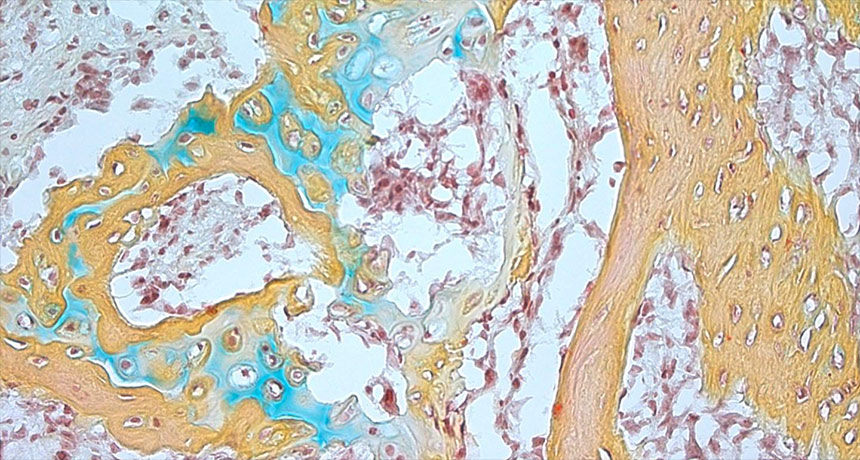
BONE TO PICK When transplanted into a mouse, human skeletal stem cells produced bone (yellow), cartilage (blue) and spongy bone (red).
Chan and Longaker et al.

BONE TO PICK When transplanted into a mouse, human skeletal stem cells produced bone (yellow), cartilage (blue) and spongy bone (red).
Chan and Longaker et al.