Convenient hydrogen storage?
Hydrogen attracts attention as a clean-burning fuel that could eliminate the United States’ dependence on fossil fuel. Finding a way of storing the gas safely and compactly in cars has been proving difficult, however.
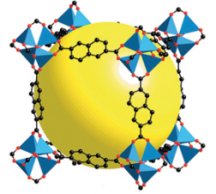
A team of researchers says it’s developed hydrogen-storage materials that, with modifications, might do the trick. In previous work, the scientists reported that varieties of their porous materials can hold large amounts of methane (SN: 6/23/01, p. 398: Perfecting Porosity). They’ve now shown that the materials, which are composed of microscopic frameworks of metal such as zinc and organic parts such as naphthalene, attract and sequester hydrogen.
The Department of Energy has specified that a good storage material should hold at least 6 percent of its weight as hydrogen. So far, however, no practical storage material has met this goal. Part of the problem is that storing the gas requires significantly compressing hydrogen at low temperatures and dangerously high pressures. In some cases, the contained hydrogen chemically bonds to the storage materials and doesn’t come out easily.
In comparison, the porous materials that Omar Yaghi of the University of Michigan in Ann Arbor and his colleagues describe in the May 16 Science can store and easily release 2 percent of their weight in hydrogen at room temperature and only 10 times atmospheric pressure. That’s the pressure found in a cigarette lighter, says Yaghi. What’s more, the work revealed a potential path to the Department of Energy’s 6 percent goal: increasing the size of the organic components in the storage framework.
****************
If you have a comment on this article that you would like considered for publication in Science News, send it to editors@sciencenews.org. Please include your name and location.







