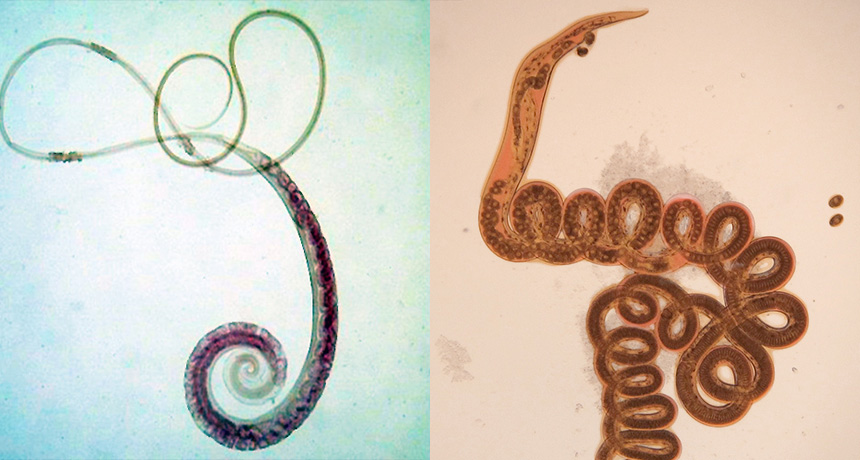
THE WORM TURNS Intestinal parasites may have an upside. Researchers have discovered that people with whipworms (Trichuris trichiura, left) and mice with Heligmosomoides polygyrus (right) have fewer inflammation-provoking bacteria than they do without the worms.
AJ Cann/Flickr (CC BY-SA 2.0); D. Davesne/Wikimedia Commons (CC BY-SA 3.0)
Parasitic worms may hold the secret to soothing inflamed bowels.