Broken Symmetry
Scientists seek mechanisms explaining development of the body’s left-right pattern
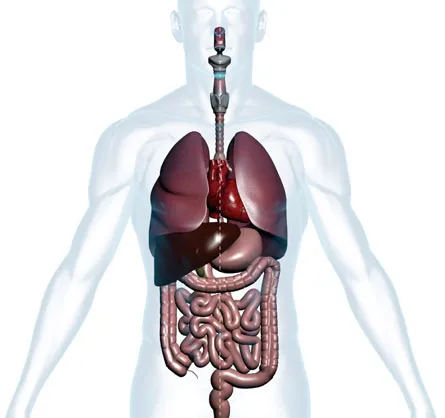
On the outside, people’s right and left sides look pretty much the same. On the inside, though, such superficial symmetry gives way to an imbalanced array of organs: The heart, spleen and stomach sit on the left side of the body, while the liver and pancreas take up the right. Even organs that at first glance appear as perfect mirror images of each other, such as the kidneys, lungs and testicles, turn out to have telltale left-right differences.
Figuring out how a body with such internal asymmetry develops from an egg (and later an embryo) with near-perfect symmetry has long stymied developmental biologists. But in recent years, scientists have identified a structure that seems to explain how mice — and possibly humans — break left-right symmetry early in development. In an 8-day-old mouse embryo, researchers discovered a short-lived, shallow pit covered with cilia. The whiplike protrusions paddle through the surrounding embryonic fluid, creating a microcurrent that flows over the pit from right to left. This leftward flow may send a signal that starts the body on its journey to an asymmetrical destination.
Many researchers believe that this cilia-powered flow is the left-right symmetry breaker in mice, and probably in fish, too. But while some other animal embryos have been shown to develop these pitlike structures, accumulating evidence suggests that frogs, rabbits and perhaps other animals have asymmetric concentrations of certain molecules long before any cilia form in the pits. Chickens don’t show any directional flow at all in their equivalent of this structure. And neither do pigs, a recent paper in Science reports. With so many inconsistencies, some scientists question whether the cilia model alone can explain symmetry breaking.
Another area of intense investigation is how this leftward flow might tell organs such as the heart and lungs which way to grow. In this year’s Annual Review of Fluid Mechanics, published in January, Nobutaka Hirokawa and colleagues at the University of Tokyo discuss one way in which left-side–determining chemicals might be transported leftward with the flow, to accumulate on one side of the embryo and begin a cascade of development. Other recent reviews have discussed different possible mechanisms.
Showing how left-right patterning emerges in human embryos is critical to understanding not just human development, but also what goes wrong in the thousands of people born each year with asymmetry disorders. These little-known diseases have a wide range of effects — some prove lethal in the first few hours of a child’s life, while other forms are harmless.
Mystery of the frozen cilia
Forty years ago, a baby girl was born blue. Doctors looking for the cause of her poor blood circulation found the answer in her heart: She had two left heart halves, pumping blood at a dangerously inefficient level. The rest of her body was symmetrical, too — with two left lungs, two left kidneys and spleens on either side of her abdomen.
Disorders in asymmetry of a single organ can cause serious health problems. Pediatric cardiologist Martina Brueckner of Yale University School of Medicine estimates that one in every 25 patients at heart clinics nationwide has some problem with asymmetry. Yet a totally reversed body asymmetry, called situs inversus, is not a problem in itself. Flipping an entire jigsaw puzzle upside down does not affect the function of each piece, but flipping a single piece certainly affects how the puzzle fits together. Those whose hearts are flipped in relation to the rest of their bodies face serious health problems. But people with situs inversus can live for years without realizing that their hearts and other organs are on the wrong side of their bodies.
In fact, it was people with situs inversus who first gave researchers the idea that cilia and fluid flow could function as symmetry breakers in an embryo.
In 1933, a Swiss doctor reported on several patients with a bizarre collection of symptoms: frequent infections in the lungs, ears and nose; in men, an inability to have children; and, in many patients, situs inversus. Named Kartagener syndrome after the doctor, the disorder affects about 1 in every 10,000 people.
In the mid-1970s, Swedish doctor Björn Afzelius made an intriguing discovery while examining four men diagnosed with Kartagener syndrome. Normally, the beating motion of the cilia that line people’s throats and ear canals pushes bacteria and debris out of these passageways. But the cilia of these men were stiff and unmoving. Afzelius and colleagues showed that in people with Kartagener syndrome, certain cilia are missing protein structures called the dynein arms, which make up the motor that allows the cilia to move. The static cilia explained the patients’ chest and ear infections, as well as the immotile sperm. But no one could figure out what cilia had to do with body asymmetry.
A biology graduate student in Hirokawa’s lab stumbled across the connection in the late 1990s, when his mutant mouse embryos died in utero. The genetically engineered mice lacked a gene key to normal cilia development. The researchers were forced to dissect the embryos at an earlier developmental stage than planned.
“It was kind of a lucky accident,” says Shigenori Nonaka, now at the National Institute for Basic Biology in Okazaki, Japan. At this time in development — 9 ½ days after fertilization — the heart is just beginning to form, visible inside a transparent envelope in the embryo.
“That’s when I noticed that some of the mutant mice had an inverted heart,” Nonaka says. In fact, nearly half of the mutant mouse embryos showed reversed asymmetry. This was, strangely enough, about the same percentage of cases of reversed asymmetry seen in people with Kartagener syndrome. In the mice, no functional cilia seemed to randomize the chance of the proper body pattern developing, the team reported in 1998 in Cell.
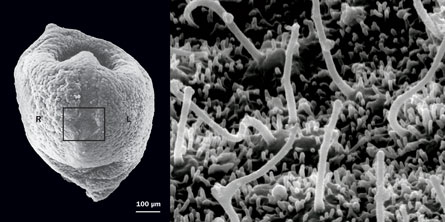
Nonaka and colleagues turned their attention to a triangular pit in the side of the embryo that eventually morphs into the beginnings of the spinal cord. In mice, this structure, called the ventral node, is covered with several hundred cilia, each rotating clockwise like a jump rope twirled overhead by a child. The researchers found that these twirling cilia were somehow pushing the surrounding fluid neatly from right to left.
The clincher came when the team artificially reversed the direction of the flow across the ciliated pit, forcing the fluid to move from left to right. In a seminal 2002 paper in Nature, Nonaka, Hiroshi Hamada of Osaka University and colleagues showed that the left side of a mouse goes with the flow. When the flow moved toward the right instead of the left, the mice developed reversed asymmetry.
“That was the big breakthrough,” says physicist Julyan Cartwright of the University of Granada in Spain. “It showed that it was fluid mechanics that was driving the process.”
The role of the cilia explained why patients with Kartagener syndrome had about a 50-50 chance of situs inversus. In the patients with frozen cilia, the fluid in the node would have had nothing to guide its flow; sometimes the flow went the “right” way, and sometimes it didn’t.
Mystery of the leftward flow
But it wasn’t clear how twirling cilia could produce a uniform leftward flow. Technically the cilia should have moved the fluid around in a vortex, the way an eggbeater creates whirlpools in a bowl of whipping cream. Instead, somehow these eggbeaters were pushing the fluid from one side of the bowl (as it were) to the other.
Cartwright, who has worked on fluid mechanics problems in a variety of biological systems, hypothesized that the orderly leftward flow could be produced if the cilia were positioned at an angle instead of straight up and down.
Consider the rotaries of an eggbeater: “If you have them vertically in a bowl of some liquid you’re mixing, it gives you a vortex, a whirlpool,” Cartwright says. “But if you tilt them over, you get flow along the surface. And that’s exactly what nodal flow is like.”
Some fluid would still flow to the right, Cartwright reasoned in a 2004 paper in the Proceedings of the National Academy of Sciences. But the rightward flow would come in contact with the walls of the pit, and the friction from this contact would slow it down. Leftward flow would be the stronger of the two.
Nonaka and Hamada confirmed Cartwright’s theory when their team caught the tilt of the cilia in action using a high-speed microscope, publishing their results in PLoS Biology in 2005.
But deciphering the first steps of asymmetry is not the same as understanding how the body develops its full asymmetry. Researchers now want to know how fluid flowing leftward in a divot on an embryo tells the body which way to grow.
Some scientists think the leftward flow may carry chemicals that turn on a sequence of left-side–producing genes. Hirokawa’s group at the University of Tokyo suggested that such chemicals could be encased in a lipid envelope, what he calls a nodal vesicular parcel, that could be carried leftward with the flow and then break upon impact with the node’s floor, spilling its chemical contents. In Nature in 2005, the researchers reported finding evidence of these parcels using time-lapse microscopy.
But others note that in such a small-scale system, where the thickness of the fluid is great compared with its volume, the impact of hitting the node floor would not provide enough force to break open such a package. To spill the chemicals in the appropriate place would require an active parcel-breaking mechanism.
“If you stop stirring, everything stops,” Cartwright says. “The parcel can’t break as if it were a ripe tomato that’s being squashed on the floor.”
Other researchers prefer the idea that some cilia are sensors that communicate a signal based on the direction of the flow. In 2003, Yale’s Brueckner and colleagues showed that the cilia at the periphery of the node are missing a dynein structure needed for movement. That suggests that outlying cilia may play a role other than generating the leftward flow and might respond passively to flow produced by their neighbors, sending signals when the cilia bend like trees in a storm.
Not by mouse alone
Scientists have found strong evidence that nodal pit fluid flow determines left from right — in a mouse. Work with zebrafish also suggests that similar flow plays a part in breaking symmetry.
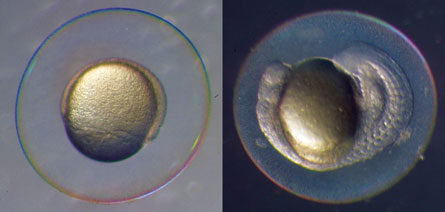
But some animals, like frogs and rabbits, appear to break symmetry well before their equivalent of the nodal pit appears. Biologist Michael Levin of Tufts University in Boston and colleagues discovered in 2005 that in frogs, symmetry may be broken as early as the first few hours after the egg is fertilized. In the May 15 Science, Harvard’s Clifford Tabin and colleagues in Germany reported that pigs do not have cilia in their nodal pits, yet still are asymmetric, so perhaps humans and other mammals rely on other early symmetry-breaking events, too.
Meanwhile, scientists have uncovered other potential mechanisms for left-right patterning. Individual cells can create a left-right axis using their own intrinsic sense of left and right, given to them by the inherently biased structure of their protein skeletons, researchers from the University of California, San Francisco and the University of California, Irvine showed in 2007. The team, led by Henry Bourne of UCSF, claimed that this mechanism could work in the absence of any external cues.
Some experts wonder if there really are distinct symmetry-breaking mechanisms that differ from animal to animal.
“My own view is that the origin of left-right asymmetry is really fundamental, and has been with us from the single-cell level,” Levin says.
While a universal symmetry breaker for all animals remains to be found, one thing almost all animals share is two genes — appropriately named Nodal and Lefty. Wherever Nodal expression is concentrated, there the left side grows, with Lefty apparently keeping Nodal in check. The presence of the genes in animals as diverse as snails, sea urchins, frogs, mice and people indicates to many that these two genes play a fundamental role in the process of telling left from right, regardless of the initial symmetry-breaking process.
“The most important event is the ability of Nodal to take slight asymmetries set up by a variety of means and translate them into a robust asymmetry,” Tabin says.
Even if fluid flow is not the first symmetry-breaking event, it may amplify an initial asymmetry, or even be a backup mechanism — what Cartwright calls a “belt-and-braces” design, with two mechanisms designed for the same purpose (such as holding up trousers). Nevertheless, many scientists agree that nodal flow plays an important role in the process of breaking symmetry at least in some animals — and given the connections between cilia and asymmetry in human disorders such as Kartagener syndrome, probably in people, too.
“It would be extraordinarily unlikely in my view that humans don’t use the same mechanism as in mice,” Tabin says.
Left-left and right-right
In people, some birth defects involving asymmetry can kill a person within the first minutes of life. Other kinds allow a somewhat normal life. And these conditions occur so early in development that it is impossible to repair the defects except through surgery. As Tabin says, doctors can’t return an individual to an embryonic state as a therapy.
But some researchers hope that understanding more about how asymmetry is produced will help physicians to tailor treatments to individuals.
The girl born blue was lucky. She survived to her teen years, had life-prolonging heart surgery and grew up to hold a regular job and even have children. But only about 60 percent of children born with two left sides survive their first five years, Brueckner says. Children with two right halves survive to the age of 5 in only about 30 percent of cases.
Brueckner and colleagues are now collecting genetic information from patients with asymmetry disorders to find out why some people survive to adulthood while others die within hours of birth. The gene library may help explain why people develop the wrong asymmetries or fail to develop asymmetry at all, she says.
Many related mysteries persist. Scientists still don’t know how an organ like the heart forms the correct way rather than in reverse, Tabin says. And though Kartagener syndrome patients with situs inversus have reversed brain hemispheres, the patients still have the same tendency to right-handedness as the rest of the population — a ratio of about 9-to-1. To Tabin, this forces biologists to ask themselves to what extent form in the brain determines function.
“We’re piecing it together in very broad strokes,” Tabin says. “I don’t worry about running out of things to do for the next two decades.”