PENCIL IT IN Scientists have used two new versions of CRISPR gene editors, which rewrite DNA like pencils instead of cutting like scissors, to change single-letter typos that cause some blood disorders.
Wildpixel/iStockphoto
New gene-editing tools can correct typos that account for about half of disease-causing genetic spelling errors.
Researchers have revamped the CRISPR/Cas9 gene editor so that it converts the DNA base adenine to guanine, biological chemist David Liu and colleagues report October 25 in Nature. In a separate study, published October 25 in Science, other researchers led by CRISPR pioneer Feng Zhang re-engineered a gene editor called CRISPR/Cas13 to correct the same typos in RNA instead of DNA.
Together with other versions of CRISPR/Cas9, the new editors offer scientists an expanded set of precision tools for correcting diseases.
CRISPR/Cas9 is a molecular scissors that snips DNA. Scientists can guide the scissors to the place they want to cut in an organism’s genetic instruction book with a guide RNA that matches DNA at the target site. The tool has been used to make mutations or correct them in animals and in human cells, including human embryos (SN: 10/14/17, p. 8).
A variety of innovations allow CRISPR/Cas9 to change genetic instructions without cutting DNA (SN: 9/3/16, p. 22). Earlier versions of these “base editors,” which target typos related to the other half of disease-causing genetic spelling errors, have already been used to alter genes in plants, fish, mice and even human embryos.
Such noncutting gene editors are possibly safer than traditional DNA-cutting versions, says Gene Yeo, an RNA biologist at the University of California, San Diego. “We know there are drawbacks to cutting DNA,” he said. Mistakes often arise when cellular machinery attempts to repair DNA breaks. And although accurate, CRISPR sometimes cuts DNA at places similar to the target, raising the possibility of introducing new mutations elsewhere. Such “permanent irreversible edits at the wrong place in the DNA could be bad,” Yeo says. “These two papers have different ways to solve that problem.”
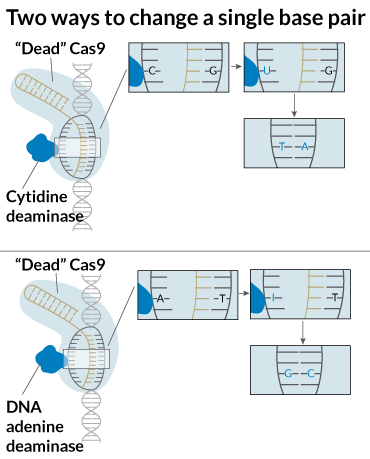
The new editors allow researchers to rewrite all four bases that store information in DNA and RNA. Those four bases are adenine (A) which pairs with thymine (T) (or uracil (U) in RNA), and guanine (G) pairs with cytosine (C). Mutations that change C-G base pairs to T-A pairs happen 100 to 500 times every day in human cells. Most of those mutations are probably benign, but some may alter a protein’s structure and function, or interfere with gene activity, leading to disease. About half of the 32,000 mutations associated with human genetic diseases are this type of C-G to T-A change, says Liu, a Howard Hughes Medical Institute investigator at Harvard University. Until now, there was little anyone could do about it, he says.
In RNA, DNA’s chemical cousin, some naturally occurring enzymes can reverse this common mutation. Such enzymes chemically convert adenine to inosine (I), which the cell interprets as G. Such RNA editing happens frequently in octopuses and other cephalopods and sometimes in humans (SN: 4/29/17, p. 6).
Zhang, of the Broad Institute of MIT and Harvard, and colleagues made an RNA-editing enzyme called ADAR2 into a programmable gene-editing tool. The team started with CRISPR/Cas13, molecular scissors that normally cut RNA. Dulling the blades let the tool grasp instead of slice. Zhang and colleagues then bolted the A-to-I converting portion of ADAR2 onto CRISPR/Cas13. Dubbed REPAIR, the conglomerate tool edited from 13 percent to about 27 percent of RNAs of two genes in human cells grown in dishes. The researchers did not detect any undesired changes.
Editing RNA is good for temporary fixes, such as shutting down inflammation-promoting proteins. But to fix many mutations, it requires permanent DNA repairs, says Liu.
In 2016, Liu’s team made a base editor that converts C to T. Chinese researchers reported in Protein & Cell on September 23 that they used the old base editor in human embryos to repair a mutation that causes the blood disorder beta-thalassemia. But that editor couldn’t make the opposite change, switching A to G.
Unlike with RNA, no enzymes naturally make the A-to-I conversion in DNA. So Nicole Gaudelli in Liu’s lab forced E. coli bacteria to evolve one. Then the researchers bolted the E. coli DNA converter, TadA, to a “dead” version of Cas9, disabled so it couldn’t cut both strands of DNA. The result was a base editor, called ABE, that could switch A-T base pairs into G-C pairs in about 50 percent of human cells tested.
This base editor works more like a pencil than scissors, Liu says. In lab dishes, Liu’s team corrected a mutation in human cells from a patient with an iron-storage blood disorder called hereditary hemochromatosis. The team also re-created beneficial mutations that allow blood cells to keep making fetal hemoglobin. Those mutations are known to protect against sickle cell anemia.
Another group reported in the October Protein & Cell that base editing appears to be safer than traditional cut-and-paste CRISPR/Cas9 editing. Liu’s results seem to support that. His team found that about 14 percent of the time cut-and-paste CRISPR/Cas9 made changes at nine of 12 possible “off-target” sites. The new A-to-G base editor altered just four of the 12 off-target sites and only 1.3 percent of the time.
That’s not to say cut-and-paste editing isn’t useful, Liu says. “Sometimes, if your task is to cut something, you’re not going to do that with a pencil. You need scissors.”
Editor’s note: Feng Zhang is on the board of trustees of the Society for Science & the Public, which publishes Science News.