore photoproduct lyase, Ellington says. It repairs an unusual type of UV damage that occurs only when a cell is so dehydrated that its DNA re-arranges from the normal double helix into a tighter, crystalline form. A cell that’s drifting in the stratosphere is “essentially in a desiccated state,” Ellington says. So having this enzyme might allow it to repair a type of DNA damage that only happens in those conditions.
Noelle Bryan
“ For me it was space
exploration. It was
uncharted territory.”
senior research manager
Ellington found that C. aetherium also uses two different photolyase enzymes. Unlike many other DNA repair proteins, these don’t require food-derived energy to do their job. Instead, they repair DNA using sunlight. This is a “huge advantage for an organism that’s trying to survive” in the atmosphere, where food is scarce but sunlight is plentiful, Ellington says. He and colleagues published the findings in March 2025 in Microorganisms.
Food scarcity may represent a major challenge for microbes in the atmosphere. Some bacteria may have other strategies for coping with it. While sampling clouds drifting over remote Réunion Island in the Indian Ocean, aerobiologist Pierre Amato at the University Clermont Auvergne in France found a strain of Methylobacterium that harnesses sunlight to produce the energy molecule ATP. This could allow it not only to repair its DNA without burning precious calories, but also to do countless other tasks, like manufacturing proteins.
Microbiologist Chris Greening of Monash University in Clayton, Australia, believes that airborne bacteria can also tap another hidden energy source. He has found that many bacteria living on trees, in soils and even drifting a few meters up in the air can oxidize hydrogen and carbon monoxide, which are trace gases in the atmosphere. He’s recently found that some microbes can even burn hydrogen at –40° Celsius, a temperature similar to the stratosphere’s. In addition to releasing energy, hydrogen oxidation actually produces water as its sole combustion product. “So, it is hydrating the cell,” Greening says. “That might be just enough for some cells to avoid desiccation.”
The work done so far has revealed a coherent story about how and why C. aetherium and some of its cousins could have evolved to survive high in the atmosphere. But many other stories may still hide among the thousands of other living cells that Bryan retrieved. She managed to grow only 12 or so species of bacteria from the stratosphere, but for every type of microbe that did grow in the lab, 100 to 1,000 others may have been present but didn’t grow in the culturing conditions that she used. Some of them might even be more surprising.
“The next question would be to understand the whole community” up there, says Burak Erkorkmaz, a microbiologist at the University of Gothenburg in Sweden.
He envisions using a method in the stratosphere that he employed several years ago, sampling air at ground level in Israel. He sequenced DNA from all of the cells that he collected, identifying hundreds of species. Doing this in the stratosphere would provide a fuller picture of the species potentially present there. It would hint at whether they, like C. aetherium, carry genes for surviving at high altitudes and how that might fit into their evolutionary niche.
Scientists are already testing that approach. In 2018, NASA’s C-20A jet sampled microbes from up to 12 kilometers over California, just below the stratosphere. Initial results showed DNA sequences for microbes that inhabit soil, rotting leaves and the anatomic nooks and crannies of humans or animals, likely derived from farmlands of the Central Valley, directly upwind.
Many questions remain. Although Bryan estimated that C. aetherium can hunker down for a few months or weeks at 20 to 36 kilometers, returning to Earth soon enough to survive the trip would still require some luck. For this reason, even C. aetherium probably makes most of its successful flights at lower altitudes, say 5 to 15 kilometers, where conditions are more mild — though still lethal for most microbes. But even so, simply finding that these bacteria can survive in the stratosphere has huge implications. It is spurring new thoughts about where life might survive in other worlds. Including ones that are hellishly hot.
From Earth to exoplanets

The lead-melting surface of Venus is inhospitable to life as we know it. But scientists have noticed some tantalizing chemical signatures in a cloud layer 48 to 60 kilometers above the surface, where temperatures and atmospheric pressures are similar to those at Earth’s surface. Potential traces of several gases, including oxygen, methane, ammonia and phosphine, have fueled speculation that Venusian microbes might inhabit those clouds, producing these gases through their metabolism.
If such cloud-dwelling microbes exist, they would have to survive periodic bouts of nastiness as they sank into lower, hotter layers of the atmosphere. In 2021, Bryan and Sara Seager, a planetary scientist at MIT, proposed in the journal Astrobiology that those Venusian microbes might survive the way C. aetherium seems to in Earth’s atmosphere: by temporarily drying out and hunkering down, then re-awakening as they’re swept back into cooler, wetter cloud layers.
Bacteria that Bryan isolated from the stratosphere in 2013 could also guide the search for life in other solar systems that are light-years away.
For example, the critters grown in the lab produced those vivid pigments that are known to protect microbes from UV radiation. Last November, astrobiologist Lígia Coelho of Cornell University suggested that scientists could use this trait to identify distant planets with microbial life in their atmospheres.
Such life might also have to protect itself from UV radiation. By analyzing pigments from Bryan’s stratospheric bacteria, including C. aetherium, Coelho identified their specific patterns of absorbance and reflectance at certain wavelengths of light.
“We might be able to capture [these patterns] with future telescopes” looking at Earth-sized exoplanets, Coelho says. The initial candidates would be small planets orbiting nearby stars such as Proxima Centauri, which is about four light-years from Earth.
“This is opening a whole new paradigm” for looking for life on exoplanets, she says — especially on Earthlike planets with a lot of liquid water and clouds that might obscure the planetary surface.
But discovering C. aetherium in the stratosphere is probably most relevant for thinking about life on Mars. C. aetherium itself probably couldn’t survive on Mars because it requires oxygen, which is largely absent there. But its sheer toughness suggests that other microscopic Earthlings probably could.
Astrobiologist Andrew Schuerger at the University of Florida in Gainesville has spent years looking for candidates. In 2025, he and Rachel Harris, an astrobiologist with Harvard and NASA, reported that a microbe called Methanosarcina barkeri comes surprisingly close.
M. barkeri normally lives in sewage, landfills, rice paddies and lake bottoms without oxygen. It obtains energy by converting carbon dioxide and hydrogen into methane.
Because M. barkeri often lives underground and underwater, one would hardly expect it to thrive in the extreme dryness and thin atmosphere on Mars. Still, Schuerger and Harris found that even at low, Marslike gas pressures — similar to Earth’s stratosphere — it continues to produce methane.
For Mars enthusiasts, that’s big news. Orbiters and rovers have repeatedly detected methane gas seeping into the planet’s atmosphere, a potential byproduct of microbial life beneath the surface. Due to the low pressure and dryness on the Martian surface, those methane-making critters might have to live kilometers underground, making them nearly impossible for a Mars rover to find.
But finding that M. barkeri is active at such low pressures “opens up the entire crust from the surface [downward]” as possible habitat on Mars, Schuerger says. If a Mars rover drilled 20 centimeters into the ground in the right place, it could potentially encounter tiny single-celled Martians — if they exist.
The niches where microbes could live just below the Martian surface might still be quite small, says astrobiologist Lynn Rothschild at NASA Ames Research Center in Mountain View, Calif. But Rothschild imagines Martian microbes using the surface of the planet the way C. aetherium seems to use Earth’s atmosphere: as a hostile environment that can be tolerated for a period time to move from one place to another.
She imagines windstorms on Mars carrying not only dust, but also Martian microbes swept up with it. Microbes “would be floating around, just looking for a place to land [where] they can survive” and grow for a while, Rothschild says. If so, then a rover might find life the way a fence finds blowing leaves.
Years may pass before the lessons from Earth’s stratosphere find their way into the search for life on Mars or Venus. But for Bryan, the excitement of it began years ago, even as she made her first tentative trips to Hobby Lobby, gathering materials that she would use to catch bacteria high up in the sky.
“For me it was space exploration,” Bryan says. “It was uncharted territory.”





