New form of hydrogen created
Negatively charged clusters might exist in outer space
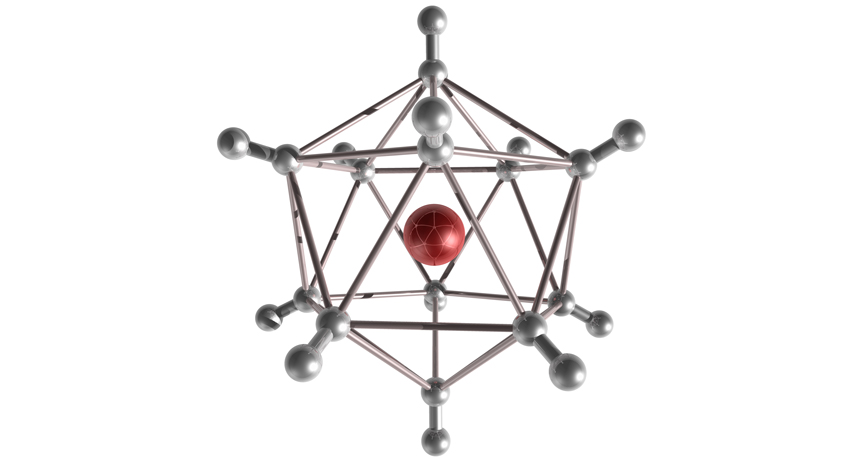
HYDROGEN HUDDLE Scientists produced negatively charged clusters of hydrogen in the lab. Hydrogen molecules made up of two hydrogen atoms (pairs of silver spheres) surround a negatively charged hydrogen ion (red sphere). The molecules arrange into an icosahedron, a shape with 12 vertices.
Andreas Mauracher