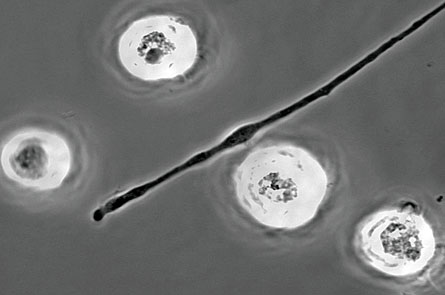
Immune cell plays good cop, bad cop
Depending on where a neuron is injured, immune cells can either help or harm
Talk about a mixed blessing. A pair of new studies shows that immune cells known as macrophages can do an injured neuron good, or can impart further harm.