Hurt Blocker
The next big pain drug may soothe sensory firestorms without side effects
Michael Morgenstern
Among a small number of related families from northern Pakistan, some individuals never feel pain in any part of their bodies. Scientists studying six such children found that by the age of 4, they all had injuries to the lips or tongue from repeatedly biting themselves. Bruises, cuts and broken bones were common, though fractures were diagnosed only long after the fact, when weird, painless limping or the inability to use a limb called attention to the injury. Tests showed that the pain-free children perceived sensations of warm and cold, tickling and pressure. They could feel the prick of a needle, but it didn’t hurt. Two had been scalded — painlessly — by hot liquids. And one boy who performed street theater by putting knives through his arms and walking on hot coals died after jumping off a roof on his 14th birthday.
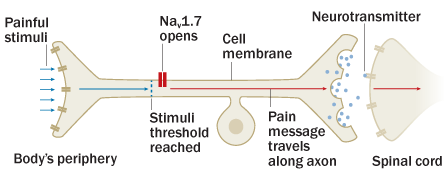
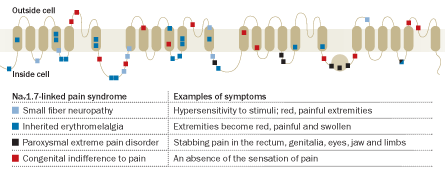
Besides their inability to feel pain, the Pakistani individuals studied by the scientists had something else in common: mutations in a gene called SCN9A. That gene encodes the instructions for a protein that forms a passageway for letting sodium ions into nerve cells. Known as Nav1.7, this particular ion channel sits on pain-sensing nerves; when a nerve is stimulated enough to warrant sending a signal to the brain, a flood of sodium ions rush into the cell.
Among the pain-free Pakistanis, various mutations in SCN9A altered the blueprints for Nav1.7 in different ways, but with the same result: The channel didn’t work. Muted nerve cells could no longer alert the brain when the body encountered something painful.
In other people, though, changes in the same gene make the channel work too well. Affected nerve cells distort or exaggerate their response, crying wolf when they encounter nonpainful stimuli or even when there is no stimulus at all. In January scientists reported seven Nav1.7-related mutations in some people with unexplained cases of small fiber neuropathy, a condition that typically entails burning pain in the feet with intermittent stabbing, aches and sensations of electric shocks or pins and needles.
Small fiber neuropathy and congenital indifference to pain (the official name for the Pakistanis’ pain-free condition) are just two of a handful of human pain disorders that have in the last decade been linked to malfunctioning Nav1.7. Though some of these conditions are relatively rare, a growing body of research suggests that Nav1.7 might play a part in more common persistent pain that follows nerve damage. Such pain can result from a bad burn or traumatic wound, or even accompany diseases such as diabetes.
As more new studies pile up, Nav1.7 is beginning to look like a nerve channel of pharmaceutical company dreams.
“Potentially, these channels are just the most amazing drug target,” says Geoff Woods of the Cambridge Institute for Medical Research in England, who led the study of the pain-free Pakistani children.
Channel Nº 1.7
Part of the excitement over Nav1.7 stems from the fact that the channel may offer a solution to an ongoing problem in designing pain drugs: To avoid unwanted side effects, medications should act only on particular targets in particular places. Today’s pain drugs aren’t so choosy.
For example, lidocaine, an anesthetic that can be applied topically or injected, targets an entire class of sodium channels. It doesn’t discriminate between channels on nerves related to pain and channels on other nerves; that’s why affected areas feel totally numb. (Because it blocks sodium channels in the heart, the drug is sometimes used to treat irregular heart rhythms.)
Aspirin, ibuprofen and their kin inactivate enzymes that promote inflammation. But these enzymes also help protect the stomach from acid, assist in kidney function and help blood clot, so the drugs can increase the risk of ulcers, kidney failure and certain cardiovascular troubles. The chronic pain drug pregabalin, which is prescribed under the brand name Lyrica for some types of pain and seizures, hits widely distributed calcium ion channels. Because of the effects on channels in the brain, the drug can cause dizziness, drowsiness and other problems.
Then there are opiates, including Vicodin, OxyContin and morphine. These work on cellular machinery in the digestive tract, spinal cord and brain, so they can cause nausea, constipation, dizziness and breathing problems, as well as being highly addictive.
But Nav1.7 is found predominantly in peripheral nerves at work in the outer territories of skin and muscle. So drugs that target this channel shouldn’t make people groggy, put them at risk for heart problems or meddle with other organs such as the liver or kidneys. The fact that the pain-free Pakistani children were otherwise healthy suggests that a Nav1.7 blocker wouldn’t interfere with other body functions. (The only secondary effect of knocking out Nav1.7 appears to be a reduced or lost sense of smell.)
Drugs aimed at Nav1.7 might provide relief, with few side effects, to people with the kind of pain that persists when peripheral nerves malfunction. Such pain is like an alarm system gone haywire.
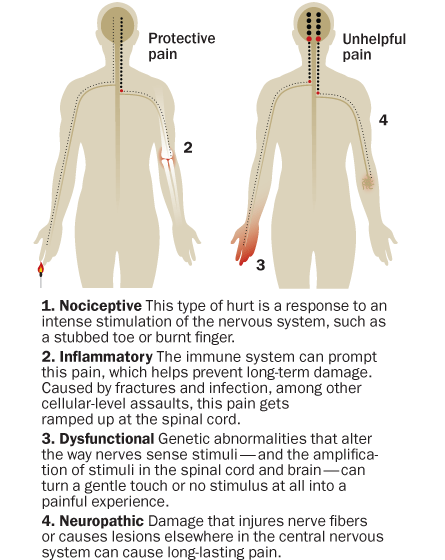
Typically pain is protective; it alerts you to impending or actual damage. Nociceptive pain (from the Latin nocere: to hurt or injure) delivers a red alert when you touch something dangerously sharp or hot. Nerve cells that sense this type of pain have a pretty high threshold, but once activated, the response is instantaneous: Your withdrawal reflex kicks in and you pull your hand away. Inflammatory pain, stimulated by immune system cells, occurs in response to injury. This pain warns you not to move a broken arm, giving the bone time to heal.
But when physical injury, metabolic disorders such as diabetes, autoimmune diseases or viral infections assault nerve cells, pain is no longer protective. Nerves can become trigger-happy, set off by something as mild as the touch of bedsheets. An estimated 20 million people in the United States experience this kind of pathological pain in their extremities.
“There are a lot of people suffering, and it is really terrible for patients,” says Catharina Faber of Maastricht University Medical Center in the Netherlands.
When examining a patient with such pain, Faber and her colleagues first look for an underlying condition that might have attacked the patient’s nerves. In many cases, there’s no obvious cause.
Research now suggests that genetic changes that alter Nav1.7 may be a culprit. The seven SCN9A mutations linked to small fiber neuropathy in a study earlier this year appeared to explain the peripheral pain of nearly 30 percent of study participants. This study — reported by Faber, Maastricht colleague Ingemar Merkies and other collaborators in the Annals of Neurology — and other recent studies suggest that much of how pain is experienced is inherited. Many scientists now believe that genetic changes altering Nav1.7, as well as other channels, may be behind a lot of chronic, pathological and mysterious pain.
Disorderly conduct
Stephen Waxman, who directs the Yale Center for Neuroscience and Regeneration Research and is a biomedical researcher with the U.S. Veterans Affairs health care system, has been investigating Nav1.7 and its defects for more than a decade. Working with Faber, Merkies and others from around the world, researchers in Waxman’s lab have identified more than two dozen mutations in SCN9A. One, reported in Brain in February, not only causes burning pain in the extremities, but may also influence development of the hands and feet. The many months of cell studies required to fully understand how each mutation influences Nav1.7’s construction and operation have been carried out for about half.
The first definitive link between mutations in SCN9A and a human pain condition came in 2004. Researchers led by Yong Yang of Peking University First Hospital in Beijing reported two mutations in a family with several members suffering from primary erythromelalgia, a condition wherein Nav1.7 channels open too readily and the body’s extremities become red, swollen and burn with pain.
Then in 2006, an international team linked mutations that alter Nav1.7 to paroxysmal extreme pain disorder. Formerly known as familial rectal pain, this lifelong condition is characterized by attacks of excruciating pain in various parts of the body, including the rectum, genitalia, eyes, jaw and limbs (the name was changed in part because the pain is not confined to the rectum). That same year, the team led by Woods reported the mutations causing an inability to feel pain in the families from northern Pakistan; additional mutations with the same effect have since been discovered worldwide.
These genetic studies cemented Nav1.7’s prominent role in human pain perception. They also offered an unusual treat: Rarely do geneticists find a tidy and direct link connecting changes in a single gene with what scientists call a loss of function and its counterpart, a gain of function. Often the picture is much more complicated, with many genes contributing to a trait or disease.
Yet SCN9A offered a textbook example: A single mutation in a single gene could cause a person to lose the ability to sense pain, while another mutation in that same gene could amp up that ability, making people feel pain even when they should not.
“The genetic demonstration was very clear. It was so clean. It’s usually not that clean,” says Simon Halegoua, director of the Center for Nervous System Disorders at Stony Brook University in New York. “That’s when multiple drug companies jumped on it.”
Halegoua saw Nav1.7’s potential earlier. In 1997, he and colleagues published research describing a sodium channel found predominantly in the peripheral nervous system. Since it was the first such sodium channel, the researchers called it peripheral nerve type 1. Later, it became known as Nav1.7 (Na for sodium and v for voltage-gated channel). Earlier studies in rats and human cells had hinted that some sodium channels favored certain body regions over others. Some did their stuff primarily in heart muscle, for example, and others acted mainly in the brain. But this was the first description of a sodium channel used mostly by peripheral nerves, at the front line of the body’s interactions with the world.
“That was the breakthrough,” says Halegoua. “It opened the door to an approach which would target those peripheral pain-sensing neurons specifically. It was a paradigm shift in the way of thinking about how to target pain. Instead of targeting it at the brain like opiates do, you would target it at the source — the first nerve that gets excited.”
Pain, pain, go away
The efforts of nearly two decades of research on Nav1.7 are now bearing fruit. London-based Convergence Pharmaceuticals has developed a compound that blocks the channel only when nerves are firing like crazy. Normal pain thresholds, such as those experienced when touching a hot stove, aren’t altered by the Nav1.7 blocker, says Simon Tate, Convergence’s chief scientific officer. The company’s compound is now in Phase II clinical testing — the stage that looks at dosage and effectiveness — for a painful condition of the lower back and limbs called lumbosacral radiculopathy. Convergence has also started a Phase II trial testing the same Nav1.7 blocker as a treatment for trigeminal neuralgia, a chronic pain condition that entails recurring brief episodes of intense, stabbing facial pain.
In January the Canadian company Xenon Pharmaceuticals reported success with an Nav1.7 blocker for treating erythromelalgia, often referred to as “man on fire syndrome.” And Pfizer and its subsidiary Icagen are working with Waxman and Yale colleague Sulayman Dib-Hajj to investigate a blocker for treating the same disorder.
Some conditions under investigation — lumbosacral radiculopathy and trigeminal neuralgia, for example — aren’t caused by inherited mutations, but appear to result from nerve damage. If an Nav1.7 blocker works in such cases, the target may prove useful for treating long-term debilitating pain more broadly. Good candidates may include the 60 to 70 percent of people with diabetes who have nervous system damage that can cause daily pain. People with traumatic injuries for which the healing time is long, such as a severe burn, may also benefit. Studies by Waxman and colleagues have found that mice without working Nav1.7 channels don’t develop the hypersensitivity to heat that typically follows a burn injury.
Beyond offering new tricks for treating pain when it strikes, studying the channel may also help doctors understand their patients’ personal pain profiles. Recent research led by Woods found that some versions of SCN9A in people without known pain disorders can confer increased sensitivity to pain. Such studies may help explain why one solider who has been shot through the arm experiences chronic post-trauma pain, while another soldier with the same wound might not. Or why some amputees have phantom limb pain and others don’t, says Waxman.
“It’s very exciting to us,” he says. “If you go into a room full of people who all look normal, 30 percent may have a lower threshold for pain and an increased likelihood of developing pain after nerve injury.”
As scientists continue to investigate the nuances of Nav1.7, some are turning to its channel brethren as well. Nav1.8 and Nav1.9, also sodium channels, are less understood than Nav1.7. But scientists do know that these channels also play a role in generating pain, and therefore may be promising targets too, says Michael Costigan, a specialist in the genetics of chronic pain at Boston Children’s Hospital. Changes in the behavior of Nav1.8 have been implicated in inflammatory pain, for instance.
Other pain-related channels being studied include transient receptor potential, or TRP, channels, which play an important role in detecting nociceptive pain, warm and cold, and the fire of wasabi and chili peppers. Some TRP blockers are under investigation for treating migraines, postsurgical pain and pain from nerve damage. And Costigan and colleagues recently reported that people with a particular version of a potassium channel gene are more likely to develop chronic pain after an injury.
The sheer number of ion channels now known to play some role in pain and the overlap of those roles highlight how important pain is for safety and survival — a fact not lost on the researchers who are trying to beat it.
There’s potential danger that comes with the promise of a superior pain drug. Total pain blockers with few side effects could be abused by athletes or others who want to ignore an injury, allowing them to do even more damage. Such drugs might also quiet warnings of a new and serious condition, such as an intestinal obstruction or a stroke. As with the Pakistani children, living pain-free might even result in severe trauma and early death.
“If we have a really effective block, it could be dangerous,” Halegoua says. “We need pain.”