UNLEASHED The genetic tool CRISPR/Cas 9 has opened up gene drive technology to provide potential solutions for a multitude of problems in global health and ecology.
Michael Morgenstern
Genies are said to have the power to grant three wishes. But genies recently released from laboratory flasks promise to fulfill nearly any wish a biologist can dream up.
End the scourge of insect-borne diseases? Check. Inoculate endangered amphibians against killer fungi? Yes. Pluck invasive species from environments where they don’t belong? As you wish.
These genies aren’t magical; they are research tools known as gene drives — clever bits of engineered DNA designed to propel themselves into the DNA of a pesky or troubled organism. A gene drive is a targeted contagion intended to spread within species, forever altering the offspring.
Gene drive enthusiasts say these genies could wipe out malaria, saving more than half a million lives each year. Invasive species, herbicide-resistant weeds and pesticide-resistant bugs could be driven out of existence. Animals that carry harmful viruses could be immunized with ease.
Scientists have sought the power of gene drives for decades. But only with the emergence of a genetic tool called CRISPR/Cas9 — the bottle opener that unleashed the genie — has gene drive technology offered the prospect of providing a speedy means to end some of the world’s greatest health and ecological scourges.
“Everything is possible with CRISPR,” says geneticist Hugo Bellen. “I’m not kidding.”
But genes designed to spread through populations and alter ecosystems could have unforeseen consequences. Researchers have designed ways to keep gene drives confined in the lab, but no such safety nets exist for gene drives released into the wild. A technology to eradicate entire species, even when those species are pests, raises ethical and regulatory issues that scientific and government agencies are just beginning to consider.
As yet, no CRISPR gene drive has been released in the wild — few have even been built. There are plenty of technical hurdles to overcome. But there is enough awareness of the peril accompanying the promise that researchers and philanthropic organizations interested in the technology recently asked the U.S. National Academy of Sciences to weigh in on gene drives. The academy’s report won’t be issued until next year, but that hasn’t stopped the debate — or gene drive science — from moving forward.
Story continues after graphic
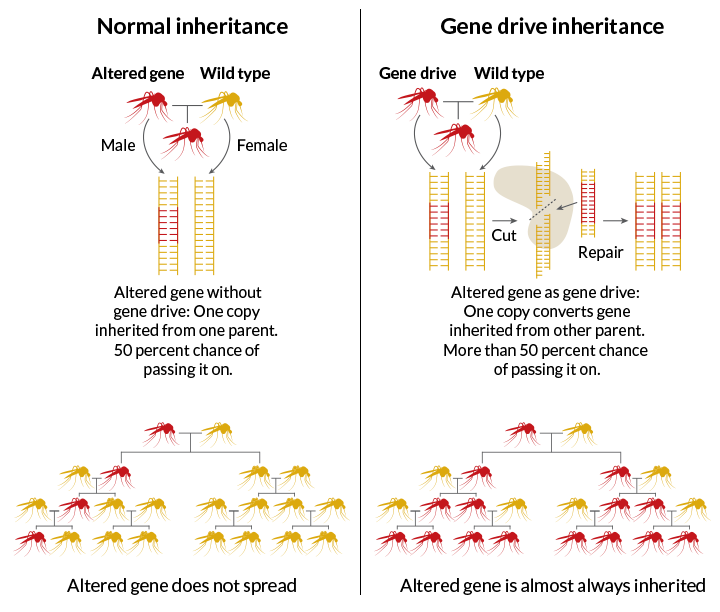
Rule breakers
Gene drives aren’t naked DNA floating around the air and water. They are molecular tools that scientists engineer into an organism’s DNA. While biologists have long imagined building gene drives, it was the arrival of CRISPR that transformed imagination into application.
CRISPR (an acronym for “clustered regularly interspaced short palindromic repeats”) refers to bits of viral DNA that bacteria have incorporated into their own genomes. With assistance from the enzyme known as Cas9, CRISPRs help bacteria defend themselves against viruses. In 2012, researchers announced that they had modified the CRISPR system into a gene-editing tool for cutting and pasting nearly any gene into any organism. Since then, CRISPR gene editing has taken biology by storm. It has become so widespread and easy that even third-graders are using it in science class, says CRISPR cocreator Jennifer Doudna of the University of California, Berkeley.
Already, researchers have used CRISPR/Cas9 to edit genes in human cells grown in lab dishes, monkeys (SN: 3/8/14, p. 7), dogs (SN: 11/28/15, p. 16), mice and pigs (SN: 11/14/15, p. 6), yeast, fruit flies, the worm Caenorhabditis elegans, zebrafish, tobacco and rice.
“I do CRISPR every day,” says Bellen, of Baylor College of Medicine in Houston. All around him, scientists are engineering fruit flies, nematode worms and mice using the bacteria-derived tool. “It’s one of those very rare events when a technology revolutionizes how you do science.”
Story continues after table
Usually, molecular biology techniques are specific to one organism, so CRISPR’s flexibility in editing genes from such a wide array of organisms is extremely attractive for researchers, says Anthony James, a molecular biologist at the University of California, Irvine.
If they can nimbly edit any gene in any organism, scientists ought to be able to design a gene drive for that species as well. That could mean that invasive species such as Australia’s pesky cane toads and other organisms that scientists haven’t been able to manipulate genetically could now become gene drive targets with CRISPR.
Gene drives are just the latest attempt at biological control systems. In the past, biologists have introduced natural enemy species to control pests. For instance, a fungus has been used to tamp down gypsy moth incursions in the northeastern United States. Gene drives aren’t much different, says gene drive pioneer Austin Burt, an evolutionary geneticist at Imperial College London. “But instead of releasing a whole species, you’re releasing a gene.”
Burt has made a career studying “selfish genetic elements,” parasitic pieces of DNA or RNA that exist only to propagate themselves. They go by other names, such as jumping genes, transposable elements, biased gene converters or meiotic drivers. Selfish elements live in a wide variety of organisms (including humans), and have devised a variety of methods for getting themselves passed on. What they have in common is the ability to circumvent the normal rules of inheritance, first described by Gregor Mendel in the 1860s.
Under Mendelian rules, a gene has a 50-50 shot at being passed from a parent to an offspring. Selfish elements don’t play by those rules. They manipulate the system to get inherited by more than 50 percent of offspring, even if it means harming the organism.
Engines of change
In 2003, Burt proposed harnessing some of these selfish entities for the greater good. He envisioned inserting a selfish element into a particular gene, creating a gene drive, which would change the inheritance rules to make sure that the drive would get passed on to a majority of a creature’s progeny.
Burt’s notion was to domesticate a microbial family of selfish elements called homing endonuclease genes. Those genes make endonucleases — DNA-cutting enzymes that target one spot in an organism’s entire genetic catalog, or genome. Like molecular scissors, the enzyme snips the target DNA if it doesn’t already have the selfish element. Next, a copy of the homing endonuclease gene with its surrounding DNA inserts itself into the gap as the cell heals the breach. Once inserted into one chromosome, the gene snips and pastes itself into the matching chromosome inherited from the other parent. So when the organism mates and divides its genetic material, both chromosomes will carry the editing machinery. In the fertilized egg, this cut-and-paste machinery would also convert the mate’s DNA. This repetitive editing allows the selfish element to drive itself into nearly every organism in a population. Gene drives could speed through a population like wildfire blazing across grasslands.
This approach has special appeal for weakening the disease-carrying dexterity of mosquitoes. A gene drive carried by 1 percent of mosquitoes in a population can be inherited so efficiently that in about 20 generations, 99 percent of all the mosquitoes will carry it, Burt calculates.
Burt’s math probably isn’t far from what might happen if a gene drive were released in the wild. “This has happened in nature,” says Nora Besansky, who studies malaria-carrying mosquitoes. A selfish element called the P element invaded the DNA of Drosophila melanogaster fruit flies in the 1950s, and “in less than 50 years, spread itself worldwide, across oceans, without any human intervention,” says Besansky, of the University of Notre Dame.
Humans might be able to direct gene drives to kill only female mosquitoes (the ones that bite and spread disease), or render the insects incapable of carrying malaria, dengue or other diseases.
To put his gene theory into practice, all Burt and his colleagues had to do was reengineer a homing endonuclease to cut a certain spot in a mosquito’s genome. Not so easy, says synthetic biologist Kevin Esvelt of Harvard Medical School. “It’s one of the hardest problems in protein engineering.”
It took years, but in 2011, Burt and colleagues announced in Nature that they had created a homing endonuclease that could find and cut a gene in mosquitoes. That experiment showed that building a gene drive in mosquitoes is possible. But gene drives that will get rid of mosquitoes or hinder their ability to transmit malaria are in the works (SN Online: 11/23/15).
In addition to homing endonucleases, scientists have been tinkering with two artificial protein systems as programmable gene-editing tools. Those tools, called zinc finger nucleases and TALENs (short for transcription activator-like effector nucleases), link a cutting enzyme to a protein that binds DNA in specific spots. Those molecules have enabled scientists to make precision edits to a menagerie of genomes (SN: 12/12/15, p. 7). But the proteins “are a burden to make,” says Bellen. Working with them is “just too painful and too slow,” he says. “If it’s not fast and efficient, it’s not good technology.”
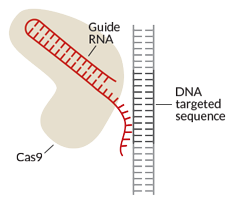
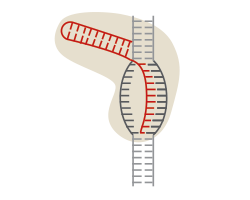
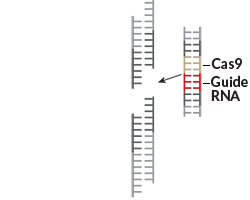
CRISPR provides the speed. Like earlier technologies, the part of the CRISPR system that snips DNA is a protein (the Cas9 enzyme). But unlike other systems, CRISPR pairs the enzyme scissors with pieces of RNA that guide it to the gene researchers want to cut. When the RNA finds a match on the information-carrying units of DNA, called bases, the Cas9 enzyme cleaves the DNA.
RNA is very easy to program. Researchers just need to select the DNA segment they want to cut and synthesize an RNA molecule that matches. The process takes days, as opposed to weeks or months for other technologies.
Researchers have wholeheartedly embraced CRISPR and used the technology to easily manipulate the genomes of many organisms in ways that would have taken years to achieve, if ever. “It’s not oh, maybe someday,” Doudna says. “It’s now.”
Fast forward
Harvard’s Esvelt was among the first to recognize that CRISPR is essentially an ultraflexible homing endonuclease that could easily be turned into a gene drive. He and colleagues laid out some of the possible uses for CRISPR gene drives in July 2014 in eLife.
At the time, no one had reported creating a gene drive using CRISPR. That soon changed. In January, Esvelt and colleagues reported online at bioRxiv.org that they had made a gene drive in yeast. In March, researchers from the University of California, San Diego reported online in Science that they had created a gene drive in fruit flies.
Those researchers, biologists Valentino Gantz and Ethan Bier, were looking for a way to easily make mutations in Drosophila fruit flies. Gantz focused on the yellow gene, which affects a fly’s color. He devised a piece of DNA carrying the gene that produces the Cas9 protein along with DNA that produces guide RNAs, which direct the drive to cut and plunk itself in the yellow gene, breaking it.
A broken yellow gene jaundices the flies, which are normally tan with dark stripes. The yellow gene is on the X chromosome. Female flies, which have two copies of the X chromosome, can inherit one copy of the broken yellow gene and retain their normal coloring. Two copies turns them golden. But males have only one X chromosome, and therefore just one copy of the gene, so any disruption will turn them yellow.
When an altered X chromosome is passed down to female offspring, Gantz reasoned, the gene drive should convert the normal X from the other parent into one with a broken yellow gene. Every female would be yellow.
“I figured this was beautiful on paper, but the odds of it really happening were pretty long,” Bier says. As it turns out, says Gantz, “it worked the first time.”
When the researchers bred female flies containing the yellow gene drive to normal males, 95 to 100 percent of both male and female progeny were yellow. If the drive hadn’t worked, and normal Mendelian inheritance rules were in effect, only 50 percent of male and no female offspring would be yellow.
The system wasn’t perfect. In 4 percent of cases, female flies were born with patches of normal cells and patches of yellow cells. One female was only half yellow. Clearly, some X chromosomes managed to outrun the drive. Still, the system worked with remarkable efficiency.
Gantz and Bier didn’t call their invention a gene drive. They named it the “mutagenic chain reaction.” By any name, it was the first time researchers had deployed a CRISPR gene drive in a multicelled organism.
Release and crash
If others can achieve the kind of efficiency that Gantz and Bier did, researchers could make a giant leap in wiping out insect-borne diseases. Theoretically, even one gene-drive–engineered organism could crash an entire population.
That possibility terrified some people when they learned about the yellow fruit fly experiment. If a gene-drive–containing organism were to escape from the lab and start breeding with its wild counterparts, it could irrevocably alter the wild population. Maybe even wipe it out.
In July, 27 scientists (Gantz and Bier among them) issued guidelines in Science for working with gene drives in the laboratory. The researchers want to keep their gene-drive experimental insects and other animals contained to protect wild populations, but also safeguard the potential humanitarian benefits of the technology.
“What’s it going to do to public trust if we accidentally release a gene drive into the wild?” Esvelt asks. He fears an accidental breach could damage malaria eradication and other much-needed public health measures.
The guidelines may help researchers avoid creating an accidental gene drive, but they don’t apply to gene drives that would actually be used in the wild, says molecular biologist Zach Adelman of Virginia Tech in Blacksburg. The whole purpose of a gene drive is to spread. Exactly how its spread will affect ecosystems isn’t known. Some people speculate that rapidly removing an invasive species could shock that system and have unknown costs. Even getting rid of disease-carrying mosquitoes might have consequences: Bats, birds and other critters that eat insects could lose a valuable food supply.
Scientists are also unclear whether gene drives could spread to closely related species. For Anopheles mosquitoes, many of which carry malaria, the answer could be yes, Besansky says.
Eight species known as the Anopheles gambiae complex of mosquitoes in Africa became separate species less than 5 million years ago, and they sometimes still interbreed, producing fertile hybrids. Gene drives might pass from one species to another through this interbreeding. But given that all but a couple of those species can carry malaria, spillover from one species into another might actually be desirable, Besansky says.
Still, many people are uncomfortable with the idea of gene drives that have the potential to eradicate entire species. Some researchers, including James of UC Irvine, prefer an approach that would prevent mosquitoes from spreading disease without reducing their numbers.
In 2012, James and colleagues reported that they had engineered Anopheles stephensi mosquitoes with genes that produce antibodies against malaria parasites. The antibodies prevented Plasmodium falciparum parasites from making sporozoites, the stage of the malaria life cycle that is infectious to humans. No sporozoites means the mosquitoes can’t pass the parasite on to humans.
With Gantz and Bier, James created a CRISPR gene drive to speed the spread of the antimalaria antibodies in mosquito populations. The team reported its work online November 23 in the Proceedings of the National Academy of Sciences (SN Online: 11/23/15). Getting the gene drive into the mosquitoes proved tricky; only two males out of more than 25,000 that were screened carried the drive. But once the drive was in the insects, males passed it to progeny with about 99 percent efficiency. Females, however, passed it to their offspring only slightly more often than Mendelian rules would suggest.
While this gene drive will not work in the wild because of the problems with female inheritance, James expects that gene-drive–carrying mosquitoes resistant to malaria will help form a front line against the disease. Any wild mosquitoes entering a zone made disease-free with a gene drive would quickly be assimilated.
Miles from reality
All of the benefits and drawbacks to gene drives are “just so hypothetical right now,” says Allison Snow, a plant population ecologist at Ohio State University. She doubts, for instance, the suggestion that weeds could be gene-drive engineered to eliminate herbicide resistance.
“These early predictions are rosy,” she says, and it is far too soon to say what will happen if such engineered weeds are ever released.
There is still time to work out the uncertainties, says Virginia Tech’s Adelman. “People are jumping the gun thinking these are going to be released any day now. It’s going to be years and years,” he says. Scientists have a number of technical hurdles to overcome.
One of the biggest barriers to making gene drives of any kind is getting them into the organism. That is harder in mosquitoes than it is in fruit flies or other lab animals.
Genetically engineering mosquitoes that will pass along a gene requires access to the organism’s eggs. At less than a millimeter long and a quarter of a millimeter wide, a mosquito egg doesn’t give researchers much room for error. Few labs have perfected the technique, James says.
When creating the antimalaria antibody gene drive, the researchers had to inject Cas9, guide RNAs and bits of DNA containing the gene drive into the egg. Cas9 appears to be toxic to mosquitoes, so the team also included a separate piece of RNA to dampen the amount of Cas9 produced. That reduced the toxicity of the enzyme, but also squelched initial insertion of the gene drive.
Ensuring the gene drive goes where it is supposed to is turning out to be tricky as well.
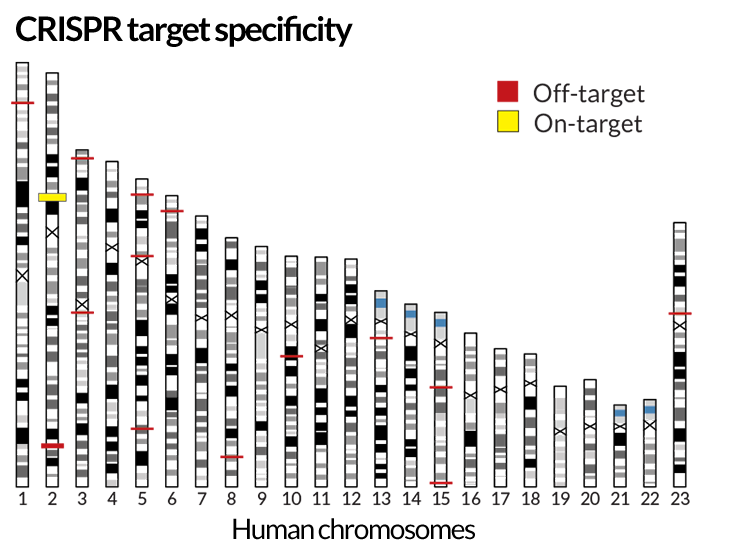
Just as some human guides give better tours than others, some guide RNAs are better than others at shepherding Cas9 to the proper spot. Guide RNAs targeting five different Aedes aegypti mosquito genes varied in efficiency from 24 to 90 percent, Adelman and colleagues reported in March in the Proceedings of the National Academy of Sciences.
Story continues after graph
In experiments with human cells, Shengdar Tsai of Massachusetts General Hospital and Harvard Medical School found that some guide RNAs nearly always lead Cas9 to the correct cutting site. Other guides take the enzyme to more than 150 “off-target” sites, Tsai and colleagues reported in the February Nature Biotechnology.
Another problem is that researchers know little about the biology of most disease-carrying critters, pests and invasive species, Burt says. That makes it hard to know which gene or genes to disrupt to sterilize or otherwise incapacitate a pest.
Even with these obstacles, CRISPR technology is moving so fast that human reaction times may not be enough to cork the bottle before the genie escapes. Scientists are scrambling to learn how to keep the genie under control, so that “make a wish” won’t turn into “be careful what you wish for.”
This article appears in the December 12, 2015, issue of Science News with the headline, “Gene drives unleashed: CRISPR brings a powerful genetic tool closer to reality.”