Reviving A Tired Heart
With a bit of encouragement, the life-giving muscle may renew itself
A generation ago, the battle to survive a heart attack was usually won or lost in the emergency room. Medical advances have now enabled more patients to win that fight and go home from the hospital — but millions of them will face another threat in the years to come.
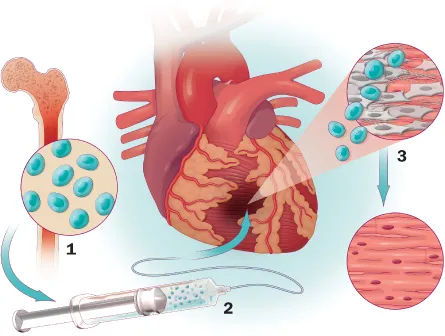
The heart has a monstrous appetite for fuel as it goes about pumping 2,500 gallons of blood a day. During a heart attack, when an artery feeding heart muscle gets choked off, the heart’s oxygen supply is interrupted. If starved of oxygen for too long, a portion of the heart can die, never to revive. Instead, lifeless muscle will be gradually replaced by an inflexible scar tissue not designed for pumping blood. To compensate, the remaining muscle pushes itself to work harder. Eventually, the heart can grow too stiff or too weak to efficiently eject the blood flowing into it, and a person lapses into heart failure.
Heart failure, a good portion of which is caused by heart attack, has become the leading reason older adults need hospital care. Efforts to improve treatments for heart failure have not yet led to a cure, and roughly half of patients diagnosed with the condition will not live five more years.
But recent experiments have lent support to a drastically new approach: awakening damaged heart muscle to regrow and beat like new. “We’re talking about transforming cardiology,” says Joshua Hare, director of the Interdisciplinary Stem Cell Institute at the University of Miami Miller School of Medicine. “This is the beginning. It’s like being at the start of antibiotics or vaccines.”
Already, hundreds of clinical trials are attempting to cultivate new muscle inside the walls of damaged hearts, even while laboratory researchers work to understand where new heart cells come from in the first place. The combination of eagerness, uncertainty and staggering clinical potential has turned heart regeneration into one of the most competitive and controversial fields of modern cardiac research. “It’s high-stakes, high-profile research in an area of great medical need,” says Richard Lee, head of the cardiovascular disease program at the Harvard Stem Cell Institute.
Keep the beat alive
Attempts to grow heart muscle occupy one section in the larger arena of stem cell research. A stem cell is like a computer with a blank hard drive waiting for software, capable of assuming any programming it gets. These cells are naturally abundant in tissues that have to frequently rebound from injury or normal wear and tear — places like the skin, liver and skeletal muscle, or bone marrow, where stem cells provide the wellspring for new blood cells.
Organs that don’t heal themselves, such as the brain and heart, are lacking in stem cells. The heart’s pumping tissue is populated by cardiomyocytes, adult cells that live and die but do not divide. Even athletes who condition their hearts to extremes have to work with the heart cells they’ve got. For the most part.
“For many years, it was thought that you’re born with a certain number of myocytes and you’re going to die with them, minus the ones you’ve lost along the way,” says Hesham Sadek, a cardiologist and stem cell biologist at the University of Texas Southwestern Medical Center at Dallas. New evidence, however, suggests that heart cells have some low-level churn — old ones expiring, new ones appearing. But the rate is too feeble for serious damage repair. It’s like a body shop that offers to fix only a few dents after a serious wreck.
Yet there is a time in life when a damaged heart grows back as easily as a tadpole sprouts a new limb. Sadek and colleagues demonstrated this fact in a simple but powerful experiment reported in February in the journal Science.
When mice were 1 day old, the researchers surgically removed the apex of the animals’ hearts. The hearts grew back, good as new. After repeating the test in different time frames, the researchers found that a mouse’s heart could regrow until the animal was about a week old — a point corresponding to the first few months following birth for humans — after which the damage was permanent. For a time, at least, the heart emerged unscathed from a seemingly devastating loss of muscle. Could an adult heart ever do the same?
“Now that we know that the mammalian heart can actually regenerate itself, the question becomes relatively easier,” Sadek says. “We can learn from the newborn heart.” He notes that his mouse research parallels the experience of doctors treating birth defects. Babies born with serious cardiac problems can recover completely if corrective surgery occurs shortly after birth. But if too many months pass before treatment, heart muscle is lost for good.
It’s not known why mammals would outgrow such a remarkable survival ability. (Some animals, including certain fish, enjoy a natural cardiovascular repair system for life.) Sadek speculates that during the course of evolution, before the human population lived long enough to grow old and have diseased arteries, a blow to the heart was usually fatal, so there wasn’t a need for a heart to heal. The challenge for scientists is to awaken a long-forgotten repair mechanism — whether by getting adult heart cells to return to their infancy or encouraging division and differentiation among a small population of existing heart stem cells.
Divide and renew
Most scientists (though there are notable exceptions) have come to accept the idea that the heart contains stem cells, though there is no agreement on how plentiful or important they may be. (Estimates for the turnover of new heart muscle range from less than 1 percent per year to as much as 20 percent.)
Among those believing that the heart is capable of self-healing is Lee, from Harvard. Using genes that give new cells a telltale fluorescent glow, Lee’s experiments suggest that after part of the muscle dies, the heart makes a weak attempt to mend. In mouse experiments that mimic the damage of a heart attack, more than 15 percent of the heart cells around the area of injury appear to be new, Lee and colleagues reported in 2007 in Nature Medicine. Later work supports that finding, he says.
“Our experiments suggest that mammals like humans have some repair capability,” he says. Recovery happens, but “it is woefully inadequate.”
Scientists don’t yet know which cells in the crippled heart are doing the dividing and why. Some researchers are tinkering with adult heart cell genetics, trying to find genes that turn a cardiomyocyte’s growth switch off or on. Studies suggest that adult heart cells often go through the preparations for dividing, making copies of their genetic material and (in mice at least) forming a new cell nucleus.
“It’s as if they try to re-enter the cell cycle,” says Jonathan Epstein, scientific director of the Penn Cardiovascular Institute at the University of Pennsylvania School of Medicine in Philadelphia. “They make new DNA; they just don’t divide.” It’s not uncommon for heart cells to develop not just one but several copies of their own genetic instructions, as if gearing up for the big event at several points in life. “The difference between a newborn heart and an adult may focus on the ability of cells to divide, and not be related to their ability to create new DNA,” he says.
Epstein and others are trying to identify the genes and proteins necessary for a cell to take the additional step of cleaving itself in two once its DNA replicates. For example, last year, in Developmental Cell, Epstein’s team reported that an enzyme called Hdac2 interacts with two other proteins in a heart cell, and together this trio of molecules impedes cell division. Hdac2 also appears to affect how genes are spooled together or packaged inside a cell, which controls their function.
Clinical trials are already under way for drugs that inhibit Hdac enzymes, though investigating cancer treatment rather than heart disease. Animal studies have suggested that taking these drugs improves cardiac function after a heart attack or in the presence of high blood pressure — but the explanation for this benefit remains unclear.
Clinical trials are also in progress to try to spur heart cell growth in people. For the most part, researchers are testing ways to give an ailing heart a dose of a patient’s own bone marrow stem cells with the hope that they will transform into heart muscle, or revive cardiac stem cells that might already lie inside. The field got a kick start in 2001, when scientists reported in Nature that they were able to regrow heart tissue in mice by coaxing injected bone marrow stem cells into taking on a new identity as heart cells.
“The net result was that a new field was born,” says Hare, from Miami. “Within a year there were papers where people had tried this in humans.” Today, hundreds of trials giving bone marrow cells to heart patients are either ongoing or already completed. Some provide stem cells shortly after a heart attack to spare heavy damage; some involve patients already in heart failure.
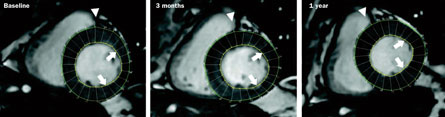
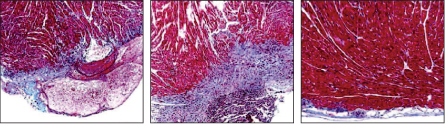
“The totality of evidence from the clinical trials is positive,” says Hare, who is conducting some of the trials and is on the board of Stem Cell Therapeutics, a Canadian-based company. “The heart is pumping more blood per beat.” In March in the journal Circulation Research, his team described a study of eight patients who received injections of bone marrow cells near an area of the heart scarred due to heart attack. After three months, the patients’ heart contractions were stronger. Even more important, their hearts later appeared to undergo some degree of “reverse remodeling,” or an attempt to return to normal anatomy, though the precise mechanism isn’t known.
Problem is, no one knows whether these kinds of improvements make any difference for a patient’s survival. “The definition of working means, ‘Do people live longer and feel better?’ That study has just been funded in Europe,” Hare says. A team of investigators coordinated by the European Society of Cardiology is aiming to recruit 3,000 volunteers, half of whom will receive bone marrow stem cell injections after a heart attack and half of whom will get a placebo. It will be one of the largest studies so far using stem cells to try to help heart patients, and will go on long enough to detect any survival advantage.
Heart of the matter
Cardiologists don’t agree about whether this clinical trial and others are a good idea. They also have concerns about the inconsistency of many laboratory studies.
With full understanding of cardiac regeneration still lacking, some researchers have cautioned against launching headfirst into human experiments. “I think it’s been reassuring and probably a little fortunate that we haven’t hurt people,” says Lee. Among the unanswered questions: Are the bone marrow stem cells turning into heart cells, or does the presence of bone marrow cells trigger some kind of dormant mechanism in stem cells already residing in the heart? Could other tissues be affected by the treatment?
“We don’t understand the mechanism enough,” Lee says.
For instance, he says, lab studies have suggested that the bone marrow stem cells do not survive well, if at all, once they are injected into the heart. If true, it means that the bone marrow cells are not becoming cardiac cells, but that some other property accounts for the study results. So patients might be receiving an injection of bone marrow cells that isn’t necessary.
A study published in September in Science Translational Medicine raises the possibility that a heart attack damages a patient’s bone marrow cells, giving them properties that make them less suited for transplantation. Other populations of stem cells might turn out to be more efficient; some scientists are working to create embryonic-like stem cells in the lab that may be even more open to a new identity than bone marrow stem cells.
“The closer we can get to understanding how these benefits occur,” Lee says, “the closer we can get to treatment.”
The scientists conducting the trials, such as Hare, believe human studies are necessary for progress, and can be done alongside vital basic research. “I would say it’s been way too slow,” he says of the pace of clinical trials. About 5 million Americans currently experience heart failure, left with options that will only slow the decline of their health but not offer a cure. He says he has no doubt that a deeper scientific appreciation will emerge with five more years of investigation. Some patients can’t wait that long. “I see patients with permanent heart damage,” he says. “Half of them are going to die while we sort out our scientific understanding.”
Even if scientists learn how to prod the growth of new heart cells, other challenges will remain. Growing muscle is not enough. The new tissue forming alongside the old still has to beat and carry electrical signals in perfect sync with neighboring cells. No one knows whether that would happen, or how to make it happen.
If scientists all agree on one point, it is this: After years of seeing progress in heart failure treatment move forward incrementally, the prospect of cardiac regeneration has energized the field, opening up a new frontier for patients who otherwise have had no chance for recovery. “It starts to get discouraging if you don’t have something on the horizon,” Lee says. After a decade of experiments, perhaps the one undisputed payoff is optimism.
What makes a weary heart |
Coronary artery disease and heart attack |
High blood pressure |
Faulty valves |
Damaged heart muscle |
Other causes |







