The antidepressant fluvoxamine can keep COVID-19 patients out of the hospital
A 10-day course may work as an easy at-home treatment for early COVID-19, a clinical trial finds
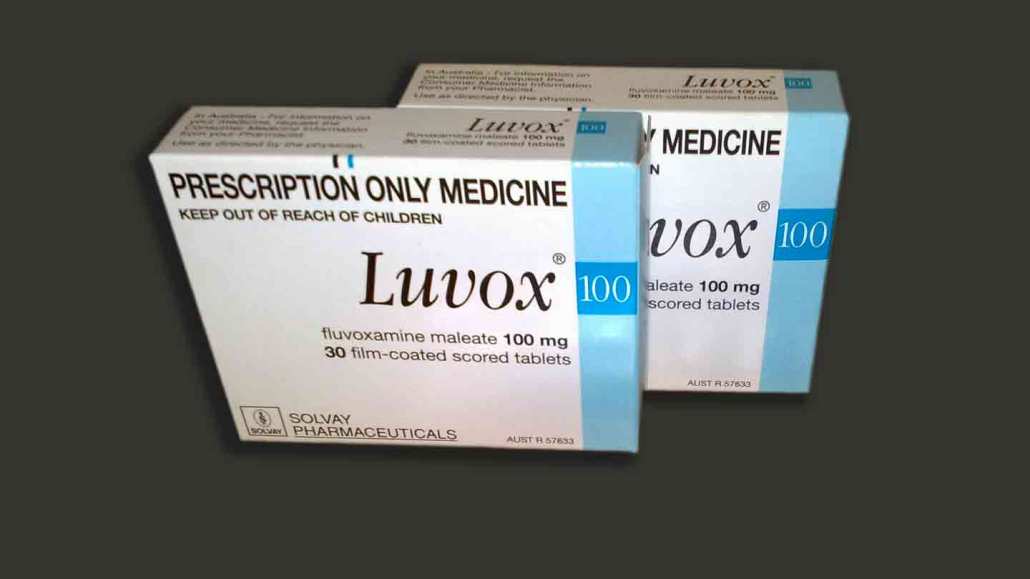
A decades-old antidepressant cut hospitalizations and deaths in COVID-19 outpatients, according to the largest trial to date testing the repurposed drug.
EDITOR182/WIKIMEDIA
An inexpensive, easy-to-take pill could be the next weapon in the arsenal against COVID-19. Taking the antidepressant fluvoxamine within days of showing symptoms of an infection can dramatically cut the risk of hospitalization and death, suggests the largest trial to date of this FDA-approved generic drug as a COVID-19 treatment.
In newly infected COVID-19 patients at high risk of complications, a 10-day course of the antidepressant fluvoxamine cut hospitalizations by two-thirds and reduced deaths by 91 percent in patients who tolerated the medicine, researchers report October 27 in the Lancet Global Health.
Fluvoxamine is commonly prescribed for obsessive-compulsive disorder and acts by increasing levels of the brain chemical serotonin between nerve cells. Aside from those effects, the drug has other biological properties that could quell inflammation triggered by COVID-19, says child psychiatrist Angela Reiersen of Washington University School of Medicine in St. Louis. She stumbled onto the idea to test fluvoxamine as a COVID-19 treatment while scouring papers during her own bout of illness early in the pandemic.
“This is an existing medicine with two to three decades of clinical use — something millions of people have taken,” says David Boulware, an infectious disease physician and researcher at the University of Minnesota Medical School in Minneapolis. “It’s available at every pharmacy in the U.S., and [a 10-day course] costs $10.” By comparison, a five-day course of Merck’s antiviral molnupiravir — another oral drug that can protect people from serious COVID-19 — carries a $700 price tag (SN: 10/1/21).
Boulware was not involved in the latest trial but helped analyze results from a smaller study showing fluvoxamine’s promise as an early COVID-19 treatment. That study tested real-world use of the antidepressant in workers who got infected during a COVID-19 outbreak at a California horse racing track last November (SN: 2/1/21).
The new analysis was conducted as part of TOGETHER — an international collaboration launched last year to test multiple repurposed medications in placebo-controlled experiments at the same time. The study’s adaptive design allows investigators to remove treatments from the study when statistical analyses show the drugs offer no benefit, as has been the case for the antimalarial drug hydroxychloroquine and for ivermectin, a drug commonly used against parasites (SN: 8/2/20).
For the new study, researchers at McMaster University in Hamilton, Canada, partnered with the research clinic Cardresearch in Brazil to recruit 1,497 unvaccinated, high-risk adults in their first week of flu-like symptoms from COVID-19. Patients at 11 clinical sites in Brazil entered the trial between January and August, and received either 100 milligrams of fluvoxamine or placebo pills twice a day for 10 days. Investigators monitored participants for another 28 days after treatment.
In the placebo group, 119 of 756 patients, or 15.7 percent, developed complications requiring hospitalization or more than six hours of emergency care. In comparison, 79 of 741, or 10.7 percent, of fluvoxamine-treated patients got that sick. Taking fluvoxamine cut emergency visits and hospitalization by 32 percent, the trial found.
Among patients who took at least 80 percent of their doses, the benefits were even stronger. About three-fourths of patients fell into that group, with the most common reason for stopping the drug being gastrointestinal complaints. Fluvoxamine cut serious complications in that group by 66 percent and reduced mortality by 91 percent. In the placebo group, 12 patients died, compared with one who received the drug.
Given the latest data and fluvoxamine’s long-standing safety record, Reiersen says, “we believe it should be used in COVID-19 for patients at high risk for morbidity and mortality from complications of the infection.” According to Boulware, the expert panel that develops U.S. COVID-19 treatment guidelines was briefed on the data in mid-September and could decide soon about recommending fluvoxamine as an early treatment.
Sign up for our newsletter
We summarize the week's scientific breakthroughs every Thursday.