How strep throat may spark OCD and anxiety in some kids
In rare cases, the body’s response to the bacterial infection appears to attack the brain

When obsessive-compulsive disorder comes on suddenly in a child, some researchers wonder if a strep infection can impact the brain.
Graphicaffe/Shutterstock, adapted by T. Tibbitts
One night in December 2013, Hans Korbmacher awoke in a fury. The book-loving, introverted 10-year-old was feverish, agitated and gnawing on his tongue. He headed downstairs, leaped onto an ottoman and threw his hands over his head, startling his parents. He was “clearly not present,” says his mother, Heather Korbmacher.
When the same thing happened two weeks later, she thought fevers may have induced Hans’ bizarre behavior. A nurse said it could be the flu. Meanwhile, Hans’ condition worsened. He was anxious and volatile. His handwriting, once a model of penmanship, morphed into angry scribbles. And he became a peculiarly picky eater.
Korbmacher, a behavioral specialist for schools in Bellingham, Wash., tried to manage Hans’ symptoms on her own. “It was working OK during those first five months, until it was absolutely not,” she says. Extreme rages came weekly and then daily, keeping Hans out of school. He punched holes in walls and ripped down curtains. The worst part: Hans was acutely aware that something was very wrong. He pleaded with his parents to make it stop. “He would beg us to kill him,” Korbmacher says.
Several doctors’ appointments later, a psychiatrist suggested that Hans’ symptoms stemmed from obsessive-compulsive disorder, or OCD. The diagnosis seemed off base to Korbmacher until she read online about a rare form of OCD with a mouthful of a name: pediatric autoimmune neuropsychiatric disorder associated with streptococcal infections, or PANDAS for short. Hans had all but one of the listed symptoms.
Korbmacher immediately had Hans tested for a strep infection. A throat swab came back negative, but blood tests revealed that he had four times the typical levels of immune molecules that the body produces in response to a strep infection.
PANDAS is as puzzling for researchers as it is for the families that struggle with it. Scientists studying the condition propose that the string of psychiatric symptoms is triggered by infection with Group A Streptococcus, one of the most common childhood illnesses (SN: 3/2/19, p. 7). In some children, a faulty immune response to the infection may mistakenly attack brain cells, causing behavior to go haywire.
But researchers have yet to nail down which brain cells are harmed. And it’s not clear why countless children get strep, yet so few develop PANDAS symptoms. With so much uncertainty about how strep could affect the brain, some experts doubt whether PANDAS is a separate disorder from OCD and whether it’s truly triggered by strep. Amid the debate, families searching for help get mixed messages.

Researchers have begun to fill in some gaps in understanding, and some say the field is on the cusp of explaining the biology underlying PANDAS. “We’re at a point where the basic science research is just progressing at lightning speed,” says Susan Swedo, the developmental pediatrician who coined the term PANDAS. But other researchers are much more reserved, saying a heap of work still remains.
Support for a connection between the body’s reaction to an infection and changes in mental health comes from recent studies linking infections and inflammation with several more common psychiatric disorders, including schizophrenia, depression, Alzheimer’s disease and autism. As scientists delve deeper into these intriguing links, the hope is that new ways to detect and treat these conditions will come into focus.
A questioned discovery
Swedo described PANDAS in the 1990s while studying a childhood condition called Sydenham chorea. The rare disorder can occur with rheumatic fever, the heart condition that can develop when a Group A strep infection goes untreated. Patients with Sydenham chorea uncontrollably jerk their limbs and bodies.
Swedo found reports suggesting that some patients with Sydenham chorea also have psychiatric problems. “Those children were noted to have a lot of obsessional fears about contamination, about harm coming to their parents,” says Swedo, a scientist emeritus at the National Institutes of Health in Bethesda, Md., and chief science officer of the PANDAS Physicians Network. The children “had separation anxiety, emotional [instability] and even frank psychotic symptoms.”
Swedo and colleagues found that some children with Sydenham chorea displayed obsessive symptoms that came on suddenly. The researchers observed a similar abrupt onset of symptoms in some children with OCD. In many of these children, surges in obsessive behaviors followed strep infections.
After evaluating a separate group of 109 children with sudden-onset OCD, Swedo’s team determined that 50 of the youngsters represented a distinct subgroup whose symptoms appeared out of the blue and then waxed and waned over time, seemingly in step with strep infections. The researchers published their findings in 1998 in the American Journal of Psychiatry.
To date, a PANDAS diagnosis is defined solely by its symptoms. There is no definitive lab test. (One company markets a PANDAS blood test, but evidence suggests it doesn’t actually work.) Swedo says it’s possible that as many as 1 in 400 children in the United States have the condition. But strep expert Ellen Wald, who also thinks that PANDAS is a distinct condition, disagrees with that estimate. “My guess is that it’s quite a bit lower than that,” says Wald, a pediatrician at the University of Wisconsin School of Medicine and Public Health in Madison.
Some experts aren’t convinced that PANDAS is even real. After Swedo first described PANDAS, skeptical clinicians aired concerns in medical journals, questioning whether the evidence supports calling PANDAS a condition. With missives lobbed back and forth between the skeptics and PANDAS proponents as recently as July, little has been resolved.
Jonathan Mink, a pediatric neurologist at the University of Rochester Medical Center in New York, is a skeptic. He says he sees around 25 patients with suspected PANDAS each year but has yet to come across a case that checks off all the criteria for the condition. Mink argues that these patients most likely have traditional OCD or Tourette syndrome, a tic disorder that involves compulsive movements or outbursts.
But parents are often disappointed with diagnoses without a clear-cut cause, Mink says. PANDAS offers a straightforward trigger. “Everybody knows about strep infections,” he says.
The link between strep and PANDAS is tenuous at best, say Mink and pediatric neurologists Donald L. Gilbert of Cincinnati Children’s Hospital and Harvey Singer of Johns Hopkins School of Medicine in a commentary in the August 2018 Journal of Pediatrics. The research that came after Swedo’s initial reports is a mixed bag; some studies find that strep occurs just as often in children diagnosed with OCD or Tourette syndrome as it does in children meeting PANDAS criteria.
These conflicting results led Swedo and other PANDAS researchers in 2012 to propose a broader condition called pediatric acute-onset neuropsychiatric syndrome, or PANS. The new diagnosis describes children who suddenly develop dramatic obsessive symptoms and other psychiatric signs with or without a preceding infection.
Wald says it may never be possible to prove without question that PANDAS stems from strep. The time between infection and the onset of psychiatric symptoms may vary from weeks to months. By then, standard strep throat swabs would come back negative. But that doesn’t negate the possibility that a strep connection, or that PANDAS itself, exists, she says. “It’s sort of time for us to say there are many unknowns here, but let’s try to keep an open mind and really try to learn about how common it is and how [to] approach it.”
Rogue immunity
Researchers studying PANDAS are focusing on wayward immune pathways that may underlie the condition. When foreign particles such as strep bacteria invade, the body unleashes proteins called antibodies to target and neutralize the threat. But sometimes this defense system turns on its host, resulting in autoimmune conditions, such as multiple sclerosis and lupus.
Since strep infections are so common, most children may have anti-strep antibodies circulating in their bloodstream from time to time. But in the case of PANDAS, these antibodies are thought to attack the brain.
In particular, some studies suggest that the rogue antibodies target a cluster of brain structures called the basal ganglia that governs motor control and emotions and has been implicated in OCD. A lab study reported in 2006 showed that antibodies from the blood of PANDAS patients latch onto tissue collected from human basal ganglia. And in a brain-imaging study of 17 PANDAS patients published in 2015 in the Journal of Child Neurology that same region showed signs of inflammation. Similar inflammation was seen in a small study in adults with traditional OCD reported in 2017 in JAMA Psychiatry.
The connection to antibodies also comes from rats exposed to strep. The animals showed deposits of antibodies in the basal ganglia as well as spikes in the brain chemical dopamine, researchers reported in 2012 in Neuropsychopharmacology. The exposed rats also exhibited behavioral quirks that mimic PANDAS symptoms, such as faulty motor control and compulsive grooming. The findings suggest that antibodies binding to this region disrupt brain signaling and may change behavior, too.
But for Mink and other skeptics, the existing data are still too shaky to mark PANDAS as a separate condition with autoimmune roots. A big problem is that studies searching for specific cells targeted within the basal ganglia and elsewhere in the brain have yielded inconsistent results. “That’s where I think the evidence is so weak that I find that there’s little to really support this as a hypothesis,” Mink says.
By contrast, researchers studying an autoimmune condition called anti-NMDA receptor encephalitis, which also spurs psychiatric symptoms, can point to a protein on certain cells in the brain where rogue antibodies bind. In this condition, antibodies attack receptors for the brain chemical glutamate. The attack brings on an array of symptoms including seizures and hallucinations.
The latest PANDAS data, however, offer a new cellular suspect. In a study reported in Brain, Behavior and Immunity in 2018, researchers looked for antibody binding in a part of the basal ganglia called the striatum. The team focused on cells called cholinergic interneurons. Other work has shown that those cells are depleted in the brain in patients with Tourette syndrome.
Infusing blood serum from five children meeting PANDAS criteria into the striatum of mouse brains, the researchers found that antibodies known to respond to strep attached to about 80 percent of cholinergic interneurons. In mouse brains given serum from children with no PANDAS symptoms, the same antibodies latched onto less than half of the cells. Preliminary work from the same laboratory suggests that cholinergic interneurons fire less when tagged by these antibodies.
Taken together, the findings may explain what anti-strep antibodies do in the brain, says lead researcher Chris Pittenger, a psychiatrist and neurobiologist at the Yale School of Medicine. But the results also raise questions: Are the findings biologically meaningful if antibodies from children without PANDAS also attached to a sizable portion of the striatal interneurons?
Pittenger doesn’t expect these findings to apply to all children with PANDAS. The condition may comprise several subtypes with distinct profiles of antibodies and neural targets. “I would call it one small step in what I hope will be a clarification of what’s going on in PANDAS and which kids actually have what pathology,” he says. Another step, Pittenger says, is to see if serum from children with traditional OCD or Tourette’s also binds excessively to cholinergic interneurons.
Breached barrier
Another team of researchers wants to know how antibodies circulating in the blood cross the blood-brain barrier to reach the brain. That border of tightly connected blood vessels typically wards off invading molecules.
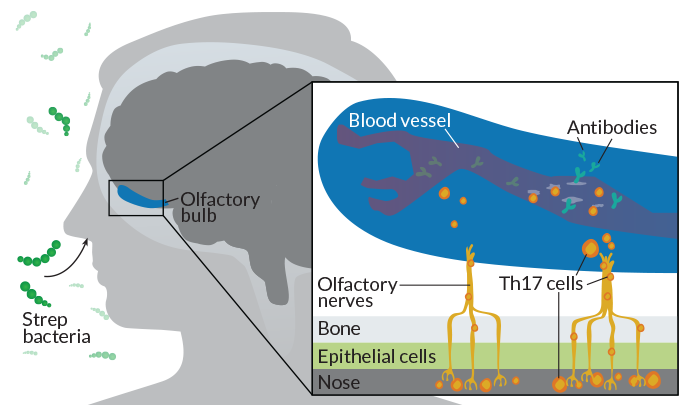
Exploring one possible pathway into the brain, neuroscientist Dritan Agalliu and colleagues at Columbia University Irving Medical Center infected the nasal passages of mice with strep bacteria and tracked the movements of a type of immune responder, T cells called Th17 cells.
“These T cells are sort of these double-edged sword cells,” Agalliu says. Like antibodies, Th17 cells help fight off infections, but can sometimes bring harm to healthy cells. In particular, Th17 cells release inflammatory molecules that can poke holes in the blood-brain barrier, which researchers have seen in multiple sclerosis.
After several rounds of strep, Th17 cells originating in the mouse equivalent of tonsils accumulated in the animals’ brains, Agalliu’s team reported in 2016 in the Journal of Clinical Investigation. Most of the T cells pooled in the olfactory bulb, a brain region that receives odor signals from nerve cells, or neurons, extending from the nasal passages. Th17 cells responding to strep may travel along this neuronal highway to infiltrate the brain, Agalliu says.
The researchers also found a leaky blood-brain barrier and deposits of antibodies in the olfactory bulb, suggesting that the Th17 cells could carve a path for the antibodies to enter the brain. To confirm this idea, the researchers repeated their experiment in mice genetically engineered to lack Th17 cells. The preliminary results show that antibodies can’t access the brain in these mice. Th17 cells may be key to allowing antibodies into the brain.
Agalliu’s work also hints at why so few children with strep show signs of PANDAS. Successive strep infections may be partly responsible, Agalliu says. His team found that Th17 cells opened a pathway to the brain only after mice were infected with strep at least three times.
Another likely contributor is a genetic predisposition (SN: 9/2/00, p. 151). In their latest unpublished work, Agalliu and colleagues have identified several genetic variants that stand out in children with PANDAS symptoms. The affected genes all regulate parts of the immune system, such as the ability to dampen an immune reaction. In children with PANDAS, Agalliu says, “the immune response seems to be persistent for a longer period of time than what we normally see.”
Seeking treatment
As researchers try to untangle the biological roots of PANDAS, families faced with alarming symptoms grapple with uncertainty about how to help their children. Clinicians still disagree on the right treatment approach, and only a few options have been studied in rigorous trials. Proposed treatments are backed mostly by case reports and small trials, many of which lacked a placebo comparison group.
Many PANDAS experts tout the use of antibiotics to treat the strep infections that may underlie the condition. In her own experience, Swedo has found that starting antibiotics at the first signs of PANDAS is enough to reverse the condition altogether in patients whose strep infections are still active but maybe hadn’t been detected and treated.
“It’s really remarkable how much of a difference prompt treatment of that infection can be,” she says. Some clinicians even recommend extended courses of antibiotics to fend off future infections. But there’s no compelling evidence that long-term antibiotics are warranted, Mink says. Strep infections tend to respond well to a single round of antibiotics, and unnecessary use could bring on side effects such as severe diarrhea. Chronic use also invites antibiotic resistance. “It’s a public health concern as well as an individual health concern,” he says.

For particularly severe cases, clinicians may turn to a more invasive treatment aimed at resetting a malfunctioning immune system. The procedure, intravenous immunoglobulin, or IVIG, is an infusion of antibodies stripped from the blood of thousands of healthy donors. The goal is for the new antibodies to bind to and disable the patient’s harmful ones. IVIG is also used to treat Guillain-Barré syndrome and other autoimmune conditions.
This was the approach Hans Korbmacher’s doctors took after seven months of antibiotics did little to improve the boy’s condition. In October 2015, nearly two years after his initial symptoms emerged, Hans received IVIG. Heather Korbmacher started noticing changes in her son about two weeks later.
One morning, she found Hans sitting peacefully at the dining room table eating breakfast. He was wearing shorts and a T-shirt instead of pajamas, the only clothes his sensitive skin could stand at the height of his illness. As Korbmacher uttered a passing “I love you” to her son. Hans said he loved her too — something she hadn’t heard in over a year. “I just stood there and cried because I knew that my son was back,” she says.
Hans improved gradually over the next six months. He is now 15 and fully recovered, says Korbmacher, who wrote a book about her family’s experience. “He is healthy in his body and healthy in his mind and able to access his life again.”

IVIG was a game changer for Hans, according to his mother. But the procedure is not without risks. Patients receiving IVIG may have allergic reactions and headaches severe enough to require hospitalization. And it’s costly. Korbmacher was stuck with an estimated $17,000 out-of-pocket cost until she convinced her insurance company to cover the procedure.
The scientific evidence backing IVIG to treat PANDAS is also unsettled. A 1999 clinical trial in 29 PANDAS patients reported in the Lancet found that the therapy suppressed obsessive-compulsive behaviors and anxiety. But a more recent trial in 35 children, published in 2016 in the Journal of the American Academy of Child & Adolescent Psychiatry, failed to show that IVIG performed any better than a placebo.
Swedo, who led both trials, says crucial design flaws stunted the later study. For example, any patient whose symptoms did not noticeably improve could receive an extra infusion of IVIG at the end of the trial. Researchers at the University of Arizona are planning a new trial with a more rigorous design to address these disparate results.
Some proposed PANDAS treatments are less controversial. PANDAS advocates and skeptics alike support the use of cognitive behavioral therapy and neuropsychiatric drugs such as selective serotonin reuptake inhibitors, which have been shown to be effective in treating childhood OCD. However, these treatments haven’t been tested in controlled trials with PANDAS patients.
Moving forward, it’s most important to get clearer data in the hands of clinicians who are seeing patients with suspected PANDAS, Mink says. For example, the field still lacks a natural history study to follow children who meet PANDAS criteria without any type of intervention. Though it would be hard to convince families to simply watch and wait, this approach would help determine whether a distinct PANDAS population truly exists, and what defines it, before treatments are tested.
Mink, who gets hate mail when he shares his skepticism publicly, says he’s open to shifting his stance if compelling findings arise. “There are reasonable people that have differing opinions here,” he says. “But what we really need to resolve those opinions is not going to be who speaks the loudest or who has the most advocacy groups on their side. It’s really going to come down to what’s the evidence.”