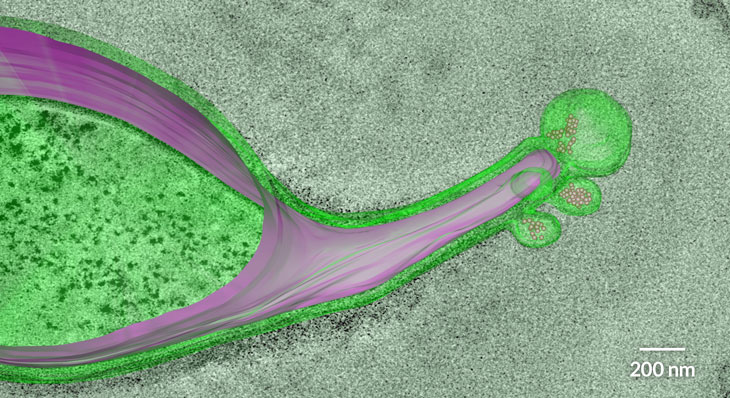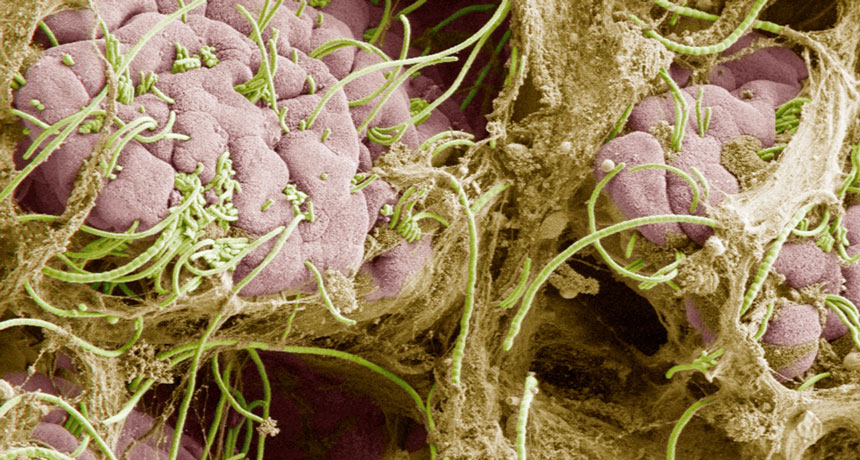
MICROBE MYSTERY Segmented filamentous bacteria (green) hook into the gut walls (pink) of many animals, including mice, birds and humans. Why the immune system doesn’t attack these bacteria, as well as other beneficial gut microbes, has been a mystery.
I.I. Ivanov and D.R. Littman/Mucosal immunology 2010