A matter of solidity
A material that oozes through itself presents a super physics puzzle
Solids are supposed to be the reliable state of matter.

Gases are flighty and flitting and expand to fill any available space. Liquids will also mold themselves to whatever shape they occupy, from soda bottle to swimming pool. Solids, though, are steadfast and unyielding, stable and dependable, like the rise and fall of tides, the guidance of the North Star or the love of a dog.
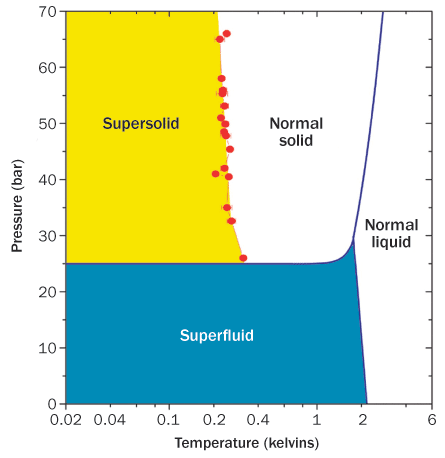
But that truism may turn out not to be so true. In the past few years, physicists have learned of a solid that doesn’t adhere to fixed rules. Within this solid — helium at very low temperatures — some atoms appear to leave their rigidly defined positions and begin moving through the rest of the material without friction. In essence, the atoms are simultaneously solid and fluid, and the material slides through itself.
Star Trek captains might be familiar with such shape-shifting, but researchers on Earth aren’t. Ever since the phenomenon, dubbed “supersolidity,” was described, physicists have struggled to explain what might be going on within the mutable material.
“Most people probably thought this would be crystal clear in 18 months,” says John Beamish, a physicist at the University of Alberta in Edmonton, Canada. “It’s a surprise that five years later we’re still not sure — which is telling you it’s more complicated than we’d thought.”
Some studies suggest that observations once attributed to supersolidity might instead result from materials behaving bizarrely for other reasons. Recent work, however, seems to strengthen the case for supersolidity. While theorists move forward with explanations for how flaws within crystals could give rise to supersolid behavior, experimentalists are building new laboratory devices to try to pin down what is really going on with this superpuzzle.
Excitement in the field runs high because of what’s at stake. Not only is supersolidity an entirely new form of material behavior, but it’s also Nobel Prize territory. The study of superfluidity, supersolidity’s older cousin, has netted a series of Nobels.
From one super state to another
Superfluidity was discovered in 1937, when researchers in England and the Soviet Union independently found that, below temperatures of 2 degrees above absolute zero, liquid helium could flow without any friction. Intrigued, theorists suggested that the atoms had morphed into a new type of quantum state — a “Bose-Einstein condensate,” in which atoms lose their individual identities and begin to flow as a collective mass.
Superfluidity has an analog in electricity. A superconducting material is one in which electrons carry electricity without any resistance. Superconductivity also occurs at low temperatures, and also because the electrons stop behaving normally and instead pair up in a Bose-Einstein condensate of their own.
By the middle of the last century, then, scientists knew that both fluids and electrons could flow without resistance thanks to quantum effects. Why not solids, too?
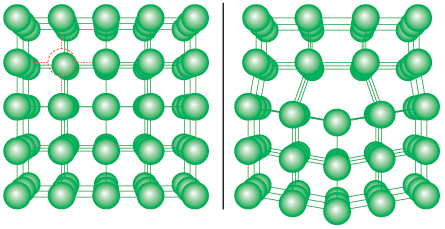
On the face of it, supersolidity seems counterintuitive, since atoms in a solid are arranged in a rigid crystal lattice. But almost all solids have empty spaces — called vacancies — where atoms are missing, like an apartment complex in which some rooms don’t have tenants. In 1969, Russian theorists proposed that quantum effects at low temperatures could cause some of these vacancies to hop from one place to another, so that different apartments become empty at different times. The moving vacancies could start to display Bose-Einstein–like behavior, and part of the solid could begin to move without friction.
If supersolidity exists, scientists thought, they should look for it in solid helium. With just two protons in its nucleus, a helium atom is relatively light. It is also not strongly attracted to other helium atoms. So if vacancies could shift around within a crystal lattice, they would do so most easily in helium.
Yet it took more than three decades for experimentalists to discover supersolidity in helium-4, which has two neutrons along with the two protons in its nucleus. The breakthrough came after John Goodkind of the University of California, San Diego reported that sound waves traveled in an unexpected manner through solid helium. Intrigued, Moses Chan of Pennsylvania State University in University Park decided to launch a new study of solid helium using a device called a torsional oscillator. This machine oscillates a sample back and forth around a central axis, like a merry-go-round spinning first one way and then the other at 1,000 times a second.
Such oscillators had been used to hunt for superfluidity because quantum materials, when placed in a spinning container, don’t spin along. Imagine rotating a bucket of water. If the water were a superfluid, it wouldn’t slosh around with the rotation but would instead sit unmoving, decoupled from the bucket’s moving sides. Supersolids, if they exist, would do the same thing. Thus, as a solid transitions to a supersolid state at low temperatures, the period of time it takes for the oscillator to rotate back and forth would drop — because less mass would be sloshing around.
Chan and his colleague Eun-Seong Kim didn’t think they had much chance of finding supersolidity. “I remember telling Eun-Seong that our chances of seeing something were close to zero — like buying a lottery ticket,” Chan says.
Yet when they filled their torsional oscillator with solid helium and spun the machine, they saw its period get shorter — presumably because some of the solid helium was becoming decoupled from the system instead of rotating with it. And when they put solid helium in a spinning device the shape of a doughnut, then blocked a portion of the doughnut before oscillating it, the effect mostly went away — suggesting that flow was indeed occurring within the helium and could be cut off at will.
That discovery, reported in 2004, touched off a rush of excitement among physicists, who thought they had finally seen the long-predicted supersolidity (SN: 1/17/04, p. 35). But the observations that followed made it far from crystal clear.
Super or not so super
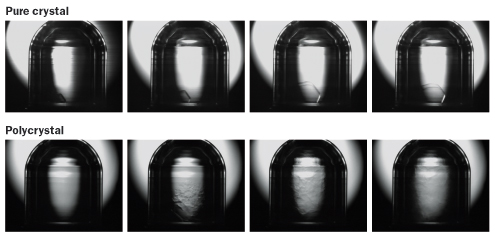
For one thing, researchers struggled with understanding what role defects played in supersolidity. In 2007, a team led by physicist John Reppy of Cornell University reported heating helium crystals long enough to heal many of the defects within. These cleaner crystals, when put into a torsional oscillator, changed the rotational period of the device far less than would be expected if supersolidity were happening. Scientists were puzzled.
That same year, doing work that didn’t use a torsional oscillator, Beamish’s group at Alberta found that at temperatures where supersolidity ought to be starting, the material stiffened up — the opposite of what might be expected for a material that could supposedly flow through itself. And helium stiffening could also explain the periodicity drop in the torsional oscillators.
“The first thing people thought of was, is all the stuff you see in the torsional oscillator just an experimental artifact?” says Beamish. If so, solid helium’s odd behavior at low temperatures could be explained by properties less exciting than supersolidity.
Yet other measurements soon showed that supersolidity and helium stiffening could both contribute to the drop in the oscillator’s period. “It seems reasonable to say there are two effects going on,” says Chan. And the contribution from supersolidity appeared to be much larger than that from stiffening.
Once again, supersolidity appeared to be on a solid footing. And once again, another challenge arose. This June, Reppy published a paper in Physical Review Letters arguing that the changes in rotational periods seen in torsional oscillator experiments are not supersolidity per se, but something else.
In this work, Reppy has for the first time squished solid helium plastically — that is, squeezed it at high pressure so that its crystal lattice begins to deform permanently, like a piece of taffy. Again, the period of the oscillator gets shorter as expected, but only at relatively high temperatures — above 100 millikelvins, or thousandths of a degree above absolute zero. At the lower temperatures at which supersolidity should be occurring, he sees little to no change in the oscillator period.
Instead of displaying supersolidity at low temperatures, Reppy writes, the helium crystals might instead be undergoing some kind of permanent deformation at higher temperatures. Such an explanation would be less sexy than supersolidity, though it would mean scientists had identified a previously unknown way that solid helium can behave.
Many in the field say Reppy’s work is provocative but does not disprove supersolidity. “The rumor of the demise of the subject of supersolidity is highly exaggerated,” Chan says. In most experiments, he says, the effect of helium decoupling from the oscillator’s sides accounts for 80 to 95 percent of the drop in period observed. In Reppy’s experiment, though, the permanent-deformation effect seems to dominate, for reasons that remain unclear.
Spotting hopping dislocations
Even as researchers work to explain Reppy’s findings in the context of supersolidity, other results are pouring in that may firm up its existence once and for all.
At the University of Massachusetts Amherst, for instance, physicist Robert Hallock has set up one of the few experimental devices that does not rely on torsional oscillators. He injects superfluid helium into a rod made of a porous glasslike substance called Vycor, which enters a cell filled with solid helium that has another Vycor rod on its far end. Hallock then watches to see if the second rod gains mass, possibly because atoms are flowing from one rod to the other. If superfluid helium enters one side and comes out the other, it could indicate that the solid helium in between is behaving as a supersolid.
In a paper under review, Hallock and graduate student Michael Ray report seeing atoms flowing between the rods at temperatures where supersolidity could be happening — 80 millikelvins. “You could interpret our experiments as that we are seeing flow associated with what might be expected for supersolid behavior,” Hallock says.
Still, he adds, “I’m much more cautious than that.” In part that’s because of what happens when the researchers lower the temperature below 80 millikelvins: The rate of flow drops dramatically, the opposite of what might be naïvely expected if supersolidity were occurring. And the flow occurs at much higher temperatures as well, up to 600 millikelvins. “Something is necessary to explain the ability of our experiment to pass atoms through the cell,” says Hallock. “Is it a supersolid? Could be. Is it for certain? I don’t know.”
Some theorists think that Hallock’s work is the closest to demonstrating true supersolidity. Anatoly Kuklov, a theoretical physicist at the City University of New York’s College of Staten Island, says the experiment shows that supersolidity exists, but probably not the kind originally envisioned in 1969. Instead of a simple shifting around of vacancies within a crystal, he says, supersolidity could occur because of atoms moving along a different kind of crystal defect, called a dislocation. Such defects, known for decades, could offer a new explanation for supersolidity’s bizarre behavior.
To picture a dislocation, imagine a book in which one page has been ripped in half horizontally. Look at the closed book from the bottom, and all pages might appear to be intact. But look at it from the top, and one page will appear to be missing. The line of that missing page is analogous to a crystal dislocation.
Numerical simulations of a helium crystal with no such dislocations showed no signs of supersolidity, Kuklov says. And experimental results from different laboratories vary widely depending on how the crystals are prepared — again suggesting that the purity of the crystals could be important. “So the effect is most likely produced,” Kuklov says, “by a network of defects which can conduct flow,” in which atoms move along the defects and allow the solid to flow like a fluid.
To test this idea, experiments are underway to see what happens to a single perfect crystal of helium, free of defects, in a torsional oscillator. Chan and Sébastien Balibar, of the École Normale Supérieure in Paris, have built a contraption where the oscillator’s sides are made of transparent sapphire, providing a window to see what’s going on inside. (Other torsional oscillators use metallic containers that obscure the view within.) Inside the researchers put the purest crystal they can make. As it oscillates, new dislocations pop into existence.
At a supersolidity workshop in Paris in late July, Balibar and Chan reported seeing single dislocations move “like violin strings” through the solid helium at rates of up to several meters per second. Such speed is impossible with everyday materials and could occur only if quantum phenomena like supersolidity were in play, Balibar says.
In a paper to appear in Physical Review Letters, Balibar and colleagues also report that these vibrating dislocations can cause ultrapure crystals of solid helium to soften. Oddly, supersolidity appears only when impurities in the crystal prevent the dislocations from moving.
Additional new evidence supporting supersolidity was reported at the Paris meeting by Kim — now at the South Korean university KAIST — and his colleague Kimitoshi Kono of the RIKEN research institute in Tokyo. They took a torsional oscillator and, as it swung back and forth, also rotated it around its axis like a spinning Earth. “For almost any classical metallurgical explanation, one that doesn’t involve superfluidity, it’s hard to imagine how rotating it would make any difference,” says Beamish. “It’s a fundamental property of superfluids that rotation makes a huge difference.” And that’s exactly what Kim reported at the conference — that mass was flowing through itself not only from the back and forth of the oscillator but also because of the added rotation.
Many say that though supersolidity hasn’t been proved beyond a shadow of a doubt, it is looking more and more likely. Clearly, solid helium at low temperatures behaves in a funky way that needs explaining — whether through supersolidity or some other phenomenon. “I think over the next year we’re going to see considerable progress,” Hallock says.
Whatever transpires in the next round of experiments, it seems certain that the unsolid nature of some solids will continue to confound. “Everyone in the field just wants to know what’s going on,” says Beamish. “There sure is something interesting, and it will be more exciting if it really is supersomething.”
Helium’s many phases
Aside from a few popular party tricks — making balloons float and voices high-pitched — helium appears to be a rather ordinary gas. But cool the element to just a few degrees above absolute zero, where it changes from a gas into a liquid, and its atoms leave the world of classical physics behind and begin to exhibit quantum behavior. These bizarre properties have led to a number of Nobel prizes for the study of liquid helium.

1. Because it has the lowest boiling point of any element, after its discovery on Earth in the late 1800s helium took center stage in the drive to reach temperatures closer and closer to absolute zero. Liquefying the element would mean hitting 4.2 kelvins, or degrees above absolute zero, in the lab. Dutch physicist Heike Kamerlingh Onnes achieved the feat in 1908, and in 1913 received the Nobel Prize in physics for his efforts. But strange discoveries were still in store.
2. As liquid helium was cooled below 4 kelvins, it boiled like crazy. But below 2.17 kelvins, the boiling suddenly stopped. This meant that helium-4, the most abundant form of the element, had two distinct liquid states, with a transition between them at 2.17 kelvins. Below that temperature, Russian physicist Pyotr Kapitsa found in 1937, liquid helium flowed without friction. This form of liquid, dubbed a superfluid, could flow through tiny holes, creep over the edge of a cup and pull itself up and out of a tube, like a fountain (pictured). Kapitsa received a Nobel Prize in 1978 for his studies into low-temperature physics.
3. Superfluidity arises when the atoms in superfluid helium join up in a quantum state called a Bose-Einstein condensate, allowing them to exhibit collective behavior. Russian condensed-matter theorist Lev Landau developed a mathematical explanation for the behavior of helium at low temperatures and was awarded a Nobel in 1962.
4. In the 1970s scientists discovered that helium-3, a rare isotope of the element, also has a superfluid state at temperatures lower than 2 millikelvins. David Lee, Douglas Osheroff and Robert Richardson shared the physics Nobel in 1996 for that find. The behavior of helium-3 appears to be even more complex than that of helium-4, and some scientists have tried to use the vortices that form in superfluid helium-3 to model the development of spacetime defects, called cosmic strings, in the early universe.
5. Helium-3 is much harder to get in a superfluid state than helium-4 because it is made of fermions, particles that cannot occupy the same quantum state. So explaining how helium-3 atoms could begin to move collectively was a theoretical puzzle. British-born physicist Anthony Leggett cracked this problem by proposing that the atoms pair up, analogous to the way electrons pair up in superconductors. Leggett won a 2003 Nobel for his contributions to the understanding of how atoms behave in superfluids.
Today, scientists are finding that the frictionless flow observed in liquid helium may also occur in the solid phase. But the jury is still out on whether supersolidity exists — and whether its discovery will lead to any Nobels. — Elizabeth Quill
Image credit: S. Balibar/Nature 2010