Cancer’s little helpers
Tiny pieces of RNA may turn cells to the dark side
When tiny hairpin-shaped molecules act up, they don’t rebel loner-style like James Dean. Instead they take on the persona of Darth Vader, crushing proteins under their command and turning acquaintances to the dark side as well. In this case, though, the fight is for control not of the universe, but of the body. And a dark-side victory could end in cancer.
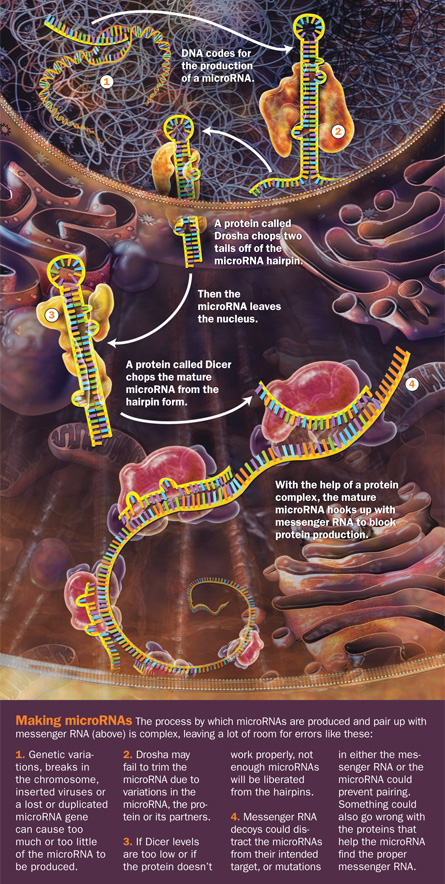
No one would have predicted a decade ago that these microRNAs, as the hairpins are called, were involved in cancer, because no one even knew that they existed in people. Mere snippets of RNA — DNA’s underappreciated cousin — these micromolecules are about 22 chemical letters long. But their size belies their power.
When on their best behavior, the molecules are competent and capable managers of the protein-building process that keeps a cell humming in perfect harmony. But when microRNAs go rogue, the results can be disastrous.
New research is revealing just how important these newly discovered molecules are. An imbalance of microRNAs can cause cancer by encouraging runaway cell growth or by dampening a cell’s defenses, and can also make the disease more stubborn. But just as Darth Vader never completely lost the young Jedi Anakin Skywalker within him, even bad microRNAs may have good in them yet. Some scientists think that therapies aimed at soothing riled-up microRNAs may help cure the very cancers that the molecules help cause.
Most of the discoveries linking microRNAs and cancer have come in the past five years. “This is extremely rapid progression,” says Curtis Harris, chief of the human carcinogenesis lab at the National Cancer Institute, based in Bethesda, Md.
Micro middle managers
The realization that such small molecules could play a big role in disease was late in coming, says Carlo Croce of Ohio State University in Columbus. “In the beginning there was no interest in microRNAs at all,” he says.
The first microRNA was discovered in 1993 in roundworms. It took another seven years before the next microRNA was found in the same organism. Though both of those microRNAs help control worm development, most scientists regarded them as biological curiosities.
But then researchers found microRNAs at work in fruit flies, people and other organisms. Those discoveries suggested that microRNAs might be important regulatory molecules for all animals, not just flukes of worm biology.
MicroRNAs work in middle management in most plant and animal cells, scientists now know. The molecules help regulate the protein-manufacturing process by essentially issuing permits decreeing when and where proteins may be built. By riding piggyback on messenger RNAs, which are copies of the protein-building blueprints contained within DNA, microRNAs prevent the instructions from reaching protein-building machinery inside cells.
While it may sound nefarious, microRNAs’ interference with protein production helps a cell maintain balance. MicroRNAs ensure that cells save energy by not making unnecessary proteins and help prevent levels of potentially harmful proteins, such as those that initiate the self-destruct program known as apoptosis, from reaching critical mass.
Each type of microRNA in a cell may potentially pair with hundreds of different types of messenger RNA, says Isidore Rigoutsos, a computational and molecular biologist at Thomas Jefferson University in Philadelphia. And each messenger RNA may have many different microRNAs piling on its back.
“It’s safe to say that microRNAs are important,” Rigoutsos says. “The difficulty is saying what are the limits of importance, and they keep being expanded more and more and more.”
Cancer connection
Croce’s lab was among the first to illustrate just how big a role the little molecules could play in people. His group showed that genes encoding microRNAs frequently go missing in tumor cells. In particular, two microRNAs, miR-15 and miR-16, are missing or found at lower than normal levels in 68 percent of chronic lymphocytic leukemia cases.
Cancer biologists usually lump microRNAs into two groups: those that protect against cancer and those that promote it (though the distinction isn’t perfect). Cancer cells tend to have lower levels of most microRNAs but have an oversupply of a few others.
In the protective corner are microRNAs such as miR-15 and miR-16. One of the many proteins regulated by those two microRNAs is BCL2, which keeps cells from pushing the self-destruct button. Cells commit suicide when they become too damaged to operate properly — an important self-defense mechanism for an organism that doesn’t want to walk around with malfunctioning cells. So cells need just the right amount of BCL2 to keep from killing themselves unnecessarily, but not so much of it that they can never die.
The microRNAs pair with messenger RNA to strike the right balance of BCL2. But when miR-15 and miR-16 levels are knocked down — which can happen if a copy of a gene is lost or if something goes wrong during microRNA manufacturing — cells make far too much BCL2, essentially disabling the self-destruct mechanism and making cells immortal. Immortality is one hallmark of cancer.
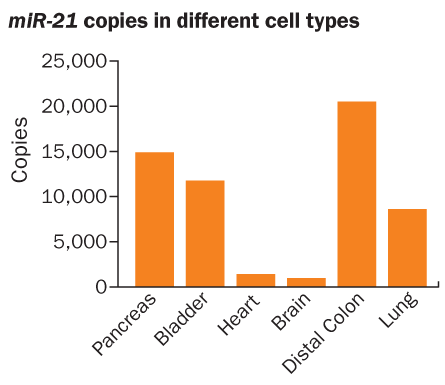
At the opposite end of the spectrum is one of the baddest microRNA bad boys, miR-21. Elevated levels cause cancer in mice, researchers from Yale University reported online August 8 in Nature. And higher than normal levels have been linked to at least 13 major types of cancer in people and to poor prognoses for people with colon, lung, breast, pancreatic or head and neck cancers, Harris says (SN: 2/2/08, p. 70).
High levels of miR-21 can slow an important cellular security system involving a protein named p53, researchers from the University of California, Santa Barbara have found. This protein performs multiple protective services, including spurring repair of damaged DNA, halting growth until damage is repaired or ending it all if repair isn’t possible (SN: 12/6/08, p. 22). Last year researchers reported in Nature that p53 helps slice microRNAs into their mature form. Too much miR-21 can strip cells of their p53 defenses, leading to cancer.
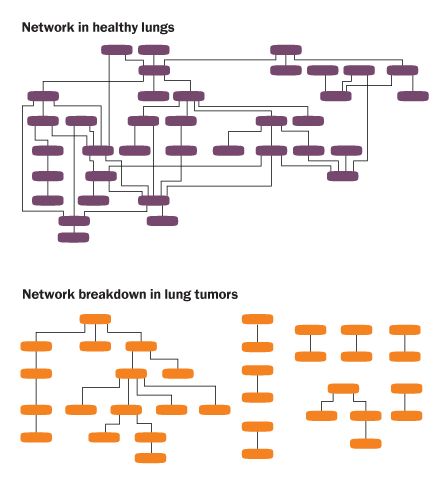
Though miR-21 has stood out among the troublemakers, Croce’s team has shown that this microRNA and others don’t work alone. The molecular managers are master networkers. In 50 different normal human tissues, microRNAs collaborate to direct cellular activities, Croce and colleagues reported online May 3 in Genome Research. The networks consist of microRNAs that help direct production of proteins, some of which, in turn, control production of other microRNAs, and so on.
But time and again, in 51 different types of cancer, Croce’s team found that the microRNAs’ teamwork had broken down. The cohesive networks disintegrated into rogue hubs of activity. These anarchist factions throw a wrench into the well-oiled machinery that usually keeps a cell healthy.
It’s a rather small wrench, though. MicroRNAs wield their power subtly, tweaking and massaging protein levels up or down a wee bit here and there instead of stopping production altogether.
“A microRNA doesn’t function like an ‘off’ switch,” says cancer biologist Dihua Yu of the University of Texas MD Anderson Cancer Center in Houston.
Even a little bump or dip in protein levels, maybe by just 5 to 10 percent, is enough to send a cell careening down the path to cancer, Croce says.
One of the most delicately balanced cancer-associated proteins is PTEN. It reins in cell growth to prevent wild replication, as seen in cancer. In the parlance of cancer research, PTEN is known as a tumor suppressor, and it works best when there is just the right amount of it.
Losing one copy of the gene for PTEN — essentially cutting protein levels in half — is enough to turn a cell cancerous, previous studies have shown. Other research has demonstrated that microRNAs, including miR-21, help govern production of PTEN. And a study reported June 24 in Nature found that a messenger RNA doppelgänger of PTEN found in healthy cells distracts PTEN-stifling microRNAs, allowing more of the protein to be made. If the twin is missing, the weight of microRNAs on messenger RNA’s back can crush protein production.
New research from Yu’s lab also suggests that reducing the amount of PTEN protein in a tumor cell even slightly is not a good idea. Higher levels of miR-21 slow down PTEN production and make breast cancer cells resistant to an anticancer agent called Herceptin, Yu and colleague Sumaiyah Rehman reported in April in Washington, D.C., at the annual meeting of the American Association for Cancer Research.
Dicer danger
PTEN isn’t the only protein that can make cancer worse. A new study shows that small changes in the amount of a protein involved in producing microRNAs can determine whether tumors stay put or spread to the rest of the body.
That discovery grew from efforts to figure out why most microRNAs are at lower levels in cancer cells but some microRNAs are overproduced. “We were intrigued by this paradox,” says Stefano Piccolo, a cellular and molecular biologist at the University of Padua in Italy.
The resolution came from an unexpected source, a protein that helps slice larger RNAs into microRNAs. This protein, Dicer, is a key component of the microRNA manufacturing machinery. Cutting levels of Dicer in half spurs on cancer because less of it leads to less microRNA, which can mean increased production of proteins that drive rapid growth. Still, cancer cells need some Dicer to survive and reproduce, since Dicer helps make microRNAs that regulate production of proteins.
So cancer cells need to control Dicer levels the way a student sets the volume on an iPod to provide background study music. The volume shouldn’t be too quiet or too loud. “You need to find that perfect middle,” Piccolo says.
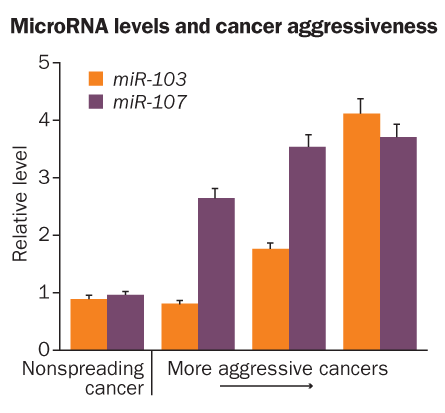
Cells dial in just the right amount of Dicer by using a family of microRNAs, miR-103.1, miR-103.2 and miR-107, Piccolo and his colleagues reported in the June 25 Cell. Those three microRNAs, which occur at high levels in some cancer cells, latch on to messenger RNAs encoding Dicer and ratchet down its production, meaning less of other microRNAs get made. But Dicer levels never drop to nothing, because the same microRNAs rely on the protein to snip them free from larger pieces of RNA.
Piccolo’s finding neatly solves the paradox of why most microRNA levels can be low in cancer cells while some are high, but his study didn’t stop there. The research also provides further evidence that low Dicer levels make cancer more dangerous.
In the study, Piccolo’s team discovered that some aggressive tumors had higher than normal levels of the microRNAs that regulate Dicer, and thus less of the protein. More of these microRNAs were also associated with breast cancer’s spread and poor prognosis in patients.
Additional experiments with tumor cells growing in lab dishes showed that the cells usually tend to cluster. But when Dicer levels are lowered to about 50 to 60 percent of normal, or levels of miR-107 are increased, cells begin migrating across the dish. Dips in Dicer levels make cells mobile, the team suggests. Less Dicer may mean less of other microRNAs that hold back production of proteins that are responsible for getting cells in gear. With fewer inhibitory microRNAs around, go-proteins can be made and cells get a move on.
Tumor cells may be taking advantage of one of Dicer’s jobs in normal cells — helping cells move around. “Cancer doesn’t invent anything,” Piccolo says.
Tiny treatment options
Getting a clue that a microRNA is involved in a problem also gives researchers a potential solution. In experiments with mice, inhibiting miR-21 made resistant tumor cells more susceptible to Herceptin, Rehman reported at the cancer research meeting.
Increasing susceptibility to anticancer drugs is just one way that microRNAs could be useful in the clinic, says oncologist Muller Fabbri of Ohio State.
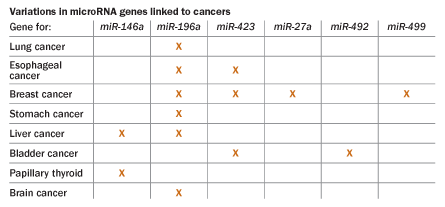
Specific microRNA levels rise or fall in different tumor types, creating a signature for that type of cancer, studies have shown. Such signatures could help correctly diagnose cancer in people whose tumors have migrated. Often pathologists can examine a brain tumor and determine that it arose from breast cancer cells, but sometimes cancer cells conceal their real birthplace. Characteristic patterns of microRNA could help identify where tumor cells originated in the 8 to 10 percent of cases when “even the pathologist doesn’t have a clue,” Fabbri says. Examining the pattern of microRNA levels in a patient’s tumor may also help doctors identify aggressive forms (SN: 2/2/08, p. 70).
In diseases such as liver cancer, researchers may be able to replace missing microRNAs or boost levels to stop the cancer, a team reported last year in Cell. And in cancers in which levels of certain microRNAs are too high, researchers can deploy decoy molecules to pull microRNAs from their targets. A team reported in January in Science that the strategy appears to work for treating hepatitis C infection in monkeys (SN: 1/2/10, p. 14).
Croce thinks that targeting several microRNAs in anarchist networks may help treat cancer with little chance of resistance developing. But such therapies are still years away. “We have to show that it is really true,” he says, “not just in experiments with mice, but in clinical trials.”
For now, no microRNA therapies are available for cancer, but researchers are watching trials of the anti-microRNA therapy against hepatitis C in people.
“The rapidity of what’s going on is what gives some of us optimism that this could have value,” Harris says. “Relatively shortly, we’re going to know the degree of importance of microRNAs.”