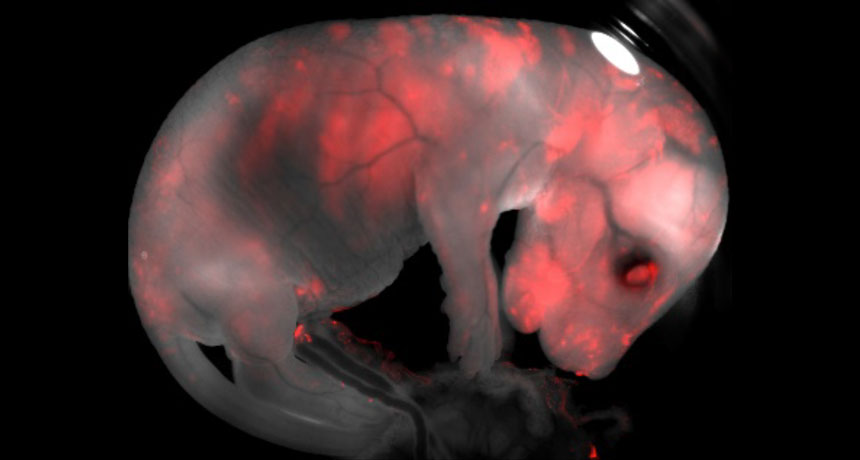
SPOT ON Researchers created a mouse embryo that contains rat cells (red). Hybrid, or chimeric, animals may eventually grow human organs to be used in transplants.
Salk Institute
Growing human organs in other animals is a small step closer to reality.
Injecting human stem cells into pig and cattle embryos created embryos that incorporate a small number of human cells, scientists report January 26 in Cell. The ultimate goal of the controversial research is to use hybrid, or chimeric, animals to produce human organs for transplant.
Farm animals incubating human organs won’t appear anytime soon, says Juan Carlos Izpisua Belmonte, a stem cell biologist at the Salk Institute for Biological Studies in La Jolla, Calif. “I feel we’re still far away from that,” says Belmonte, who led the work. It has taken his group four years “just to deliver a message that, yes, human cells can integrate into a pig.”
While human-animal chimera work is still in its infancy (and faces ethical and funding hurdles, see sidebar), hybrids of rats and mice are already hinting that growing an organ from one species in another is a viable strategy for curing some diseases. Researchers report January 25 in Nature that they grew mouse pancreases in rats. Mouse insulin-producing cells were extracted from the rat-grown organs and transplanted into diabetic mice, curing their diabetes. Transplanted cells kept the mice’s blood sugar normal for more than a year even though the mice were not given immune-suppressing drugs to prevent rejection after the first five days following the transplant. That finding raises the hope that animal-grown organs tailored to individual patients could be transplanted without fear of rejection.
The mouse-grown pancreas wasn’t big enough to transplant back into a rat. So for the new study, Nakauchi and colleagues reversed the experiment, growing a mouse pancreas in a rat. The researchers used a genetic trick to ensure that the pancreas was composed mostly of mouse cells, breeding rats that cannot make a pancreas because of defects in the Pdx-1 gene. Normally rats without a pancreas die shortly after birth. But animals that got mouse cells were able to grow a functioning pancreas.
In the study reported in Cell, Belmonte and colleagues also worked with rodents, creating mice growing rat organs. Using the gene-editing tool CRISPR/Cas9, the researchers disabled Pdx-1 and genes involved in heart and eye development so that mice couldn’t grow functioning versions of those organs. Rat stem cells injected into mouse embryos filled in, growing functional organs, including one the researchers didn’t expect. Rats don’t have gallbladders, but rat stem cells introduced into mice embryos are able to form gallbladders, the researchers reported.
This finding illustrates that the host environment has significant control over what happens to stem cells, says Daniel Garry, a transplant cardiologist and stem cell biologist at the University of Minnesota in Minneapolis. “That’s pretty cool biology.” The result also indicates that it may not be possible to re-create in lab dishes the mechanical forces, chemical signals and other conditions an organ needs to develop properly.
In the case of the human-pig chimeras, researchers weren’t sure how well the human cells would fare, Garry says. For all scientists knew, human stem cells might take over pig embryos. “That would be very much unwanted,” Garry says.
But the Salk researchers and colleagues found that human cells don’t incorporate efficiently into pig embryos. Of 2,075 pig embryos injected with human stem cells and transferred to sows’ uteruses, only 186 grew for 21 to 28 days — as long as the researchers allowed development to continue. Of those, 67 contained human cells. Most of the chimeric embryos were underdeveloped, indicating that human cells may interfere with normal pig development. Early cattle embryos appeared to accept human cells more easily, but cows are more difficult to work with than pigs.
More work is needed to improve growth of human cells in pig embryos. The Salk researchers hope to use CRISPR/Cas9 to engineer pigs to lack certain organs just as they did with rodents, says Jun Wu, a stem cell biologist on Belmonte’s team. Human cells able to supply the missing organs might have a growth advantage and survive better.
Research Hurdles
Some people are not excited about the prospect of growing human organs in animals. “People view chimeras as monsters,” says Stanford stem cell biologist Hiromitsu Nakauchi, who is attempting to make pigs and sheep that carry human organs intended for transplant.
Part of the discomfort many people feel stems from concerns that the chimeras might be “too human,” giving pigs human brains or raising the possibility that breeding a chimeric animal could produce a human baby, says Robert Streiffer, a bioethicist at the University of Wisconsin‒Madison.
In some countries, creating human-animal chimeras is banned. Researchers in the United States are allowed to conduct human-animal chimera work using private or state funding, but not with federal money.
The U.S. National Institutes of Health currently has a moratorium on funding such research. In August, NIH announced intentions to lift the funding ban pending finalization of policy governing such research, but there is no final regulation yet. The proposed rules would expand prohibitions on introducing human stem cells into early nonhuman primate embryos and strengthen bans on breeding any animal that might produce human eggs or sperm, says Carrie Wolinetz, associate director of science policy for NIH. A new oversight committee would also be created to address issues of ethics and animal welfare raised by such research, Wolinetz says. “We’re trying to allow the research to move forward responsibly,” she says.
Bioethicist Françoise Baylis contends that there is no ethical way to continue chimera work because it is based on a faulty assumption that human life is more valuable than that of nonhuman beings. “The hope that one can ‘forever’ avoid the tough ethical questions by simply ensuring that the nonhuman animals are not ‘substantively humanized’ is flawed (short-sighted),” Baylis, of Dalhousie University in Halifax, Canada, wrote in an e-mail.







