Bird flu can jump to mammals. Should we worry?
Wild bears, skunks and sea lions are just some of the mammals infected with H5N1

A deadly version of avian influenza is behind an outbreak in wild birds, including pelicans in Lima, Peru (shown) and domestic poultry. The virus has also been detected in mammals, although there is no evidence of widespread transmission in animals other than birds.
ERNESTO BENAVIDES/AFP via Getty Images
An uncomfortable truth is that there is another influenza pandemic in humankind’s future. Whether it will be a relative of the lethal avian flu strain currently wreaking havoc in bird populations around the globe is anyone’s guess.
Because the virus, called H5N1, can be deadly to birds, mammals and people, researchers closely monitor reports of new cases. Worryingly, a new variant of H5N1 that emerged in 2020 has not only spread farther than ever before among birds, but has also spilled over into other animals, raising the specter of a human outbreak (SN: 12/12/22).
The variant was linked to a seal die-off in Maine last summer. In October, there was an H5N1 outbreak on a mink farm in Spain, researchers reported in January in Eurosurveillance. (It’s unclear how the mink were exposed, but the animals were fed poultry by-products.) Sea lions off the coast of Peru and wild bears, foxes and skunks, which prey upon or scavenge birds, in the United States and Europe have also tested positive for the virus.
Globally, hundreds of millions of domestic poultry have been culled or died from the new variant. It’s also likely that millions of wild birds have died, though few governmental agencies are counting, says Michelle Wille a viral ecologist at the University of Sydney who studies avian influenza. “This virus is catastrophic for bird populations.”
A handful of human cases have also been reported, though there’s no evidence that the virus is spreading among people. Of seven cases, six people recovered and one person from China died. In February, health officials in China reported an eighth case in a woman whose current condition is unknown.
What’s more, four of the reported human cases — including a U.S. case from Colorado and two workers linked to the Spanish mink farm — were in people who didn’t have any respiratory symptoms. That leaves open the possibility that those people were not truly infected. Instead, tests may have picked up viral contamination, say in the nose, that the people breathed in while handling infected birds.
The impossibility of predicting which avian influenza viruses might make the jump to people and spark an outbreak is in part related to knowledge gaps. These bird pathogens don’t typically easily infect or circulate among mammals including humans. And scientists don’t have a full grasp on how these viruses might need to change for human transmission to occur.
For now, it’s encouraging that so few people have gotten infected amid such a large outbreak among birds and other animals, says Marie Culhane, a food animal veterinarian at the University of Minnesota in St. Paul. Still, experts around the globe are diligently watching for any signs the virus may be evolving to spread more easily between people.
The good news is that flu drugs and vaccines that work against the virus already exist, Wille says. Compared with where the world was when the coronavirus behind the COVID-19 pandemic came on the scene, “we are already ahead of the game.”
How the virus would need to change to spread among people is a big unknown
This new iteration of bird flu is what’s called a highly pathogenic avian influenza, one that is particularly lethal for both domestic and wild birds. Aquatic birds such as ducks naturally carry avian flus with no or minor signs of infection. But when influenza viruses shuffle between poultry and waterfowl, variants with changes that make them lethal to birds can emerge and spread.
Avian viruses can be severe or even deadly for people. Since 2003, there have been 873 human cases of H5N1 infections reported to the World Health Organization. A little less than half of those people died. In February, an 11-year-old girl in Cambodia died after she developed severe pneumonia from an avian flu virus, the country’s first reported infection since 2014. Her father was also infected with the virus — a different variant than the one behind the widespread outbreak in birds —though he has not developed symptoms. It’s unknown how the two people were exposed.
Some of what scientists know about H5N1’s pandemic potential comes from controversial research on ferrets done more than a decade ago (SN: 6/21/13). Experiments showed that some changes to proteins that help the virus break into cells and make more copies of itself could help the virus travel through the air to infect ferrets, a common laboratory stand-in for humans in influenza research.
While researchers know these mutations are important in lab settings, it’s still unclear how crucial those changes are in the real world, says Jonathan Runstadler, a disease ecologist and virologist at Tufts University’s Cummings School of Veterinary Medicine in North Grafton, Mass.
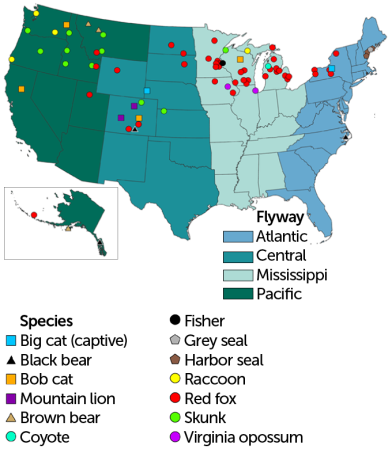
Infected mammals
A variety of mammal species have tested positive for avian influenza across the United States. Each symbol represents the counties that have reported cases. States colors indicate the routes birds take, called flyways, as they migrate south for the winter or north for the summer.
Detections of the new H5N1 bird flu variant in U.S. mammals, 2022–2023
Viruses change constantly, but not all genetic tweaks work together. A change may help one version of the virus transmit better, while also hurting another variant and making it less likely to spread.
“We’re not sure how critical or how big a difference or how much to worry about those mutations when they happen in the wild,” Runstadler says. “Or when they happen five years down the road when there are other changes in the virus’s genetic background that are impacting those [original] mutations.”
That doesn’t stop researchers from trying to pinpoint specific changes. Runstadler and his team look for viruses in nature that have jumped into new animals and work backward to figure out which mutations were crucial. And virologist Louise Moncla says her lab is trying to develop ways to scan entire genetic blueprints of viruses from past outbreaks to look for signatures of a virus that can jump between different animal species.
“There’s a ton that we don’t know about avian influenza viruses and host switching,” says Moncla, of the University of Pennsylvania.
Genetic analyses of H5N1 circulating on the mink farm in Spain, for instance, revealed a change known to help the virus infect mice and mammalian cells grown in the lab. Such a change could make it easier for the virus to spread among mammals, including people. There could have been mink-to-mink transmission on the farm, the researchers concluded, but it remains unclear how much of a role that specific mutation played in the outbreak.
It’s a numbers game for when influenza viruses with the ability to transmit among mammals might make the jump from birds, Runstadler says. “The more chances you give the virus to spill over and adapt, the higher the risk will be that one of those adaptations will be effective [at helping the virus spread among other animals] or take root and be a real problem.”
The ongoing outbreak is still a big problem for birds
Irrespective of our inability to forecast human’s future with H5N1, it’s clear that many species of birds — and some other animals that eat them — are dying now. And more species of birds are dying in this outbreak than previous ones, Culhane and Wille say.
“We have seen huge outbreaks in raptors and seabirds, which were never really affected before,” Wille says. It’s possible that genetic changes have helped the virus to spread more efficiently among birds than previous versions of H5N1, but that’s unknown. “There are a number of studies underway to try and figure it out,” Wille says.

Historically, these deadly avian flus have not been a persistent problem in the Americas, Moncla says. Sporadic outbreaks of H5N1 variants are typically limited to places such as parts of Asia, where the virus has circulated in birds since its emergence in the late 1990s, and northern Africa.
North America’s last big avian flu outbreak was in 2015, when experts detected more than 200 cases of a different bird flu virus in commercial and backyard poultry across the United States. The poultry industry culled more than 45 million birds to stop that virus’s spread, Culhane says. “But it didn’t go away from the rest of the world.”
The latest version of H5N1 arrived on North American shores from Europe in late 2021, first popping up in Canada in Newfoundland and Labrador. From there, it spread south into the United States, where so far tens of millions of domestic poultry have been culled to prevent transmission on farms where the virus has been detected. By December 2022, the virus had made it to South America. In Peru, tens of thousands of pelicans and more than 700 sea lions have died since mid-January.
It’s important to understand exactly how nonbird animals are getting exposed, Culhane says. Highly pathogenic avian influenzas infect every organ of a bird’s body. So, a fox chowing down on an infected bird is exposing its own mouth, nose and stomach to a lot of virus as it eats its meal.
For now, experts are keeping an eye on infected animals to raise the alarm early if H5N1 starts transmitting among mammals.
“I do think that the mink outbreak, and then the sea lion outbreak, is a wake-up call,” Moncla says. “We should be doing our very best to implement all the science we can to try and understand what’s happening with these viruses so that if the situation does change, we are better prepared.”







