Predicting Parkinson’s
Researchers search for early warnings in the brain
By Carolyn Gramling

In the painting “Nude with Parkinson’s,” a woman kneels forward onto her elbows, bent under the weight of swirls of red, orange, and black. “[It] shows my struggle to maintain balance while being pummeled by the demands of everyday life,” says artist and Parkinson’s patient Carol McLeod. As her symptoms worsen, she adds, keeping up with those demands is a constant, and growing, battle.
This work was one of several hundred paintings, poems, and songs on exhibit last February at the first annual World Parkinson Congress in Washington, D.C. As participants examined the artwork produced as a therapeutic exercise by Parkinson’s patients, many in the most-debilitating stages of the disease, scientists spoke to attentive audiences of other scientists, medical professionals, patients, and caregivers. Much of the exciting research at the meeting focused not on people struggling with late, severe symptoms of Parkinson’s disease but on those in whom the illness had not yet become, or was just beginning to become, apparent.
The disease’s progressive, debilitating effects result from the slow but inexorable die-out of nerve cells in a region of the central brain called the substantia nigra. A variety of insults can trigger that cell death. The vulnerable cells produce dopamine, one of the neurotransmitters that manage the flow of signals from the brain to the body’s muscles.
As the brain cells die, there’s less dopamine available to transmit these signals, so Parkinson’s patients endure muscle tremors, difficulty balancing, slowed movement, dementia, and, eventually, paralysis. Because dopamine also regulates mood, patients short on that neurotransmitter may suffer from depression.
Currently, doctors use its characteristic motor symptoms to diagnose Parkinson’s. By the time those overt signs become apparent, however, a great deal of damage to the nerve cells has already been done, says David Eidelberg, a neurologist at New York University School of Medicine. Today’s therapies, primarily dopamine replacement, can slow that progress. Yet as the disease progresses, motor symptoms respond less well to dopamine-replacement treatment.
Medical scientists know what a Parkinson’s patient’s brain typically looks like both when symptoms first appear and as patients follow the largely predictable path of the disease. The challenge now is to characterize what happens before visible symptoms of the disease show up. “We need a reliable and objective measure” to find those signs, Eidelberg says. If physicians could detect Parkinson’s disease earlier, they might learn how to arrest damage before there’s permanent impairment.
For early diagnosis, scientists are looking for genetic signatures, brain-activity patterns, and blood cell characteristics that indicate the disease.
“Since Parkinson’s disease can be initiated by multiple triggers… there must be some kind of [common] pathway that defines the response to the injury,” says Howard Federoff of Albert Einstein College of Medicine in Rochester, N.Y. “If we understand that pathway, we might be able to identify new strategies for therapy.”
Genetic complexity
Researchers want to develop a test that measures a biological characteristic, or biomarker, that would indicate a propensity for the disease or would change in a specific way during the early Parkinson’s stages.
Markers that predict whether a person will develop Parkinson’s have proved elusive. The disease has a hereditary component, but the genetics of Parkinson’s is so far much murkier than that of Huntington’s disease, another neurodegenerative disorder.
“Genetic testing for Huntington’s [disease] is easy,” says neurologist Martha Nance of Struthers Parkinson’s Center in St. Louis Park, Minn. A person will acquire Huntington’s if he or she has a defect in a single, specific gene. This certainty is described as 100 percent penetrance.
The case is less straightforward for Parkinson’s. At least four different genes appear to be related to the onset of the disease, Nance says. So far, clinical tests suggest that a defect in any one of three of the genes doesn’t always result in Parkinson’s disease. The fourth gene has a penetrance of nearly 100 percent, but the disease may not show up until age 80.
Faults in the four genes appear to trigger nerve cell death in different ways. A defect in the PARK1 gene can cause the concentration of the protein alpha-synuclein to become so high that clumps deposit around and damage dopamine-producing cells. Defects in another gene, called DJ-1, prevent it from protecting dopamine cells against highly reactive oxygen molecules.
The tau gene, which has been linked to other neurodegenerative diseases, such as Alzheimer’s, has also been connected to late-onset Parkinson’s. And in 2004, scientists identified what appears to be the most-direct genetic link to the disease: the LRRK2 gene, which encodes for a protein that controls other proteins’ activities.
But genes aren’t the only triggers for the disease. At least one environmental contaminant, an industrial chemical called MPTP, has been directly linked to the onset of Parkinson’s, and researchers have found that some herbicides, pesticides, and heavy metals may cause symptoms similar to those of the disease.
Most people diagnosed with Parkinson’s say they would have wanted to know whether they had the disease well before symptoms appeared, according to a survey that Nance and her colleagues recently conducted at the Struthers Parkinson’s Center. Furthermore, a majority of relatives of patients said that they would choose to take a Parkinson’s test if one were available, the researchers reported at the World Parkinson Congress.
Visible losses
Like a time-lapse series of snapshots, repeated imaging of the substantia nigra can both track the disease’s progression and monitor the effects of treatment.
The primary tools of the trade are positron-emission tomography (PET) and single-photon–emission computed tomography (SPECT) scans, with which scientists can find and measure changes in dopamine and other neurochemicals in the brain. Imaging can also reveal the effect of Parkinson’s therapies such as levodopa, which replaces depleted dopamine, and deep-brain stimulation, in which doctors install in the brain a pacemakerlike device that sends electric pulses to target areas to prevent the signals that cause tremors and rigidity (SN: 3/12/05, p. 174: Available to subscribers at Inner-brain electrode may curb depression).
Many in the imaging field are now looking for markers that will indicate whether a patient has the disease before the characteristic motor symptoms appear, Eidelberg says.
In 2002, researchers in Ireland tracked dopamine production in the brains of people with minor muscle tremors but who hadn’t been diagnosed with Parkinson’s disease. After the participants received a gamma-ray–emitting compound that binds to dopamine-producing brain tissue, a computerized tomography scanner assessed dopamine production. With this method, the researchers found that only 5 of the 50 patients showed normal dopamine production. This trait proved to be a better predictor of whether the participants would later develop Parkinson’s than did the severity of their minor tremors, the researchers found (SN: 12/14/02, p. 382: Available to subscribers at Imaging Parkinson’s).
Eidelberg and his group are working to uncover even earlier signs of illness. Although dopamine has been the primary target for imaging, it may not be the most effective one, Eidelberg says.
“Dopamine is already two-thirds diminished by the time symptoms develop,” he says. As the brain’s dopamine concentrations continue to decrease, he adds, it becomes even harder to accurately measure changes in the neurotransmitter’s concentrations.
Scientists have known for a decade that loss of smell, excessive daytime sleepiness, and some behavioral disorders can foreshadow the motor symptoms of Parkinson’s disease. These early symptoms aren’t specific enough for a definitive diagnosis, but they hint that the disease may begin in regions of the brain other than the substantia nigra, Eidelberg says.
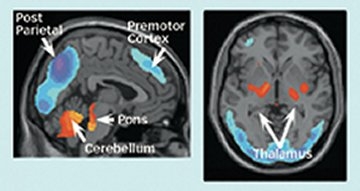
In 1994, Eidelberg visualized cellular metabolism by measuring fluoro-deoxy-glucose (FDG), which cells take up when they’re active. Parkinson’s patients showed an overactive pattern of metabolism in several brain regions related to motor function. The elevated activity occurred not just in the subcortical brain, which includes the substantia nigra, but also in the motor cortex, which issues neural commands to the body’s muscles, and the cerebellum, which regulates posture and balance.
Chengke Tang, who works with Eidelberg, recently used a radioactive isotope of oxygen to look at blood-flow patterns in the brain. Tang found a strong correlation between the altered flow in Parkinson’s patients and Eidelberg’s pattern of aberrant brain metabolism. Tang presented his findings at the February Parkinson’s meeting.
Parkinson’s disease produces changes in cognitive as well as motor areas of the brain, Dutch neuropathologist Heiko Braak and his colleagues reported in 2003. They noted the changes late in the course of the disease, when dementia often occurs.
Eidelberg’s team is looking for a Parkinson’s-specific pattern in cognitive areas of the brain in late-stage patients who are experiencing dementia. If a pattern can be established, Eidelberg says, a single PET image may serve to monitor both motor and nonmotor aspects of the disease.
It’s in the blood
Other scientists are looking at blood chemistry as a window on the brain. Federoff suggests that there may be a common biochemical signature that would indicate the presence of Parkinson’s, whether the disease arises from genetic or environmental factors.
“Our hypothesis is that the blood shares many cellular-signaling pathways with the nervous system,” Federoff says. If so, blood would be affected by many of the factors that influence the brain. “Some of the same biochemical dysfunctions in white blood cells may also be disordered within the brain,” he adds.
Federoff and his colleague Timothy Mhyre of the University of Rochester College of Medicine have been working with white blood cells to identify RNA and protein patterns that are associated with the onset and progression of Parkinson’s and Alzheimer’s diseases. The goal, Mhyre says, is to create profiles that distinguish Parkinson’s patients from people with other neurological disorders as well as from neurologically healthy people.
He and Federoff examined white blood cells from groups of Parkinson’s patients with cognitive impairment, Parkinson’s patients with normal cognitive function, Alzheimer’s patients, and people with no illness.
Molecular signatures in the cells were indeed distinct from group to group in preliminary results that Mhyre and Federoff presented in February at the World Parkinson Congress. Still, they warn, it’s a long step from identifying the signatures to predicting who will develop the disease.
No matter how early scientists can make a Parkinson’s diagnosis, physicians still will need an effective strategy to prevent nerve cell death. Mhyre says that if the patterns that he and his colleagues have discovered reveal the earliest steps in the sequence of events that leads to the loss of dopamine neurons, “we may be able to develop new targets for therapies.”
The biomarkers currently being developed to identify the disease and monitor its impact will make future treatments more effective, Eidelberg notes. “Opportunity favors the prepared mind,” he says.







