Brain chemical lost in Parkinson’s may contribute to its own demise
In a hopeful note, treating dopamine-producing nerve cells with antioxidants lessened damage
DOPAMINE DAMAGE In Parkinson's disease, a dangerous form of the chemical messenger dopamine may help destroy the nerve cells that produce it, a new study suggests.
Naeblys/istockphoto
The brain chemical missing in Parkinson’s disease may have a hand in its own death. Dopamine, the neurotransmitter that helps keep body movements fluid, can kick off a toxic chain reaction that ultimately kills the nerve cells that make it, a new study suggests.
By studying lab dishes of human nerve cells, or neurons, derived from Parkinson’s patients, researchers found that a harmful form of dopamine can inflict damage on cells in multiple ways. The result, published online September 7 in Science, “brings multiple pieces of the puzzle together,” says neuroscientist Teresa Hastings of the University of Pittsburgh School of Medicine.
The finding also hints at a potential treatment for the estimated 10 million people worldwide with Parkinson’s: Less cellular damage occurred when some of the neurons were treated early on with antioxidants, molecules that can scoop up harmful chemicals inside cells.
Study coauthor Dimitri Krainc, a neurologist and neuroscientist at Northwestern University Feinberg School of Medicine in Chicago, and colleagues took skin biopsies from healthy people and people with one of two types of Parkinson’s disease, inherited or spontaneously arising. The researchers then coaxed these skin cells into becoming dopamine-producing neurons. These cells were similar to those found in the substantia nigra, the movement-related region of the brain that degenerates in Parkinson’s.
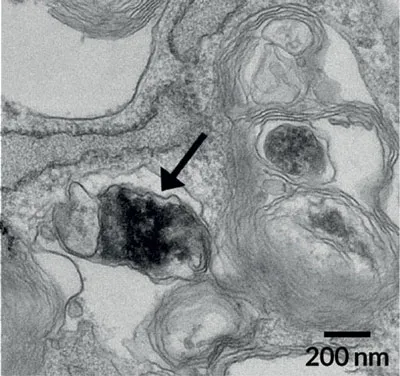
This dangerous form of dopamine seemed to kick off other types of cellular trouble. Defects in the cells’ lysosomes, cellular cleanup machines, soon followed. So did the accumulation of a protein called alpha-synuclein, which is known to play a big role in Parkinson’s disease.
Those findings are “direct experimental evidence from human cells that the very chemical lost in Parkinson’s disease contributes to its own demise,” says analytical neurochemist Dominic Hare, of the Florey Institute of Neuroscience and Mental Health in Melbourne, Australia. Because these cells churn out dopamine, they are more susceptible to dopamine’s potential destructive forces, he says.
When researchers treated neurons carrying a mutation that causes inherited Parkinson’s with several different types of antioxidants, the damage was lessened. To work in people, antioxidants would need to cross the blood-brain barrier, a difficult task, and reach the mitochondria in the brain. And this would need to happen early, probably even before symptoms appear, Krainc says.
“Without this human model, we would not have been able to untangle the pathway,” Krainc says. In dishes of mouse neurons with Parkinson’s-related mutations, dopamine didn’t kick off the same toxic cascade, a difference that might be due to human neurons containing more dopamine than mice neurons. Dopamine-producing neurons in mice and people “have some very fundamental differences,” Krainc says. And those differences might help explain why discoveries in mice haven’t translated to treatments for people with Parkinson’s, he says.
Over the past few decades, scientists have been accumulating evidence that oxidized dopamine can contribute to Parkinson’s disease, Hastings says. Given that knowledge, the new results are expected, she says, but still welcome confirmation of the idea.
These toxic cellular events occurred in lab dishes, not actual brains. “Cell cultures aren’t the perfect re-creation of what’s going on in the human brain,” Hare cautions. But these types of experiments are “the next best thing for monitoring the chemical changes” in these neurons, he says.
Editor’s note: This story was updated September 11, 2017, to correct Dominic Hare’s affiliation.







