Quantum dots get a second chance to shine
A new look at colorful semiconductor particles for visualizing living cells
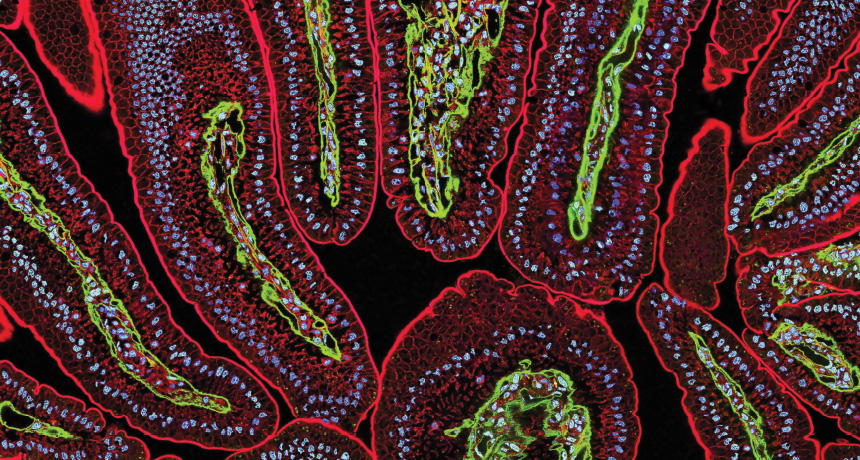
DAZZLING DOTS Tiny semiconductor particles called quantum dots light up mouse intestines in this microscope image. Red and green represent proteins illuminated by fluorescing quantum dots. Cell nuclei are stained blue with a traditional dye not involving quantum dots.
Provided by Thermo Fisher Scientific. Image by Thomas Deerinck and Mark Ellisman/National Center for Microscopy and Imaging Research
Warren Chan helped invent a research field and then watched it nearly die.
The chemist and biomedical engineer at the University of Toronto specializes in quantum dots, tiny semiconductor particles that glow in a rainbow of colors when zapped with a laser. Fifteen years ago, quantum dots were all the rage. Scientists dreamed of the wild things they could do with them (SN: 6/3/06, p. 344).
Perhaps quantum dots could glow in a futuristic, superbright television screen. Or maybe doctors could hook them to a cancer-fighting drug to watch the medicine spread through the body and attack the tumor. In 1998, Chan and Shuming Nie, then at Indiana University in Bloomington, were among the first to describe how quantum dots could light up the inner workings of living cells.
In the years since, the television screens became reality: Quantum dot displays with vibrant colors are hot items at consumer electronics shows. But researchers struggled to get quantum dots to perform safely and predictably in living cells. Chan’s dreams remained wild.
More recently, though, the barriers to quantum dot progress seem to be crumbling. Within the last few months, Chan has finally succeeded in building one of the devices from his dreams. It’s a smartphone attached to a lab-on-a-chip, which uses quantum dots to detect viruses such as HIV or hepatitis lurking in a person’s blood. It is almost as accurate as a traditional lab test, while a lot cheaper and portable. Doctors could use it outside of hospitals.
Other researchers are shrinking quantum dots, making them tiny enough to track the flow of single particles among brain cells and other microscopic systems. Some scientists are taking an apparent drawback of quantum dots — that their brightness flickers in and out like a firefly’s — and turning it into an advantage by creating incredibly detailed images of cell innards.
Quantum dots still may not launch the revolution that some biologists had hoped for, but the field is clawing its way back to relevance. The ultimate goal now is to provide a new way to illuminate how cells work, turning a series of small laboratory advances into something that helps doctors and patients better track and fight disease.
“I still believe quantum dots are very cool,” Chan says.
Minisemiconductors
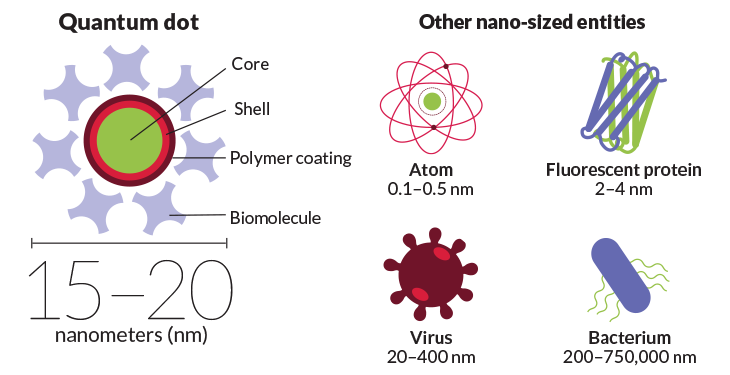
Quantum dots combine everyday chemistry with extraordinary physics. They are made of standard semiconductor materials such as cadmium selenide, but their size gives them astonishing properties. The core of a typical quantum dot measures from a few to more than 20 nanometers, or billionths of a meter, across. It is so small that its electrons behave much as they would in an individual atom, observing quantum mechanical rules (hence the “quantum” in its name). Electrons jump between individual energy states, in the process emitting photons of a particular wavelength of light.
Because they can be produced in a variety of small sizes, quantum dots can be “tuned” to emit light in whatever wavelength a scientist wants. A larger quantum dot emits red light, while a small one glows blue. By tweaking the size of the dots, researchers can coax them to sparkle in a rainbow of colors.
Quantum dots are also much brighter and last longer than fluorescent dyes, the traditional tool for staining living cells. That makes the dots good candidates for studying how drugs flow within the body and how proteins and other biological molecules go about doing their jobs over hours or even weeks.
When quantum dots first entered the scene, some experts thought they might eclipse fluorescent dyes and become the material of choice for studying living cells. There’s just one problem, though, and it’s major: Cadmium, the heart of most commercially produced quantum dots because it glows so brightly and reliably, is toxic in high doses. In the early 2000s, a series of papers showed how quantum dots made with cadmium selenide could actually kill cells in a petri dish. Those studies scared many biologists away from working with the dots, says Russ Algar, a biochemist at the University of British Columbia in Vancouver.
Algar thinks that toxicity fears have unfairly stalled the field. Studies in live animals suggest that cadmium can be used at levels that aren’t toxic to the body, he notes. And studies in cell cultures generally don’t last long enough for researchers to have to worry about whether cadmium is killing the cells. In addition, quantum dots are not all alike — many of them are packaged inside outer layers that safely contain the cadmium, or at least mitigate how much of it leaks into its environment. “This idea that quantum dots should be written off because they contain a heavy metal isn’t a fair assessment,” Algar says. “That’s potentially closing the door on some opportunities.”
Size matters
While some researchers try to work out, or work around, the toxicity issues, others have found a way to turn quantum dots’ unpredictable blinking into an asset. The flickering may have something to do with cycles of electrical charging and discharging in the dots’ cores. The blinking makes it hard to continuously track every move of a particle within a cell.
At the University of Illinois at Urbana-Champaign, biophysicist Paul Selvin has embraced the blink. He uses a technique known as super-resolution fluorescence microscopy, whose inventors shared the 2014 Nobel Prize in chemistry (SN: 11/1/14, p. 15). It relies on flickering particles to take sharper images of very small molecules than would otherwise be possible.
Normally, the wavelength of light in a microscope limits the size of the items that researchers can see; anything tinier than about half of the light’s wavelength (about 200 nanometers for visible light) simply can’t be spotted in an ordinary optical microscope. But scientists can get around this limit by taking a handful of molecules and making them glow, a few at a time, in the area they want to image. By allowing a few particles to light up, and then turning them off and letting another few particles glow, the researchers can superimpose the images and come up with a picture that is higher resolution than before.
Although super-resolution microscopy usually uses traditional fluorescent dyes, quantum dots can do the job well, Selvin says. In 2013 in Nano Letters, his team reported taking super-resolution pictures of epidermal growth factor receptors, molecules involved in the growth of breast cancer cells, using quantum dots. Understanding how the receptors work could help scientists learn more about how the cancer spreads.
More recently, Selvin has used the technique to track the step-by-step action of proteins that act as molecular motors to move other proteins around within a cell. “These quantum dots are really pretty amazing,” he says. “They are so good that you can see in all three dimensions.”
Even though quantum dots measure just nanometers across, that can still be a bit too big for tight spaces like the junctions between cells. Most commercially available quantum dots are 20 nanometers or more across. Their cores are much smaller, but a thick shell surrounds the core to safely insulate the cadmium metal from the environment. An additional polymer coating allows the dot to flow freely within a cell. These commercial quantum dots can be used for biological imaging, but they aren’t always ideal for squeezing into all parts of a cell.
Many research teams have been working to fashion thinner shells that still sequester the cadmium. Selvin, for instance, takes a core of cadmium selenide and then adds a sulfur atom and a chain of 11 carbon atoms. He coats the whole thing in a polymer to make it move easily in water. If the layers are added at high temperatures in the absence of oxygen, the coating molecules form a dense, tight layer around the core. The result: quantum dots that are only about 9 nanometers across. Selvin and collaborators made the dots glow at four different wavelengths. The glow lasted for more than a month, the team reported last year in Angewandte Chemie.
The small quantum dots are only about one-third as bright as a larger commercial dot, but that’s still bright enough for Selvin. In a lab dish he hooks them up to AMPA receptors — molecules in living nerve cells, or neurons — and tracks how they travel.
The dots are small enough to squeeze into the synaptic cleft, a space about 20 to 40 nanometers across where one neuron passes a signal to another. “The smaller dots can actually get inside,” Selvin says. So “we’re getting a very different view of how these synapses work.”
For instance, the small quantum dots showed that most of the action happens within the synapse itself, not at locations just outside the synapse. Watching the whole thing with big quantum dots had misled scientists into thinking that the area outside the synapse, which was the only place the big dots could go, was perhaps more important than it is, Selvin says.
Magnetic moves
There are times when quantum dots don’t have to be small to be helpful. MIT chemist Moungi Bawendi has used the dots to help build “supernanoparticles” with a very attractive property: They are magnetic and can be guided to a specific place in a cell by applying a magnetic field. Ordinary quantum dots and fluorescent dyes can’t do that.
Bawendi’s team coaxed cadmium-based quantum dots to cluster around magnetic iron oxide nanoparticles, like a ring of wagons encircling a cattle herd. Together they form one larger cluster about 100 nanometers across.
Because the supernanoparticle is magnetic, researchers can steer it in a particular direction by flipping on a magnetic field. Then they can watch the brightly glowing collection as it travels within a cell.
Story continues below infographic
Supernanoparticles may become a sort of Swiss army knife for biological imaging — a single package containing many handy tools, Bawendi and his colleagues reported last year in Nature Communications. One day scientists might even use them to steer drugs to a particular location within the body, he says.
Another disease-fighting quantum dot tool is being developed at Imperial College London, inspired by a baby’s rattle. Biochemist Molly Stevens and her colleagues have built a quantum dot–filled “quantum rattle” that is no toy; it seeks out and destroys cancerous tissue by heating it.
Instead of using cadmium selenide, Stevens makes her quantum dots out of gold. Tiny gold particles, less than 2 nanometers across, can absorb near-infrared light and transform it into heat. These nano-sized golden warriors penetrate diseased tissue and deliver a heated punch right where it counts.
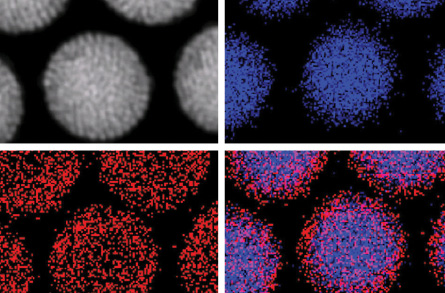
Stevens and her colleagues build the rattle using a hollow shell of silica, about 150 nanometers across. They place gold quantum dots inside along with larger particles of gold. When zapped with a laser, the quantum dots glow and give off heat.
The researchers injected the quantum rattle into tumors in mice, then blasted the tumors with a laser. Tumors containing the quantum rattle began to show signs of cells dying, unlike tumors that had been injected with hollow nanoparticles that did not contain quantum dots rattling around inside.
Quantum rattles might also help deliver cancer-fighting drugs directly to tumors, Stevens’ team reported in February in the Proceedings of the National Academy of Sciences. In lab tests, rattles loaded with doxorubicin plus quantum dots released the drug into the surroundings a third as rapidly as silica shells containing doxorubicin but no quantum dots. A slow, extended-release delivery method could make it easier for cancer patients to tolerate their drugs.
Not all of these quantum dot discoveries will make it into your doctor’s office. The supernanoparticles may be too big to flow smoothly through many types of living cells. And the quantum rattle may not work better than other approaches for ferrying drugs directly into cells. But one quantum dot application is now almost ready for prime time — and it comes from Chan in Toronto.
In March, in the journal ACS Nano, Chan’s team reported on a quantum dot bar code device that can diagnose three different viral infections. A person places a drop of DNA from blood onto a tiny chip, and a smartphone snaps a picture and can read out whether a virus is present.
The chip is coated with microscopic beads containing quantum dots. Each bead is coated with a material designed to recognize a particular strand of DNA — for instance, a sequence that is specific to a hepatitis virus. If there is virus in a blood sample, the DNA will connect to the beads designed to detect hepatitis. If there is HIV in the sample, the DNA will connect instead to the HIV beads. “It really took about 10 years to get the chemistry to work,” Chan says.

Doctors can already do this type of test in a hospital, using fluorescent dyes, but making it portable and part of a smartphone opens new options. “We want to develop a device that’s cheap and cost-effective so we could do the detection anywhere,” Chan says. His team is using 3-D printers to fashion plastic cases to hold the beaded chip, smartphone and lasers together; the whole setup costs well under $100.
Quantum dots are bright enough for a smartphone camera to pick up their glow, and with the bar coding, multiple tests can be run at the same time. The device can detect virus-positive samples about 80 to 90 percent as accurately as a traditional laboratory test, Chan says. He is working on improving that detection rate and thinks he can start selling the quantum dot bar coder soon.
Such smartphone-based tests may be the future of quantum dots, says Algar. “If we can leverage their really excellent fluorescence properties, then we can get away with instrumentation or devices that are less complex than in the lab,” he says. “Press a button on your smartphone, and if you can do some sort of useful measurements or diagnostic test that would be a great thing.”
All thanks to tiny glowing specks that may be finally coming into their own.
This article appears in the July 11, 2015, Science News with the headline, “Dazzling dots: Tiny quantum particles illuminate the body’s innermost workings.”