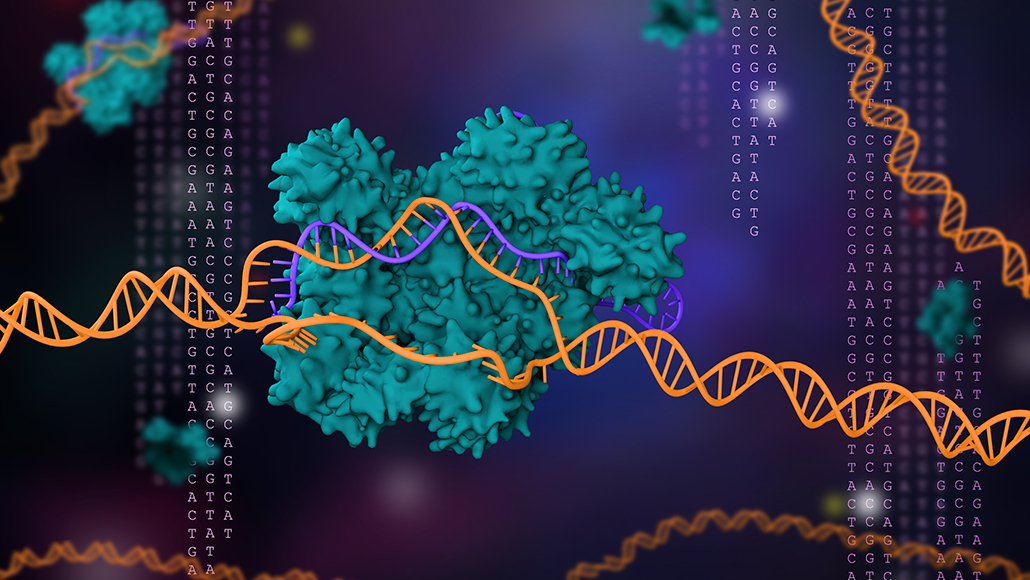
Researchers Emmanuelle Charpentier and Jennifer Doudna devised a powerful gene-editing tool called CRISPR/Cas9 (illustrated). The gene editor has revolutionized molecular genetics and gene therapy and netted the duo the 2020 Nobel Prize in chemistry.
Meletios Verras/iStock/Getty Images Plus