How a seahorse dad is like a pregnant woman

Big-belly seahorses always look pregnant. But when a male is carrying a brood, his body undergoes changes to genes that are similar to those in other animals that give live birth, a new study finds.
Joanne Merriam/Wikimedia Commons (CC-BY-SA 3.0)
Seahorses aren’t odd simply for their horselike shape. They (and their syngnathid fish kin, seadragons and pipefish) are the only vertebrates in which the males take on the burden of pregnancy. You might think that since having pregnant guys is really weird, some keen scientists would have figured everything out about the phenomenon. But a lot of questions remain, in part because it doesn’t look the same in all species. In some, the seahorse brood pouch — the male alternative to the uterus — serves as a simple site for fertilized eggs to attach while they develop. In others, the eggs, after mom deposits them in the brood pouch, are fully enclosed and protected from the outside world.
The big-belly seahorse (Hippocampus abdominalis) is at the far end of that spectrum, with the most complex type of seahorse pregnancy. To get a better picture of what is going on during this period of seahorse life, Camilla Whittington of the University of Sydney and colleagues analyzed gene expression (the patterns of genes turned on and off) in the big-belly seahorse during conception, egg development and birth. They found several commonalities linking the seahorse, reptiles that give live birth and us. The study appeared September 1 in Molecular Biology and Evolution.
A male seahorse becomes pregnant when a female transfers her eggs into his pouch, where they are fertilized. (It’s a clever system for males that ensures that they are taking care of their own kids.) The pouch has to undergo some restructuring, such as growing bigger, to accommodate and protect the eggs, and there are changes in genes that regulate cell growth, proliferation and death, Whittington and her colleagues found.
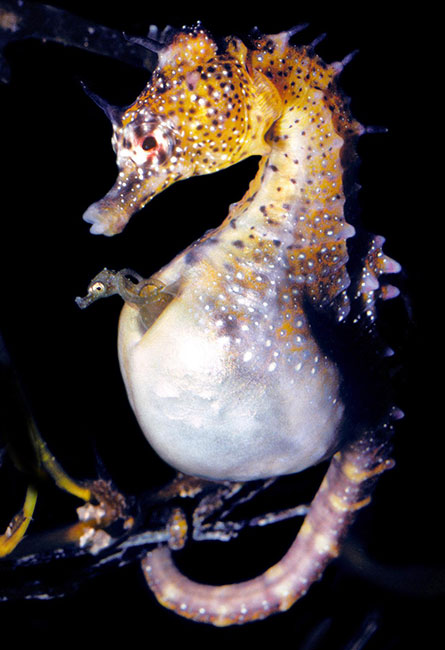
When a male seahorse isn’t pregnant, his brood pouch is open to the environment. But it seals up when the eggs arrive, creating a great place for bacteria or other pathogens to proliferate. Presumably to deal with this threat, changes occur in immune function during male pregnancy, with genes for bacterial resistance, pathogen recognition and antifungal activity turning up. And after the big day arrives and the kids are born, genes for tissue repair and remodeling (when tissues are reorganized or renovated) quickly turn on, letting dad get back his prepregnancy body fast enough to make any human mom jealous.
Live birth has evolved at least 150 times among vertebrates, and the gene activity changes in the big-belly seahorse are similar to ones in other species that give birth, including other fish, reptiles and mammals, the researchers note.
Last year, I visited Whittington in Sydney to talk about her work on platypus venom. She told me then that she was also studying the evolution of live birth. She was curious whether all of these animals that independently evolved live birth are using similar genetic pathways to get there. “Is there some set of genes that are particularly good for providing the functions of pregnancy?” she wondered.
Whittington and other scientists will need a lot more genetic data from species that become pregnant to determine just how similar we are. And only then will the researchers be able to tell the story of how this complex form of reproduction came about so many times in so many different critters.







