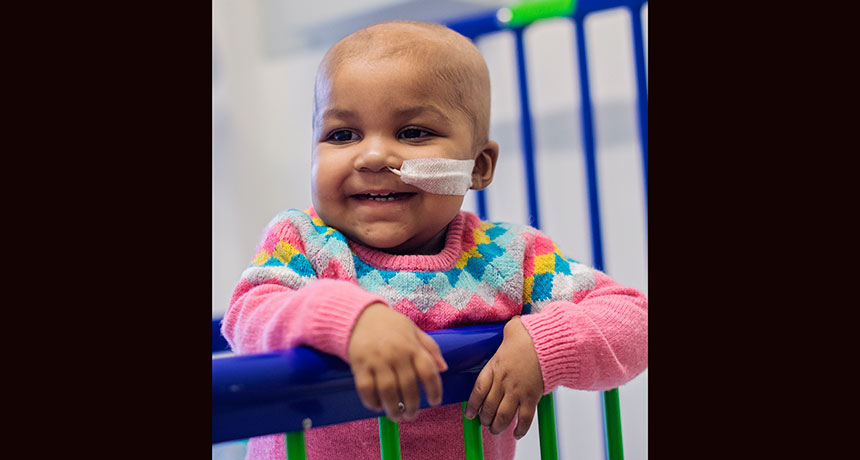
TEST CASE Layla, shown here at 16 months, had an experimental treatment for leukemia. She is the first person to receive a therapy involving gene editing with molecular scalpels called TALENs.
Sharon Leese/GOSH

TEST CASE Layla, shown here at 16 months, had an experimental treatment for leukemia. She is the first person to receive a therapy involving gene editing with molecular scalpels called TALENs.
Sharon Leese/GOSH