American and Dutch physicists reach new low temperature
Physicists compete in a race to the bottom with a finish line that can never be reached.
Read the full article (PDF) | Vote on future topic | Search archives
June 10, 1933 | Vol. 23 | No. 635
American and Dutch physicists reach new low temperature
The greatest cold produced and measured by man has now been pushed to within a quarter of a degree of absolute zero, that unattainable heatless point where all motion of the molecules cease and where gas would exert no pressure whatever.
Two groups of research workers, one at the University of California and the other in Holland, using novel methods identical in principle, have arrived at the extraordinary low temperatures of 0.25 degrees absolute and “certainly below 0.27 degrees absolute,” respectively.
The University of California scientists are Drs. W.F. Giauque and D.P. MacDougall, while the Dutch scientists are Prof. W.J. de Haas and E.C. Wiersma of Leyden and Prof. H.A. Kramers of Utrecht. The Americans did their work earlier and published first, and so they now hold the record.
Dr. Heike Kamerlingh Onnes, the pioneer in low temperature research who worked at Leyden, Holland, used the method of lowering temperature by reducing the vapor pressure of liquid helium. He reached a temperature of 0.82 degrees absolute and the same method was used by his successor Dr. W.H. Keesom of Leyden last year to attain 0.71 degrees.
The new low temperature records have been made by taking advantage of the fact that when a substance is magnetized, it heats up. Using liquid helium, made by cooling, liquefying, and solidifying of air, and then liquefying hydrogen to cool the helium, a substance is cooled as low as possible. Then it is magnetized. It heats up. Liquid helium is used to remove that heat. Then it is demagnetized, taking care to keep it heat-insulated. It becomes colder as a result of the demagnetization. Thus lower temperatures than ever before attained have been reached. Technically the method is referred to as “adiabatic demagnetization of paramagnetic salts.”
UPDATE | December 31, 2011
Physicists keep going lower
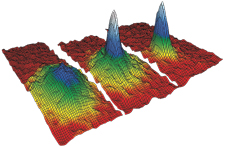 In 1995,
scientists observed a gas of rubidium atoms becoming a Bose-Einstein condensate (velocity-distribution data shown)
In 1995,
scientists observed a gas of rubidium atoms becoming a Bose-Einstein condensate (velocity-distribution data shown)
The third law of thermodynamics prohibits scientists from reaching absolute zero, but that doesn’t stop the competitive among them from trying to get as close as possible.
From the 1930s into the 1990s, Science News printed at least three dozen stories on the race to low temperatures, tracking new records as they fell from thousandths of a degree above absolute zero (millikelvins) to millionths (microkelvins) to billionths (nanokelvins).
A big breakthrough came in 1995, when researchers Eric Cornell and Carl Wieman cooled a gas of rubidium atoms to around 20 nanokelvins. As the temperature dropped, a new state of matter appeared. The atoms lost their individuality and began to behave as a superatom. Called a Bose-Einstein condensate, the exotic material had been predicted in the 1920s by Albert Einstein and Indian physicist Satyendra Nath Bose.
The 1995 discovery inspired a flurry of activity in the ultracold field. Scientists rushed in to explore the condensate’s bizarre properties and to gain insights into fundamental physical questions. But those efforts didn’t derail the race. If anything, more contestants emerged as scientists began cooling their own chosen stuff in an effort to uncover unusual phenomena.
In 2003, a team led by MIT’s Wolfgang Ketterle, who shared the 2001 Nobel Prize in physics with Cornell and Wieman for their ultracold efforts, created a Bose-Einstein condensate of sodium atoms at 450 picokelvins. This year in Physical Review Letters, Ketterle and colleagues report cooling what’s called a “spin mixture” to a record-low 50 picokelvins (one twenty-billionth of a degree above absolute zero).
And it’s not just atoms getting all the attention. Parallel races exist for molecules (SN: 12/20/08, p. 22) and for mechanical objects cooled with the touch of light (SN: 5/7/11, p. 24). —Elizabeth Quill
Credit: BEC: NIST, JILA, CU-Boulder