More than a decade ago, theoreticians predicted that nitrogen, the major constituent of air, could assume a three-dimensional, polymeric structure. Now, chemists have made this polymeric nitrogen, and they say it might someday serve as a lightweight, high-energy storage material that could outperform conventional explosives, rocket fuels, and even automotive fuel.
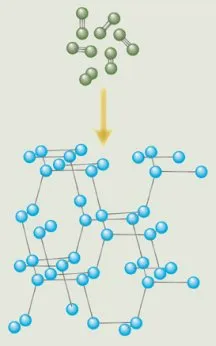
In its most stable configuration, nitrogen is a two-atom molecule—a pair of nitrogen atoms locked together by a triple bond. According to computer simulations, however, nitrogen atoms rearranged into a 3-D network could pack several times as much energy per unit of volume than do some of the most powerful explosives available today.
The simulations specify that each nitrogen atom in the network be bound via single bonds to three other nitrogen atoms. The energy in those three single bonds surpasses that of the triple bond in nitrogen’s conventional, two-atom form. Not only could polymeric nitrogen store and release large amounts of energy, but the only by-product would be ordinary nitrogen gas, which is environmentally benign.
Over the years, several groups around the world have attempted to create polymeric nitrogen in the lab. Like the process for making synthetic diamonds, production of polymeric nitrogen requires extremely high temperatures and pressures (SN: 9/14/02, p. 165: Better-Built Diamonds: Fast growth, purity may multiply uses).
Now, Mikhail Eremets of the Max Planck Institute for Chemistry in Mainz, Germany, and his colleagues report success where others have failed.
In the August Nature Materials, the researchers chronicle how they made the elusive polymeric nitrogen. First, they injected a small sample of nitrogen gas into a disk-shaped chamber inside a diamond-anvil cell. The chamber measured about 50 micrometers in diameter and 10 µm in height. Next, the researchers began introducing wrenching pressures inside the cell. They then heated the sample with a laser until the nitrogen began to solidify and turn dark.
At approximately 1,725°C and 115 gigapascals—a million times the pressure of the atmosphere at sea level—something remarkable happened. The sample transformed into a transparent crystal. Eremets calls it “nitrogen diamond” because it’s similar to diamond in appearance and structure. X-ray and optical analyses of the sample confirmed that the nitrogen had indeed become polymeric.
Christian Mailhiot of the Lawrence Livermore National Laboratory in California says that he’s thrilled to learn of the results, calling the research “a significant breakthrough.” Mailhiot was part of the original team that in 1992 predicted the existence of polymeric nitrogen. “It’s not very often that theorists predict a material beforehand,” he says.
Alain Polian of the Pierre and Marie Curie University in Paris was also impressed, noting that “the experiment required great skill.” However, there is one snag: The researchers have yet to recover the nitrogen crystal intact under ambient conditions because their cell didn’t survive the decompression.
Eremets says his team is currently tackling that problem as well as searching for more-practical ways to make the material.






