Another Side to Statins
Heart-healthy drugs show promise against inflammation, cancer and the flu
It’s been quite a ride for statins. Lipitor, Zocor, Crestor and their relatives are fast becoming household names. These cholesterol-lowering drugs have delivered a body blow to heart disease and stroke, two of the top four killers in Western society.
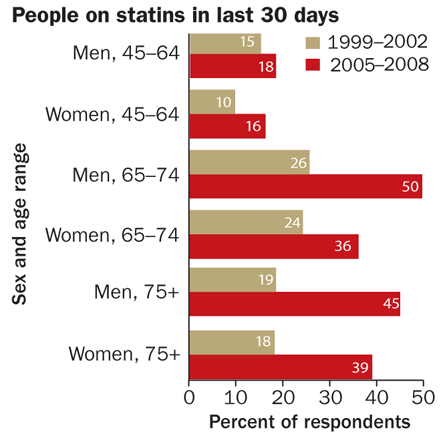
All success comes with a price: Some scientists express concerns over the widespread use and possible overprescription of statins. Still, there’s no denying the evidence that they have cardiovascular benefits in many people. The drugs’ track record has earned high praise for the research team that first devised them — and even higher rewards for the companies cranking them out.
Just when it seems that business can’t get any better, another upside to statins is now emerging. Research suggests that the drugs have potent effects against diseases they weren’t designed to fight. Statins might make people less susceptible to fatal pneumonia, better able to survive a head injury and more apt to dodge a nasty case of the flu.
No one is dancing in the streets just yet. These and many other findings arise from studies that track comparable populations but don’t randomly assign participants to one treatment or another. While useful for picking up trends, such population analyses can’t always be trusted, says Steven Lipkin, a physician and geneticist at Weill Cornell Medical College in New York City. These studies have been compared to economic models that have predicted “10 of the last four recessions,” he says. Evidence is indirect and can be misleading.
Recently, though, laboratory research by Lipkin and others is adding weight to population findings. Cell-based work shows that aside from slowing cholesterol production, statins derail compounds that exacerbate inflammation — an all-around pain in the neck, and elsewhere. What’s more, randomized trials in which people get statins or dummy pills, the gold standard for testing drugs, have shown a knockdown of rheumatoid arthritis and improvements in other immune-related ailments. Such findings are boosting enthusiasm among some researchers.
When statins first hit pharmacy shelves in the late 1980s, the tone was decidedly different. Many scientists worried that the drugs might cause cancer or have other unforgivable side effects. The discussion has come full circle now as scientists seek to sort out whether the drugs might treat cancer.
“It’s definitely heartening. It’s exciting,” says Carol Prives, a molecular biologist at Columbia University. “But it’s a large jump from [laboratory] data to any clinical changes.”
Tackling inflammation
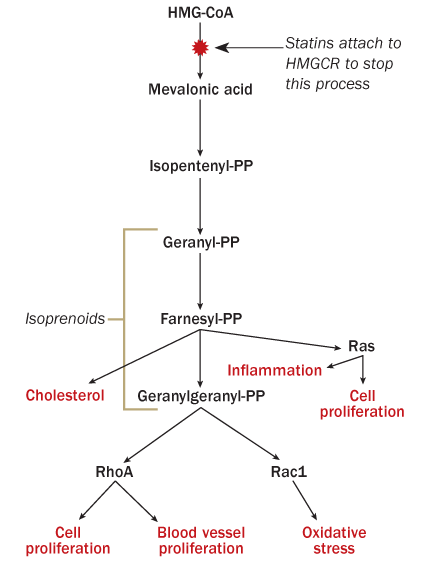
Statins have already made one giant leap, from the early days of drug development to today’s broadening interest. Japanese biochemist Akira Endo and his colleagues devised the first rudimentary statin in the 1970s, building on earlier work that had deciphered how the body makes cholesterol. His team screened thousands of fungal components to see if any of them bogged down cholesterol manufacture in cells. The painstaking effort unearthed a compound that reduced LDL — the “bad” form of cholesterol. The compound works by neutralizing an enzyme called HMGCR (for 3-hydroxy-3-methylglutaryl coenzyme-A reductase), halting the chain reaction that leads to cholesterol production inside cells. Scientists at Merck later devised their own version of this first statin, and the rest is Big Pharma history.
But over time it became clear that lowering cholesterol was only part of the story. Although some patients still got heart attacks, statins consistently overachieved: The drugs prevented more heart attacks than would be attributable to cholesterol alone. Paul Ridker, a cardiologist at the Harvard School of Public Health and Brigham and Women’s Hospital in Boston, thought statins’ success might speak to a central puzzle of heart disease: At least 15 to 20 percent of people who suffer a heart attack lack any apparent risk factor for the Big One, such as high cholesterol, hypertension, diabetes or a smoking habit.
Some researchers had already speculated that the hidden culprit might be chronic inflammation in the body, since plaques in the heart’s coronary arteries contain inflammatory immune cells. Meanwhile, lab work and studies looking at statins’ effects on the heart had suggested the drugs played a role against inflammation.
Ridker and colleagues decided to actually tackle the question: Did statins help prevent heart disease by reducing inflammation? His team assigned healthy people with normal LDL but elevated inflammation to get either Crestor or a placebo. The five-year trial was stopped early when it became clear that the statin recipients were having fewer heart attacks and strokes, the researchers reported in 2008 in the New England Journal of Medicine.
The following year, a second analysis of the data showed that roughly twice as many people getting placebos as receiving Crestor had experienced an episode of deep-vein thrombosis — a blood clot, often in the leg. Because clotting in veins is linked to inflammation and not plaque formation, the researchers concluded that statins’ anticlotting effects in this case were independent of its benefits against heart attacks.
“Statins are twofers,” Ridker says. “They both lower cholesterol and seem to inhibit inflammation.”
Suddenly earlier studies hinting at an anti-inflammatory role for statins took on a new gloss, as did work by scientists who, in relative obscurity, had been looking into statins’ effects on infections, trauma and other inflammation-related assaults on the body.
Multipronged switch
A steady stream of lab studies have since solidified the scientific basis for this inflammation-fighting effect. By attaching to HMGCR, statins flip a switch that does more than reduce cholesterol. The flip also knocks down production of compounds known as isoprenoids. Data now show that the knockdown influences basic immune function in a host of ways — a largely unexpected discovery.
“The whole history of science is filled with serendipity,” Lipkin says.
James Liao, a vascular biologist and cardiologist at Harvard and Brigham and Women’s Hospital, and others have tracked the fallout of statins’ other career in recent years. The researchers have found that inhibiting isoprenoids prevents some immune cells from secreting inflammatory proteins and hampers other cells’ ability to exacerbate existing inflammation. Statins even seem to tilt some immune cells away from an inflammatory bent toward a more quiescent role, and provide a bonus by maintaining good health in the all-important cells lining the insides of blood vessels. Such findings “have put into question whether cholesterol was entirely, or even mainly, the benefit of statin therapy,” Liao says.
Make no mistake, inflammation is a helpful response to infection or injury and has served humans well over time, particularly in the rough-and-tumble Stone Age. But inflammatory cells and proteins can overstay their welcome, damaging healthy tissues in blood vessels, swollen joints and infected lungs.
Statins’ additional role “opens up a whole new therapeutic window,” Liao says. There is plenty of work out there for an easy-to-take anti-inflammatory.
Randomized trials have shown that statins may ease rheumatoid arthritis and tame inflammation in a lethal condition called sepsis, a whole-body immune reaction to bacterial infection. Statins may also treat osteoarthritis, researchers reported in 2010: Statins prevent cartilage loss in mice and decrease enzymes known to abet inflammation and degrade cartilage in human cells. And then there’s trauma. Anti-inflammatory effects are consistent with a 2011 study finding that older people taking statins were much less likely than nonusers to die in the hospital following a head injury.
Liao says statins’ immune benefits go beyond fighting inflammation. The drugs don’t appear to wipe out inflammation wholesale, as a steroid drug might, he says, but instead bring overwrought inflammation signalers “back to a normal state.”
Some scientists even argue that the protection offered could extend to the inflammatory onslaught brought on by infections such as influenza. Severe flu strikes 35 million people annually. Patients already taking a statin are less likely to die of the disease than are nonusers, researchers at state health departments across the United States reported in January. Danish researchers tracking patients with pneumonia, a flu complication, reported that those on statins are less likely to die of the condition.
Despite this evidence, applying statins in the clinic could be tricky, says Edward Walsh, an infectious-disease physician at the University of Rochester in New York. “It’s conceivable that you need to be on statins ahead of time, and I wouldn’t advise anybody to take statins just because they might get the flu,” he says. “But on the other hand, if we were faced with a really deadly flu pandemic and — good gosh — we didn’t have a vaccine that worked? You could easily see how people might turn to anything that has potential.”
David Fedson, a retired physician and flu researcher, has made such concerns his scientific mission. He argues that documented cases of bird flu jumping to humans in Southeast Asia make a deadly flu pandemic a plausible threat. Population studies and biological findings from the last several years suggest that statins could be lifesavers in such a scenario by blunting the flu’s impact on the body. Annual stockpiles of flu vaccine and antiflu drugs are nowhere near adequate to stop a 1918-level flu, particularly in poor countries, says Fedson, who formerly worked at the University of Virginia. The annual version of the flu vaccine might not even work against such a novel virus. “We need something else,” he says.
Statins are widely available, easy to store and simple to take. Drug manufacturers in India already make a generic version of Zocor and ship it widely to developing countries at as little as 10 to 15 cents a dose, Fedson says. With appropriate planning and testing, he says, “on the first pandemic day, everyone in the world could be treated. What this means in terms of global equity is stunning. From Hyderabad to Houston, you give everyone an equal chance to live.”
Not all the same
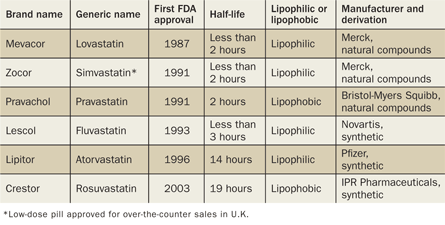
Whether statins will ascend to such wider use — and if so, which versions will work best — is still an open question. They are not all uniform. Some statins, such as Zocor, don’t linger in the blood for long, Liao says. That’s why doctors tell people to take them at night, when the liver actively makes cholesterol.
A short half-life might limit a statin’s effectiveness against diseases not related solely to cholesterol. “If inflammation is happening around the clock,” Liao says, “I’d like to have a statin that works 24 hours a day.” Other statins are released over a longer time frame.
Zocor, though, might have an advantage in fighting disease more broadly, because it can gain entry into most cells. Zocor, Lipitor, Lescol and Mevacor are all lipophilic — they “like” fats — meaning they dissolve easily into most any cell’s membrane, passing through it like a ghost through a wall. On the other end of the spectrum are Crestor and Pravachol. These lipophobic (“fat-fearing”) statins are reluctant to dissolve into cell membranes. Makers of these drugs have by design equipped them to enter into liver cells preferentially.
A stark contrast emerges between lipophilic and lipophobic statins in studies of another menace that statins are now showing promise against: cancer.
At first blush, statins would seem to have little to offer against cancer. Yet many population studies have suggested that taking some statins can lessen a person’s cancer risk. In an example reported in 2011, researchers tracked the fate of nearly every breast cancer patient diagnosed in Denmark over an eight-year span. Those taking simvastatin, the generic name for lipophilic Zocor, had 10 percent fewer cancer recurrences during a decade or so of follow-up than women not getting a statin. Women taking lipophobic statins didn’t fare any better than nonusers. Similarly, lab tests have shown that Zocor and Mevacor readily kill ovarian, uterine and cervical cancer cells in lab dishes, but lipophobic Pravachol does not.
Recent research provides a biological explanation for how statins, if they can get into a cell, might fight certain cancers. Statins appear to sabotage procancerous machinery within the cell by neutralizing the effect of a cancer double-agent called p53, Prives and colleague William Freed-Pastor reported in the Jan. 20 Cell.
The normally helpful p53 protein undergoes a Jekyll-to-Hyde transformation in roughly half of all cancers, including many aggressive ones. In breast tumors, mutant p53 ceases to check aberrant cell growth, instead frequently switching on 11 genes that encode proteins in the very chain reaction by which cholesterol and isoprenoids are produced. Prives and Freed-Pastor showed in cell-based experiments that adding a statin substantially impedes this disaster, limiting the ability of mutant p53 to switch on these genes. When the researchers treated breast cancer cells with Zocor in a lab, the cells died or showed slower growth.
There are other biological arguments in the case for statins against cancer. By thwarting isoprenoid production, Lipkin says, statins also disable a process that triggers activation of a proliferation molecule called Ras, which has been implicated in several cancers. Other evidence suggests that starving a tumor cell of cholesterol might slow runaway cell growth because cells use cholesterol as a building block for their fatty membranes.
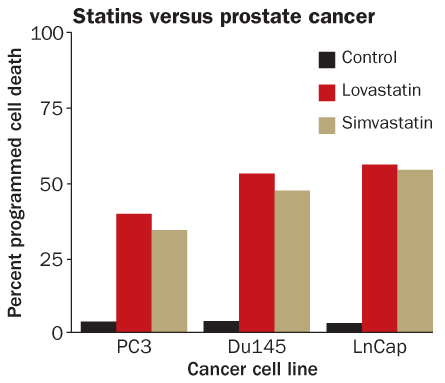
Several studies have linked high cholesterol with prostate cancer risk, and there is molecular evidence that bottling up HMGCR thwarts this cancer. A 2008 study showed that prostate cancer cell lines exposed to statins are more apt to undergo stalled growth or programmed cell suicide than unexposed cells. “The prostate cancer data actually look pretty strong,” Lipkin says.
Men who underwent prostate removal for cancer were less likely to have a recurrence if they were on statins beforehand than men not getting the drugs, Johns Hopkins University researchers reported last year. While this population finding doesn’t prove cause and effect, “what we’re seeing does make sense in the biological data. It’s nice that it hangs together,” says Alison Mondul, an epidemiologist at the National Cancer Institute who coauthored the study while at Johns Hopkins.
Some say it ain’t so
Inflammation and cancer aren’t the only conditions beyond the heart that statins might address. Recent studies suggest that statins might counteract dementia, cataracts, multiple sclerosis, Parkinson’s and depression. A National Institutes of Health website lists hundreds of clinical trials — finished, ongoing or still recruiting participants — that focus on statins’ impact outside of heart disease or stroke. But some scientists remain unconvinced that statins hold more wonder drug potential.
Beatrice Golomb, a physician and neurobiologist at the University of California, San Diego, notes that studies have found some pretty bad side effects associated with statins, including muscle pain and liver damage, as well as perhaps memory loss and diabetes. Such side effects, combined with the widespread prescribing of such drugs, have led to a backlash against statins among some scientists.
Golomb says population studies aren’t nearly adequate to outweigh these doubts. “This is a class of studies that should not be used for causal inferences of a drug and its outcomes,” she says, citing the “healthy-user effect.” People who obtain statins might have better insurance, more education and better access to health care than people who don’t get the drugs, skewing the outcomes.
But physician and epidemiologist Reimar Thomsen of Aarhus University in Denmark says that while the United States does have wide disparities in health coverage, Denmark doesn’t. His population study of statins and pneumonia included people covered by the same health system. “Everyone goes to the hospital for free — funded by taxes,” he says.
Factors such as the variation in statins’ ability to permeate cell membranes might explain some of the mixed results seen in recent studies. What’s more, some patients might be hardwired to benefit more from statins than others, Lipkin says. He cites a 2005 population study that found that colorectal cancer appears to be vulnerable to statins in some people but not all.
Intrigued by that finding, Lipkin and his colleagues tested more than 4,000 people, half with colorectal cancer, looking for alterations in 40 genes known to play a role in cholesterol synthesis and metabolism in cells. The analysis showed that one-fifth of the people made a slightly altered form of the HMGCR enzyme, a form that statins didn’t latch on to effectively.
“People with this variant don’t respond well to statins,” Lipkin says. These patients got poorer cholesterol-lowering effects and were less likely to share in statins’ protection against cancer.
Pinning down these and other vagaries of statins’ effects might clarify who would benefit from the drugs, and in what ways. In one trial, researchers are now randomly assigning patients who have undergone colorectal cancer surgery to get statins or a placebo along with other medications. The researchers will see which patients develop precancerous growths, or cancer itself. But results from this and other trials could be a long time coming.
Though some doctors still balk at the counterintuitive nature of giving a statin to someone with normal cholesterol, Thomsen believes the general thinking on statins is changing, if gradually.
“Five years ago, in an intensive care unit, doctors would stop statins, thinking there were other things to worry about,” he says. “Now, I think the attitude is that you should keep a heavily inflamed patient on statins in the ICU. It might be dangerous to stop them.”