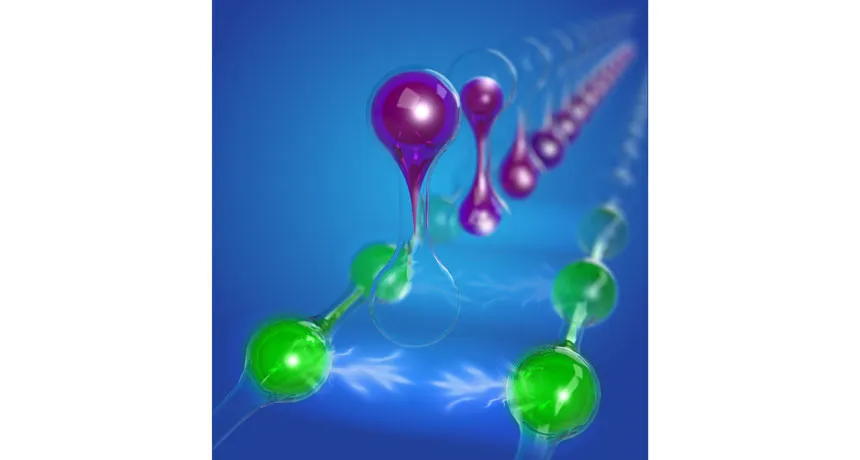
ELECTRONS ATTRACT Although electrons normally repel one another, a new study shows that two electrons (green in this illustration) can attract, thanks to their repulsion from other electrons (purple).
Avishai Benyamini and the graphics unit in Weizmann Institute
Standoffish electrons typically keep one another at arm’s length, repelling their neighbors. But surprisingly, under certain circumstances, this repulsion can cause pairs of electrons to soften their stance toward one another and attract instead, new research shows. The effect may be the key to someday producing a new type of high-temperature superconductor, scientists report in the July 21 Nature.
Though the effect was first predicted over 50 years ago, previous attempts to coerce electrons to behave in this chummy way have failed. Like charges repel, so negatively charged electrons ordinarily rebuff one another. But now researchers have validated the counterintuitive idea that an attraction between electrons can emerge. “Somehow, you have [this] magic that out of all this repulsion you can create attraction,” says study coauthor Shahal Ilani, a physicist at the Weizmann Institute of Science in Rehovot, Israel.
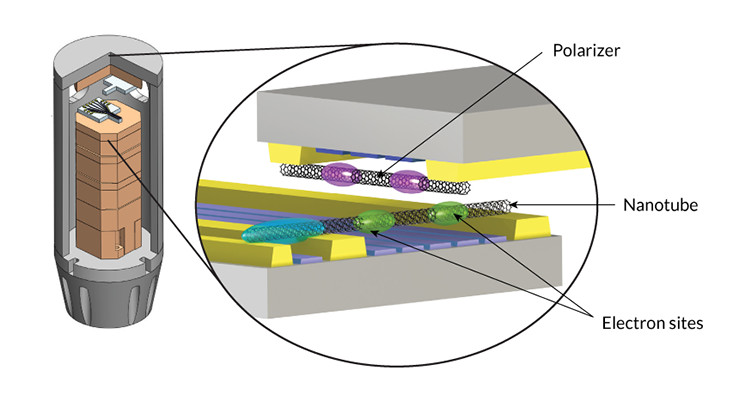
Ilani and colleagues produced the effect in a bare-bones system of electrons in carbon nanotubes. Operating at temperatures just above absolute zero, the system is made up of two perpendicular carbon nanotubes — hollow cylinders of carbon atoms — about 1 nanometer in diameter.
Two electrons sit at sites inside the first nanotube. Left to their own devices, those two electrons repel one another. A second nanotube, known as the “polarizer,” acts as the “glue” that allows the two electrons to attract. When the scientists brought the two nanotubes close together, says Ilani, “the electrons in the first nanotube changed their nature; they became attractive instead of repulsive.”
This flip is due to the nature of the polarizer. It contains one electron, which is located at one of two sites in the carbon nanotube — either between the first nanotube’s pair of electrons or farther away. The pair of electrons in the first nanotube repels the polarizer’s electron, kicking it from the near to the far site. And the electron’s absence leaves behind a positively charged vacancy, which attracts the pair of electrons toward it — and toward each other.
Come together
To demonstrate how normally repellant electrons can attract, scientists created a system of electrons in nanotubes, shown below. The apparatus, left, is housed inside a cooling system. Inside are two perpendicular carbon nanotubes (right). The lower nanotube contains two electrons located at two sites along the tube (green). The top nanotube, known as the polarizer, contains one electron, which can be located at one of two sites (purple). Researchers found that when the nanotubes were brought close together, the electrons at the green sites were attracted to one another, due to their interaction with the polarizer.
It’s a “tour de force,” says Takis Kontos, a physicist at the École Normale Supérieure in Paris, who wrote a commentary on the paper in the same issue of Nature. Although the system the scientists created is very simple, he says, “the whole experiment built around it is extremely complex.”
Electrons are known to attract in certain situations. In conventional superconductors, electrons pair up due to their interactions with ions in the material. This buddy system allows superconductors to conduct electricity without resistance. But such superconductors must be cooled to very low temperatures for this effect to occur.
But in 1964, physicist William Little of Stanford University theorized that electron pairs could likewise attract due to their interactions with other electrons, instead of ions. Such pairs should stay linked at higher temperatures. This realization sparked hopes that a material with these attracting electrons could be a room-temperature superconductor, which would open up a wealth of technological possibilities for efficiently transmitting and storing energy.
It’s yet to be seen whether the effect can produce a superconductor, and whether such a superconductor might work at higher temperatures — the new discovery shows only that the attraction can occur due to electrons’ repulsion. It’s “the first important step,” says Ilani. Now, scientists can start thinking of how to build “interesting new materials that are very different than what you can find in nature.”






