Proteins turned on by opium and similar substances in the body have now been caught in action. Two new snapshots show how cellular proteins lasso molecules in the opium family, revealing the 3-D structure of such pairings for the first time.
The work represents a major step toward designing more specific analgesics and other drugs that lack opioids’ nasty side effects, two teams of researchers report online March 21 in Nature.
“Both are landmark studies,” says Gavril Pasternak, a neuroscientist who designs opioids at the Sloan-Kettering Institute in New York City, and who wasn’t involved in either study. “These structures will quickly be utilized with goal of developing nonaddicting painkillers and new ways to combat drug abuse.”
Proteins that respond to opium and opiumlike molecules protrude from the surfaces of cells throughout the brain, spinal cord and gut. The body’s own hormones and brain chemicals such as endorphins can bind to these proteins to turn the molecular switches on and off to control pain, regulate breathing and change mood.
Many of today’s most powerful painkillers work by switching on one of these proteins, called the mu opioid receptor. But the relief this provides comes at a price. Derivatives of opium, such as morphine and codeine, are addictive and can cause breathing problems and constipation.
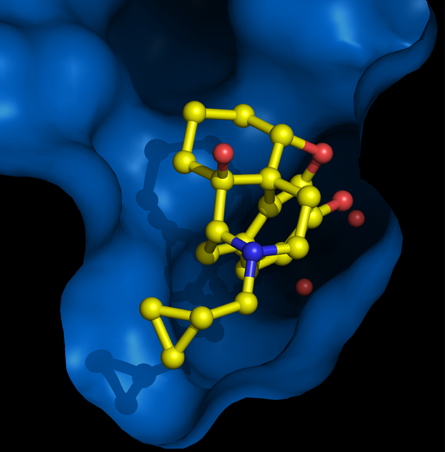
To better understand how these drugs work, an international team of researchers for the first time crystallized a small morphinelike molecule attached to a mu receptor — a technically difficult task that requires isolating the pair of molecules without unsticking them from each other. X-rays revealed how one molecule lined up with the other.
This structure “may prove to be important in understanding why some opioids are more addictive than others,” says Sébastien Granier, a molecular biologist at Stanford University and a coauthor on the mu receptor paper. Although the small molecule his team tested has the opposite effect of morphine — it deactivates the mu receptor — knowing how the molecule docks should help improve computer simulations of various drugs that can turn the protein on or off.
In the second study, Seva Katritch of the Scripps Research Institute in La Jolla, Calif., and colleagues looked in detail at how the experimental drug JDTic sticks to a kappa opioid receptor — a switch that’s turned on by the plant-derived hallucinogen Salvinorin A. “Kappa opioid receptors are especially interesting because of their emerging distinct role in regulation of stress,” says Katritch. JDTic, which flips the switch off, is being tested as a treatment for drug abuse.
Each team’s test molecule latched on to its receptor at multiple points in the revealed structures. Small differences in the attachment sites may help to explain why some molecules are more promiscuous than others. Designing new drugs that bind only with specific receptors could help to cut down on side effects.
“Everyone in the field has been waiting to see these crystal structures,” says Jane Aldrich, a medicinal chemist at the University of Kansas in Lawrence. “Now we can look at how particular parts of the molecules interact.”
The receptor pockets that grabbed ahold of the molecules also proved to be surprisingly large and open. This extra space should give drug designers a bit of wiggle room — and the flexibility to tack on extra atoms to drugs like morphine in the hopes of making them less problematic.







