In the middle of a cattle ranch near Gerlach, Nev., enclosed by a corrugated metal fence, are small pools of steaming water. Close to the surface of these pools, water temperatures reach about 90° Celsius; deeper down, it’s even hotter. Landowners have sectioned off the area around the pools and installed an overflow pipe to keep the water from seeping out and harming livestock or people.
Despite the dangers involved, one September day Joel Graham leaned over the barrier and plunged in his gloved hand to scoop up sediment and water samples.
At this locale, the biologist from the University of Maryland, Baltimore was fishing for microorganisms that can thrive at very high temperatures. Such extremophiles contain reaction-driving proteins called enzymes unlike any found in less harrowing environs.
Enzymes show up in all living things — from the simplest microbes to humans — and are essential for survival. They do jobs such as digesting food and helping to make sure genetic material gets properly copied. While a lot of known enzymes live and work best around the temperature of the human body, Graham and colleagues are among a cadre of scientists looking for new enzymes that can function when conditions get superheated.
The collective hope? That such enzymes could be co-opted to perform industrial processes. In many of the world’s factories, setting the thermostat on high keeps reactions moving quickly and prevents bacteria and other microbes from contaminating or eating up desired products. Papermaking, textile manufacturing and some food processing might all benefit from the help of heat-loving enzymes.
Graham’s team is particularly interested in a class of enzymes that help break down the grassy parts of plants, an area where researchers have made recent progress. Finding such enzymes, and employing them in large quantities, could lead to biofuel production on an industrial scale, helping society reduce its dependence on fossil fuels.
Some scientists are hoping a newly discovered enzyme pulled from the field may be an ideal high-temperature grass muncher. Other researchers, instead of getting their hands dirty in hot pools, are designing never-before-seen heat-loving enzymes in the lab. Many lab enzymes are in some way inspired by versions already found in nature.
“The environment contains a lot of solutions to a lot of human problems,” Graham says. “If you find a directed way to go after what you’re looking for, it’s actually not that hard to find something new and useful.”
Enzyme-palooza
People have been using enzymes to perform chemical reactions outside the body for millennia. Food processing with enzymes, for example, dates back at least 4,000 years, when people stored milk in the excised stomachs of animals. Digestive enzymes in an animal’s gut caused milk to curdle, forming other dairy products like curds and whey.
Today, a cocktail of various proteins help coagulate milk to make solid curds for cheese. Other enzymes soften cotton during textile manufacturing or treat paper in the pulp industry. Enzymes even lend a hand in the synthesis of drugs and supplements such as vitamin E.
But enzyme-driven processes these days can get much more involved than just throwing starting materials into an animal gut. Often materials need to undergo additional reactions, treatment with a solvent or dyeing with a chemical, before or after an enzyme can do its job. For many of these reactions, heat is required as a catalyst.
The problem: Most known enzymes work only at conditions matching those of the organism from which they came. So temperatures have to be repeatedly ramped up and lowered at various stages of a multistep process. “You have to cool it down for the biological part and heat it back up for the next step,” says Vicki Thompson of Idaho National Laboratory in Idaho Falls. “That makes the process more expensive, and it’s wasteful of energy.”
During low-temperature stages, materials can also be vulnerable to attack from a plethora of microbes.
The breakdown of long chains of glucose molecules from plants, or cellulose, faces such troubles. Cellulose stored in corn stover — the grassy part of corn that people don’t eat and a potential biofuel source — is tied up in complex chemical arrangements, making it hard for enzymes working alone to get access.
So the biomass has to be mashed up a bit first. It’s heated and treated with corrosive chemicals like acids or salty solutions, which expose the cellulose. But the plant products have to be neutralized and cooled before a collection of room-temperature enzymes can digest the material into usable sugars.
Cellulose-breaking enzymes, called cellulases, made by organisms that normally thrive under extreme conditions, in places like hot springs or hydrothermal vents, could offer a solution.
“You can take those enzymes and use them under the industrial conditions that you are interested in,” explains Thompson, who has been studying organisms that live in extreme environments and the proteins they make for more than a decade.
Pulled from a pool
Biologist Thomas Brock unearthed one of the first known extremophiles from hot pools in Yellowstone National Park in Wyoming in the 1960s. Later named Thermus aquaticus, the bacterium thrived best at temperatures around 70° C. This finding suggested life could exist in all kinds of places that were previously thought of as dead zones.
Explorations in volcanic soil, hot vents, deep seas and salt deserts have turned up thousands of extremophiles since, says Thompson. A sizable portion love the heat. Notably, a heat-loving, DNA-building enzyme from T. aquaticus has been a major boon for genetic engineering and forensics. To analyze DNA from collected samples, scientists need large amounts of uncontaminated copies. By simply adding the enzyme, Taq polymerase, to a starting strand of DNA and other genetic ingredients, scientists can make lots of DNA copies with no contamination to worry about.
Over the last two decades, Frank Robb of the University of Maryland, Baltimore has scoured some of the hottest corners of the Earth — from the hot springs at Yellowstone to the deep-sea vents of the Okinawa Trough — in search of interesting heat-loving enzymes. An ability to break down cellulose is one sought-after skill in the “help wanted” ads.
Robb and Graham want cellulases that can do their thing at temperatures of 100° C or even higher. The more types the better, since some enzymes specialize in cutting the cellulose into chunks and some break it down even further into glucose.
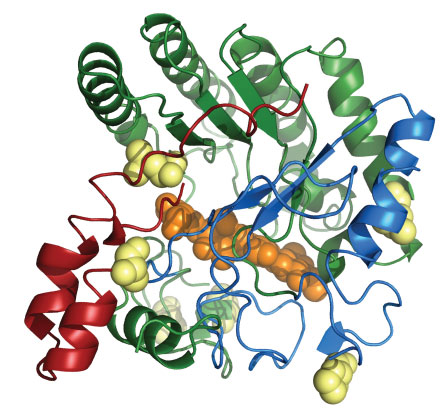
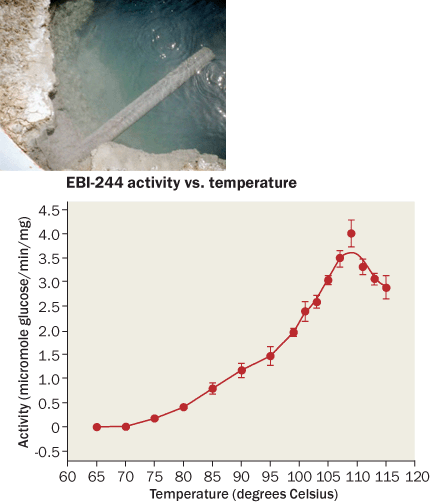
Recently, after examining genetic material from samples collected at the hot springs in Nevada, Robb and Graham turned up an enzyme called EBI-244 that can perform one of the early cellulose breakdown steps at temperatures as high as 109° C. Though other heat-loving cellulases have previously been found, none do the job as well as EBI-244 at such high temperatures.
“Nobody ever thought or had seen an enzyme that worked at those temperatures, and it’s so stable at those temperatures,” says Isaac Cann of the University of Illinois at Urbana-Champaign, who also studies cellulases and their role in biofuels production.
And the enzyme, described in Nature Communications last year, can also survive in acidic conditions and put up with harsh solvents.
Researchers can’t collect nearly enough heat-loving cellulases in the field to run a cellulose-degrading factory (kilogram amounts are needed). Instead, scientists typically hook up the genetic material that codes for a specific enzyme to a bacterium’s or yeast’s molecular machinery, tricking the organism into producing the protein. But for many enzymes, EBI-244 included, it has been a challenge.
“It’s just a painstaking and tedious process to get suitable quantities,” says Douglas Clark, a biochemical engineer at the University of California, Berkeley who collaborated with Robb and Graham in identifying EBI-244.
Despite the barriers, the discovery of EBI-244 is exciting because of what scientists can learn from it. The DNA that codes for the enzyme is unlike the DNA for any known cellulase; it was traced back to a never-before-seen extremophile. “It’s very strange to find one that doesn’t fit into a well-defined category,” Graham says. “That was the most exciting part.”
The next challenge will be to study the enzyme’s physical structure, to unlock its heat-loving secrets.
“We would love to know more about it,” says Clark. “Could we begin to use the information gained from studying this enzyme to engineer not only other cellulases, but other proteins?”
Into the lab
Rather than hunt for enzymes that can function under extreme conditions in the field, chemical engineer Frances Arnold of Caltech makes her own. She brings the principles of breeding to the lab: guiding the development of new and better proteins using known proteins as parents.
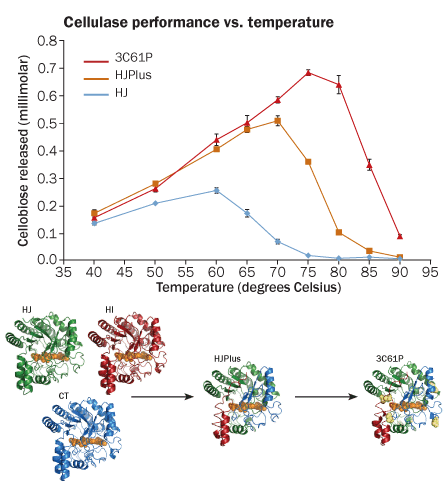
Arnold’s lab starts off by using a computer program to put together stretches of amino acids, the building blocks of proteins, from different enzymes. By mixing and matching chunks from each parent, the team’s computer software figures out what types of children would emerge if these enzymes could “breed.” The idea is to pick parents with different features — like ones that thrive in high heat, or can grow up in large quantities — to produce a child with the best of all the qualities.
Arnold and colleagues mine the results, looking for prospective children with the most promising futures — that is, with proteins likely to perform interesting functions and assemble into a desired shape in the lab. The children can then undergo small-scale changes that make them even better.
“You’re better off making it in the laboratory, because no natural enzyme will fulfill all the requirements of your desired applications,” says Arnold.
Arnold produces her computer-based cellulases with the help of simple organisms in the lab. She doesn’t try to grow huge loads of her heat-loving cellulases, but she does start with proteins from filamentous fungi, which are known to grow up easily.
After a recent cellulase “breeding,” Arnold’s team reported making new enzymes that could work at about 70° C. Some of the parent enzymes could break down cellulose only at temperatures around 57° C. Since that report, Arnold has been able to coax more proteins to work at similar temperatures.
She has since sold licenses for making these cellulose-breaking prodigies to private companies, though she doesn’t yet know whether the enzymes are being used for large-scale plant munching.
Despite the recent breakthroughs, there’s a lot of work to be done before heat-loving enzymes can usher in an era of cheap biofuels.
Individual proteins, whether unearthed from hot zones or made in the lab, will have to come together as members of a cellulose-converting team. Once many different heat-loving enzymes are found, scientists need to get them to work well as a group.
“Cellulases are a particularly hard problem because it’s not just one enzyme. It’s a whole family of enzymes. If you improve one component, it does not mean you have a better product,” Arnold says. “You have to improve many components and then find the right mixture of those components. It’s a challenging thing.”
Once the right team is found, scientists still have to overcome the hurdle of inexpensively making a lot of proteins in the right ratios. The same hurdle will probably hold for enzymes in other heat-dependent processes.
“We haven’t yet figured out all the rules for making highly stable enzymes that also do what they need to do and can be produced in an economical fashion,” Arnold says. “We’re trying.”






