How does early pregnancy lower breast cancer risk? Odd cells could offer clues
In mice, pregnancy appears to slow the buildup of suspicious mammary cells

For women, giving birth by their 20s may cut lifetime breast cancer risk, but no one knows how. New clues may come from abnormal cells in mice.
freestocks.org/pexels
A cellular Frankenstein’s monster has breathed new life into an old breast cancer mystery.
For decades, scientists have known that early-in-life pregnancy somehow dials down the risk of breast cancer as women age. No one has pinpointed how, but a group of strange mammary cells could help crack the case. In female mice, these cells build up over time — unless an animal has been pregnant, scientists report January 21 in Nature Communications.
That’s a hint that pregnancy is blocking the buildup, and that might be a good thing. The cells have undergone an identity crisis that might spur cancer progression, says Shaheen Sikandar, a cancer and stem cell biologist at the University of California, Santa Cruz. If the cells do indeed nudge tissue toward disease, preventing their accumulation might also prevent breast cancer, she says. “We are trying to see why these cells are showing up, and if there’s a way we can stop them.”
For women, giving birth by their 20s may cut lifetime breast cancer risk, though estimates of how much vary. Some clues about the phenomenon come from young mice, whose mammary tissues undergo molecular changes after pregnancy. But no one had a good sense of what was going on in older animals, after the pregnancy protection effect kicks in, Sikandar says.
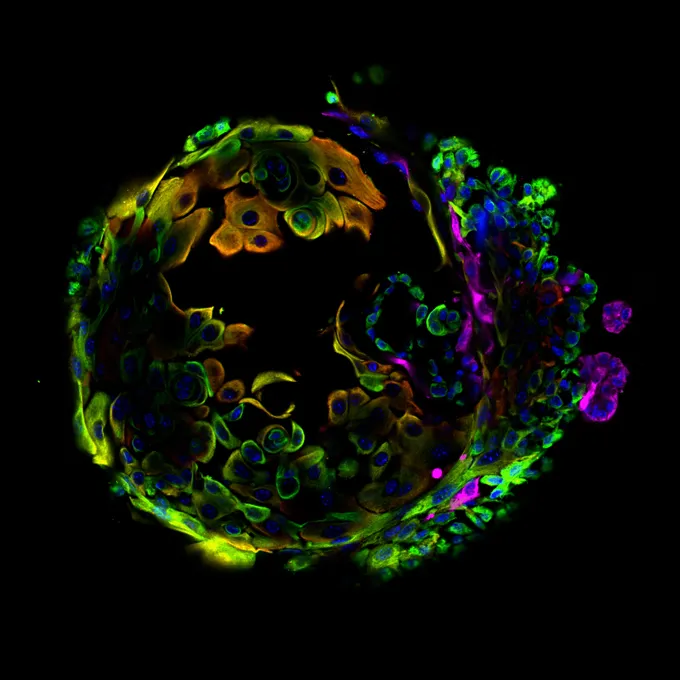
Her team compared mice that had been pregnant with those that hadn’t and examined both groups at different ages — up to 18 months old, the equivalent of 55 to 65 in human years. The researchers probed mammary tissue using a technique that let them identify cells and see what each one was up to.
In aged mice that hadn’t been pregnant, the team found cells with peculiar features. They were hybrid, for one. Instead of matching either of the two main mammary cell types, the hybrid cells were a mash-up of both. This kind of altered identity may be a key driver of cancer, a different team of researchers studying lung tumors reported in a separate study published January 21 in Nature.
The hybrid cells Sikandar’s team discovered produce an inflammatory molecule called IL33. Injected into young mice, IL33 seemed to age mammary tissue, compelling it to form hybrid cells, like those in older, never-pregnant mice, the researchers found. It’s possible that early pregnancy offers a cellular reset, halting the pileup of these irregular cells, Sikandar says. But many questions remain.
Mice and humans are very different animals, so it’s not clear whether the team’s observations will hold up in people, says Mark LaBarge, a breast cancer researcher at City of Hope in Duarte, Calif. It’s also too early to say whether these hybrid cells are an engine of cancer at all. Sikandar agrees. “We think they are bad,” she says. “They show all the signs of being bad.” But the team is doing the experiments now to find out.
For LaBarge, the study has sparked “cautious optimism and curiosity.” By looking in mice, Sikandar’s team has found a “really interesting population of cells,” he says, and that’s a reason to investigate further.